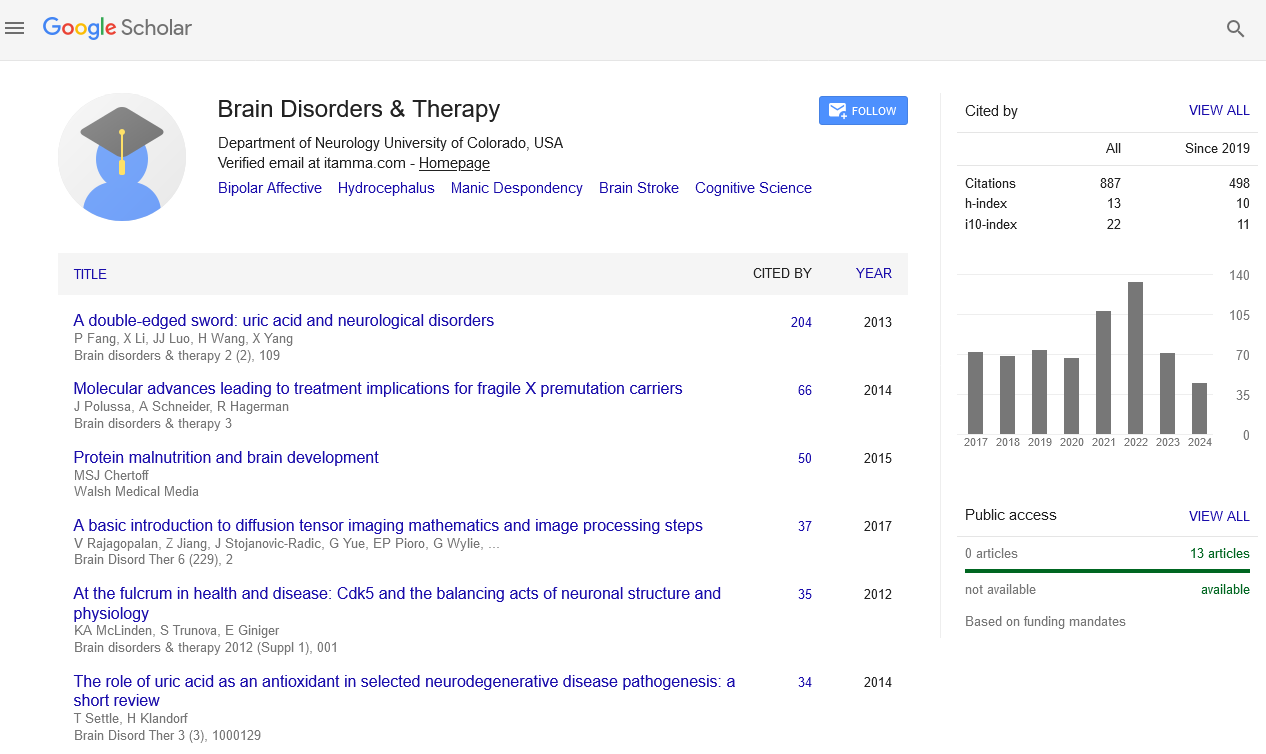
PMC/PubMed Indexed Articles
Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Targeting Neurotoxic N-Terminal Pyroglutamated Abeta (Pgluaò) with inhibitors of Glutaminyl Cyclase (Qc) and Pgluaò-specific antibodies has reached clinical stage
International Conference on Brain Disorders & Therapeutics
August 24-26, 2015 London, UK
Hans-Ulrich Demuth
Fraunhofer Institute for Cell Therapy and Immunology, Germany
Scientific Tracks Abstracts: Brain Disord Ther
Abstract:
Alzheimer�??s disease (AD) is characterized by neuron-loss and neuro-inflammation. Although N-truncated and N-pyroglutamated Aβ-peptides (pGluAβ) are known as prominent constituents of plaques in AD-brain, their importance was overseen and pathways leading to their formation not understood. Because of their abundance, resistance to proteolysis, such N-terminally modified peptides can be important for initiation of pathological cascades leading to AD. Our work uncovers, that N-terminal pGluAβ-formation is catalyzed by QC1. QC-expression is upregulated in the cortex of individuals with AD and correlated with the appearance of pGlu-modified Aβ. Oral application of QC-inhibitors resulted in reduced pGlu3Aβ42 burden, but surprisingly also to the attenuation of the 1.000fold higher amounts of total Aβ in transgenic AD-models1. These observations led to the hypothesis that pGluAβ can seed Aβ-oligomerization by self- and co-aggregation with other monomeric Aβ-species2. Amounts of less than 10nM pGlu3Aβ42 generated cytotoxic oligomers which are over 20 fold more stable than oligomers of �??classical�?� full-length Aβ-peptides. Such mixed pGlu3Aβ42-oligomers propagate their toxic structure in a prion-like manner. Moreover, the neurotoxicity unfolds to be tau-dependent in cell culture as well as in animal models. There, specific neuronal expression of pGluAβ provides in vivo evidence for profound pGluAβ neurotoxicity and gliosis induction2. Hence, a drug development program was entering regulatory testing 2010. Two phase 1: Single (SAD) and multiple ascending dose (MAD) trials of the compound PQ912 in healthy volunteers were conducted. They revealed PQ912 safe and well tolerated. Dose-proportional pharmacokinetics and a strong pharmacodynamic relationship were observed in plasma and CSF justifying studies involving patients. PQ912 is the first QC-inhibitor for treatment of AD since March 2015 in phase 2. Our research concentrates further on posttranslational modifications of Aβ leading to alternative pathways of the turnover of precursor protein APP.
Biography :
Hans-Ulrich Demuth has completed his PhD at the age of 28 and his doctorate of sciences at the age of 36 years from Martin-Luther-University, Halle/S., Germany. He did postdoctoral studies at University of Kansas and the Uppsala Biomedical Research Center. He has inaugurated and co-founded Probiodrug in 1997, the company which has developed the concept of inhibition of Dipeptidylpeptidase-4 as treatment of type-2 diabetes (on the market since 2006). He has published more than 300 papers in reputed journals and has been serving as an editorial board member and reviewer of repute. In 2013 he founded the Fraunhofer-IZI department MWT.