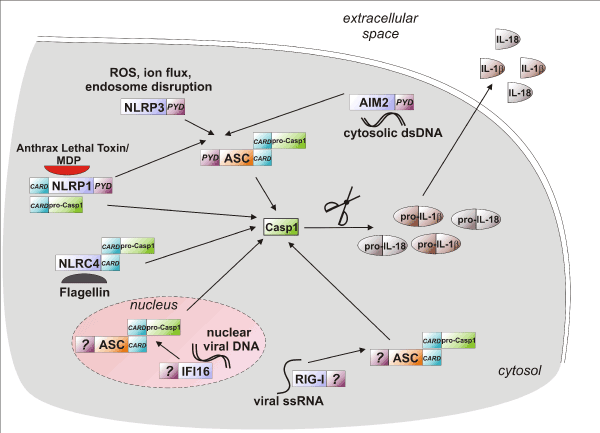
ASC, apoptosis-associated speck-like protein containing a caspase recruitment domain; CARD, caspase activation and recruitment domain; Casp1, enzymatically active caspase-1; dsDNA, double stranded deoxyribonucleic acid; IFI16, interferon gamma-inducible protein 16; IL-18, interleukin-18; IL-1β, interleukin-1β; MDP, muramyl dipeptide; NLRC4, NLR family CARD domain-containing protein 4; NLRP1, NOD-like receptor family, pyrin domain containing 1; NLRP3, NOD-like receptor family, pyrin domain containing 3; pro-Casp1, procaspase-1; PYD, pyrin domain; RIG-I, retinoic acid inducible gene-I; ROS, reactive oxygen species; ssRNA, single stranded ribonucleic acid.