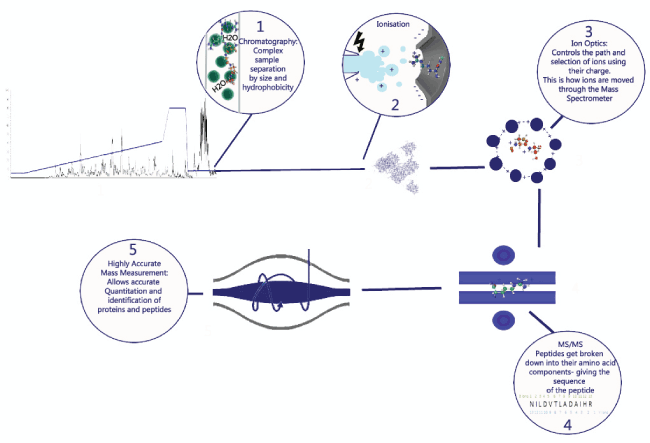
1. Peptides in solution are first separated by high pressure liquid reversephase chromatography, which separates peptides by size and hydrophobicity.
2. The peptides elute from the column at an electrospray tip, where a charge is applied to generate positively charged ions.
3. The resulting ions can now be controlled due to their positive charge and separated by mass. This is done throughout the mass spectrometer using resonating magnetic frequencies, which allow selection of ions according to their mass and charge.
4. Selected ions can also be collided with an inert gas to fragment them, giving a characteristic fragmentation spectrum from which the amino acid sequence of that peptide can be deduced.
5. Highly accurate mass measurement, in conjunction with rapid speed of analysis, makes LC-MS/MS a useful tool in the analysis of complex protein and peptide mixtures.