 |
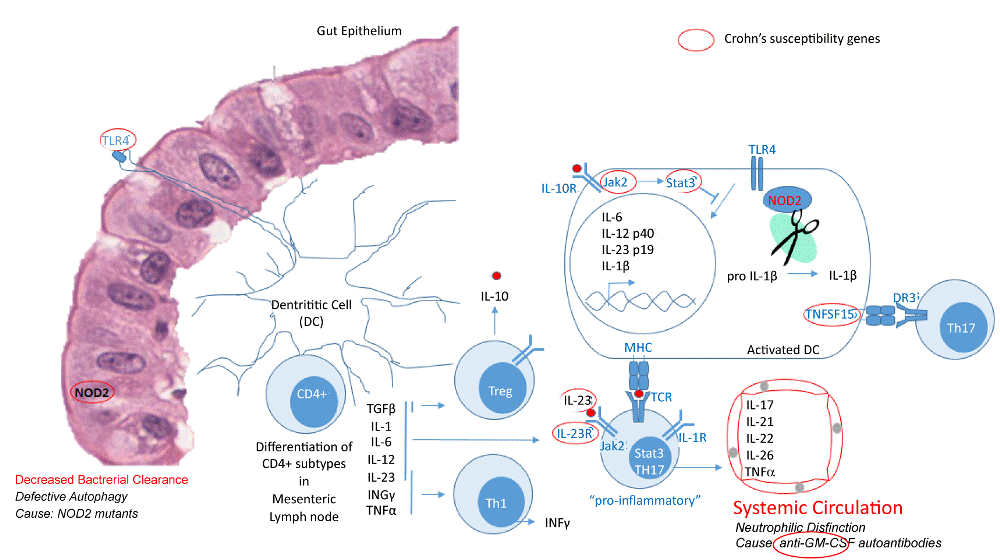
| Figure 1: Gene polymorphisms and autoantibodies associated with Crohn’s disease are involved in inflammatory (innate) and adaptive immune responses. The production of inflammatory cells depend on growth factors as in the case of GM-CSF for neutrophils. Anti-GM-CSF antibodies suppress the production of neutrophils, hence the decreased bacterial clearance leading to decreased epithelial integrity. Increased levels of antibodies against specific bacterial peptides are associated with an increased risk of severe Crohn's disease. The most common mutants in Crohn’s disease, occur in autophagy related genes (susceptibility genes) such as NOD2, which give a prolonged inflammatory response. In addition, NOD2 is involved in the formation of a TLR4 stimulated inflammasome in dendritic cells, that promotes cleavage of pro-Ilβ to the active cytokine IL1β, and the production of IL-23 (another susceptibility gene). Other polymorphisms associated with Crohn’s disease involve genes that are important for the differentiation of CD4+ cells into special T cell subtypes and T cell function. Dysregulation of IL23R, IL12B, Jak2, Stat3, CCR6 and TNFSF15 through functional polymorphisms, interferes with the pro-inflammatory response involving Th17 cells which has been shown to have an important role in intestinal inflammation. In addition, Stat3 was also shown to regulate the inhibitory signals initiated by IL-10 secreted by regulatory T cells (Treg). Hence, autophagy response and Th17 signals are important mediators of inflammation in Crohn’s disease. |