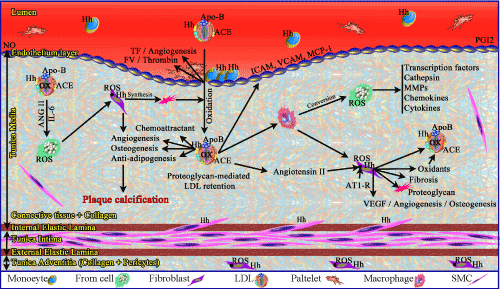
Figure 2: Pathogenesis of atherosclerotic plaque. Normal arteriole is composed of endothelium and the subendothelium or BM. An intact endothelium produces PGI2
and NO, which retain hemostatic components in an inactive form and inhibits monocyte and platelet aggregation. An injured endothelium releases Vwf and TF to start
coagulation and exposes BM to the circulation to stimulate circulatory platelets and monocytes to the place of injury. This forms a plaque for repairing the injury and
prevention of blood loss. BM composed of a collagen and elastic fibers as well as SMCs that provide support around vessels. IEL and EEL line two sides of tunica
intima and separate SMCs from the media and adventitial layers. In normal state, intact IEL prevents migration of SMCs into media space, however in atherosclerotic
state, the intima layer growth and macrophage-cathepsis K degradates IEL. This leads to migration of SMCs from intima to media for release of collagen and plaque
stabilization. Moreover, pericyte myofibroblasts in atherosclerotic plaque is able to differentiate into osteoblasts and chondrocytes, adventitial myofibrolasts, calcifying
vascular cells, valvular interstitial cells and foam cell macrophages, which release cytokines, chemokines and transcription factors. They, as well as fibroblasts, express
enzymes generating oxidants (ROS). The processe of plaque formaton and inflammation starts by trapping of LDL containing ACE and Hedgehog (Hh) molecules
and binding of them to the proteoglycan (lipid core). Hedgehog molecules function for T cell activation and cytokine production and SMC proliferation and migration
into plaque. It functions in regenerative processes including chemotaxis, revascularization, anti-adipogenesis and osteogenesis that are crucial in plaque calcification.
Thereafter, LDL follows the oxidative modification processes and forms ox-LDL, which is taken up by macrophages and form foam cells. IL-6 and Ang2 produced by
ox-LDL activate foam cell. Ox-LDL also enhances expression of the adhesion and chemoattractant molecules (VCAM-1, ICAM-1, MCP-1) by endothelial cells, which
lead to leukocyte recruitment to the plaque. ACE of LDL converts Ang I to Ang II, which activates (AT1-R) on fibroblasts and produce both proteoglycans and oxidants
by fibrobelast/myofibroblasts. Hh particle is assumed to be present on the monocytes, LDL, fibroblasts and SMCs. TF and factor V are assumed to have a role in
plaque calcification (Dashty, et al, BioEssays, 2012).
BM: Basement Membrane; PGI2: Prostacyclin; NO: Nitric Oxide; vWF: vonwilebrand Factor; TF: Tissue Factor; SMC: Smooth Muscle Cells; IEL: Internal Elastic
Laminas; EEL: External Elastic Laminas; ROS: Reactive Oxygen Species; Ox-LDL: Oxidized-Low Density Lipoprotein; IL: Interleukin; Ang: Angiopoietin; VCAM:
Vascular Cell Adhesion Protein; ICAM: Intercellular Adhesion Molecule; MCP: Monocyte Chemoattractant Protein; ECs: Endothelial Cells. ACE: Angiotensin-Converting
Enzyme; AT1-R: Angiotensin II Type I Receptors. |