 |
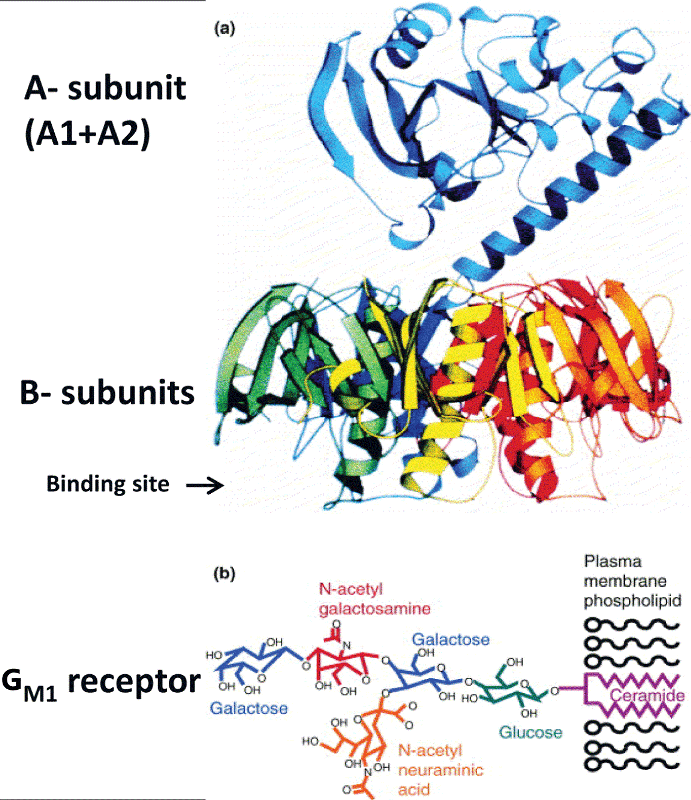
| Figure 1: The molecular structure of HLT-I [similar to HLT-II except for the type of receptor (55)]. The family of LT and LT-II toxins contains an enzymatically active A1 domain and the A2 domain that allows for non-covalent interaction of the A subunit and the B-subunit pentamer to give holotoxin. Importantly, this molecular structure can be exploited to engineer chimeras with other proteins or epitopes. In the native structure of the holotoxin, A1 points away from the A2-B pentamer complex. Inserts can be engineered at the N terminus of A2 subunit to replace A1 (responsible for toxic activity) with the target of interest and thus still allow appropriate assembly. Such chimeric proteins assemble, do not interfere with the immunological function of the binding subunit of the toxins, LT-B, and are non-toxic. Alternatively, fusions can be made directly to the C-terminus of the B subunits in the absence of the A subunits (A1 and A2). The binding site of the B-subunits cross-links five moieties of GM1 via interaction with their pentasaccharide moieties exposed upon the lymphocyte cell surface. GM1 is integrated into the plasma membrane through its hydrophobic ceramide moiety. Diagram: a) A1/A2-B5 holotoxin; b) GM1 receptor. |