 |
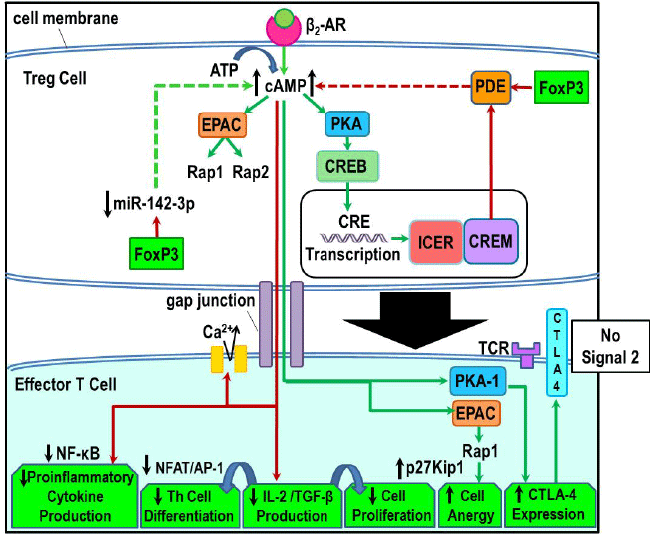
| Figure 13: Cyclic AMP-mediated regulation of Th cells by Treg cells. Treg cells predominantly express β2-ARs on their cell surface (top center). Ligand activation of these receptors stimulates cAMP production, which induces a cascade of PKA-dependent cAMP response element-binding protein (CREB) phosphorylation and subsequent transcription of ICER and CREM. CREM inhibits PDE activity, which prevents the breakdown of cAMP, and therefore promotes higher intracellular cAMP in Treg cells (upper right side). Elevated cAMP also increases EPAC activity which acts to increase Rap 1 and Rap 2 GTPase activity. Rap1/2 is implicated in promoting Treg cell regulation of Th cells. FoxP3, a phenotypic marker of Treg cells, regulate intracellular cAMP concentrations by at least two mechanisms. FoxP3 inhibits PDE activity, promoting greater intracellular levels of cAMP. FoxP3 also down-regulates miR- 142-3p (upper left side) to keep the AC/9/cAMP pathway active, a role that is opposite to that played by this microRNA in other Th cell subsets. The cAMP generated in Treg cells and transferred to Th cells via gap junctions (center of figure) can exert both suppressive and enhancing actions on Th cells. These suppressive and enhancing effects are illustrated in the lower left and right of the figure, respectively. Greater cAMP concentrations inhibit proinflammatory cytokine production by suppressing NF-κB and Th cell differentiation by blocking IL-2 and TGF-β production, via reduced NFAT/AP-1 binding to DNA binding sites (lower right). Lower influx of calcium is induced by cAMP, which negatively regulates NFAT protein kinase. Increased cAMP concentrations in Th cells induce PKA-dependent CTLA-4 upregulation, a negative regulator of contact-dependent T cell receptor (TCR) activation (lower right), preventing binding of CD28 (signal 2) necessary for T cell activation. EPAC transduces cellular actions of cAMP in Th cells that promote anergy via the activation of Rap1 (lower right). (Green arrows, excitatory; red arrows, inhibitory). |