|
| Michelle R. Yagoda MD1* and Eugene H Gans PhD2 |
| 1Facial Plastic Surgeon, Private Practice, and Associate Attending Surgeon, Manhattan Eye, Ear and Throat Hospital, Lenox Hill Hospital, North Shore-LIJ Health System, New York, USA |
| 2President and Pharmaceutical Consultant, Hastings Senior Associates, New York, USA |
| Corresponding Author : |
Michelle R. Yagoda MD
1025 Fifth Avenue, New York 10028, USA
Tel: 212-434-1210
Fax: 212-535-8155
E-mail: info@dryagoda.com |
| |
| Received March 05, 2014; Accepted April 26, 2014; Published April 28, 2014 |
| |
|
Citation: Yagoda MR, Gans EH (2012) A Nutritional Supplement Formulated with Peptides, Lipids, Collagen and Hyaluronic Acid Optimizes Key Aspects of Physical Appearance in Nails, Hair and Skin. J Nutr Food Sci S5:002. doi: |
| |
|
Copyright: © 2014 Yagoda MR, et al. This is an open-access article distributed
under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. |
| |
Related article at
 Pubmed Pubmed  Scholar Google Scholar Google |
| |
Visit for more related articles at
 Journal of Nutrition & Food Sciences Journal of Nutrition & Food Sciences |
| |
| Keywords |
| |
| Absorption; Aging; Beauty; Nutrition; Physiognomy;
Supplementation |
| |
| Introduction |
| |
| Symptoms of dryness in the skin, hair and nails increase with age
and with certain medical, environmental and physiologic conditions—
affecting up to 40% of people over 18 years old [1-5]. Topical therapies
can be limited by the skin’s inherent barrier function, and simply
ingesting a balanced and varied diet may not be enough to prevent
nutritional deficiencies. |
| |
| According to the Center for Disease Control, nutritional deficiencies
could impact as many as 33% of the population as a result of altered
gastrointestinal absorption [6]. |
| |
| In as early as 1968, Bender noted the effect of age on absorption,
[7] and others noted an increase in amino acids following graded
dietary intake of whey protein [8]. A deficiency in dietary protein was
determined to cause reduced cellular turnover, decreased DNA repair,
fragmentation of collagen and elastin, and the resultant skin wrinkling
and laxity [9]. In addition, dietary proteins were noted to control lipid
metabolism [10] and affect intercellular water retention in the stratum
corneum [11]. As a result, a decrease in the level of skin lipids was noted
to cause skin dryness [9]. |
| |
| Hydrolyzed collagen was shown to stimulate chondrocytes in vitro,
[12] in human serum, [13] and in animal models [14-16]. Additionally,
dietary supplementation with hyaluronic acid was shown to increase
skin moisture content and smoothness, and decrease wrinkles [17]. |
| |
| Beauty nutritional supplementation in a powder formulation
containing peptides and lipids—the building blocks necessary for healthy skin, hair and nails—as well as hyaluronic acid and collagen,
was clinically evaluated to determine its benefits for the skin, hair and
nails. |
| |
| Materials and Methods |
| |
| Inclusion/Exclusion criteria |
| |
| A total of 54 women were selected for two trials of the supplement
formula, one lasting 8 weeks, the other 60 weeks. Ages of study
participants ranged from 26 to 68 years old (mean=49). To qualify for
participation, each individual was required to exhibit one or more severe
or moderately-severe skin, hair or nail problem in one or more areas
(dullness, dryness, brittleness, unmanageability of the hair; dryness,
cracking, breaking and splitting of the nails; and, dryness, roughness,
cracking, peeling and scaling of the skin), or mild to moderate
problem in two or more areas, where problem severity was rated as
follows: 0=none; 1-2=mild; 3-4=mild-to-moderate; 5-6=moderate;
7-8=moderately-severe; 9-10=severe. |
| |
| Precluded from the study was anyone with a dermatological diagnosis of skin, nail or hair disorder, anyone pregnant, or anyone
with an allergy to soy or milk products. Participants were restricted
from using additional topical or medically prescribed hair, skin or
nail products during the trial period with the exception of overthe-
counter, non-medicated shampoo and conditioner and nonsalicylated
or glycolated cleanser. Participants were also restricted
from using additional nutritional supplements; any topical hair
strengthening and/or hair growth products; and topical and/or
internal nail products. All patients were instructed to report any
deviation from the above restrictions, as well as any weight loss or
illness during the trial period. |
| |
| Blinding and dosing |
| |
| Each of the 54 participants meeting the inclusion criteria was
randomly assigned into either a placebo or active group for one of the
two studies. Both the 8-week study and the 60 week cross-over study
were double-blinded studies. In the 8-week study, participants were
randomized at a ratio of 2:1, with double the number of participants
assigned to the active group. |
| |
| Active and placebo products were independently labeled by subject
number, so that neither the investigator nor the subject knew which
subjects were getting which test materials. Both active and placebo
powders were packaged in identical 1-pound cans, with aluminum lids
and aluminum protected, cardboard sides. Each can had a pull-tab to
open the can and a plastic lid to close it. Each can contained a precalibrated
scoop to measure the 20-gram daily dose. |
| |
| (1) Active=Protein/Oil in a Carbohydrate containing vehicle. The
formula contained safflower and canola oils, both unsaturated, as the
lipid sources; hydrolyzed soy, whey protein concentrate and collagen
as the protein sources; and maltodextrins as the carbohydrate sources.
The powder was flavored with vanilla and off-white in color. There were
approximately 5 grams of lipid and 5 grams of protein in each 20-gram
dose. Additional ingredients included calcium, lecithin, magnesium,
vitamin E, zinc, copper, manganese, biotin and riboflavin. |
| |
| (2) Placebo=The Carbohydrate vehicle alone was matched to
the Active Protein/Oil/Carbohydrate powder in color and flavor. Additional ingredients included calcium, lecithin, magnesium, vitamin
E, zinc, copper, manganese, biotin and riboflavin. |
| |
| Each participant was instructed to take 20 grams of powder every
morning before breakfast, 7 days each week, by filling the provided 20
gram scoop with the respective powder, dispersing it in 3-4 ounces of
water, and then swallowing the dispersion all at once. |
| |
| Evaluation schedule |
| |
| In the 8-week study, there were 34 participants with 23 randomly
placed into the active group, and 11 in the placebo group. Evaluations
were performed at Baseline and at weeks 3, 4, 6 and 8. |
| |
| In the 60-week double-blinded cross-over study, there were 20
participants enrolled with 10 randomly placed in the active group, and
10 in the placebo group. Evaluations were performed at Baseline and at
week 4, 8, 14, 20, 26, 34, 38, 44, 52, and 60. 8 of the 10 in the active group
completed the study as two were relocated for work. In this cross-over
study, one group of 10 was given active for 52 weeks, and then switched
to placebo for an additional 8 weeks. The other group of 10 was given
placebo for 52 weeks, and then switched to active for an additional 8
weeks. |
| |
| Rating |
| |
| The skin, hair and nails of each participant were evaluated on
each rating day by two trained clinical investigators: Elias Packman,
Sc.D., Professor of Pharmacology, Philadelphia College of Pharmacy
and Science, and Ruth Jeffkin, RN, of the Institute for Applie1d
Pharmaceutical Research, Limited. |
| |
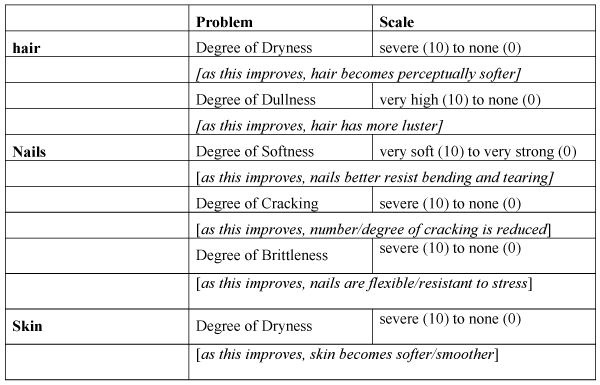
| Clinical investigators rated 6 symptoms: dullness and dryness of
the hair; softness, cracking and brittleness of the nails; and dryness of
the skin. They used a 10-point rating scale where 0=none; 1-2=mild;
3-4=mild-to-moderate; 5-6=moderate; 7-8=moderately-severe;
9-10=severe (Figure 1). |
| |
| Separately, all participants rated themselves using the same
rating scale. Neither the investigators nor the participants knew
the other’s score. In cases of non-concurrence, the lower of the two ratings was used— i.e., the smaller benefit/change. Both investigators
and participants were trained to use the rating scale in Table 1 in a
standardized and reproducible fashion using published methods [18-
20]. |
| |
| Results |
| |
| 54 participants were enrolled in the studies and 52 participants
completed the full trials. No participants were disqualified from the
study, but two withdrew from the double-blinded cross-over study
when they were relocated for employment purposes. |
| |
| No significant adverse effects were reported. One participant
reported minimal gas and bloating on the first day of product
consumption, but there were no other reported incidences by this
participant associated with subsequent ingestion, or by any other
participant. |
| |
| 8 week study |
| |
| In the 8-week study, the severity of nail cracking and brittleness
decreased by 79.1% (p<.001) and 80.7% (p<.001), respectively, in
the active group, while those in the placebo group experienced a
respective 62.3% (p=.001) and 68.5% (p=.002) improvement over the
same 8-week time period. Nail softness, cracking and brittleness, all
statistically and significantly decreased from baseline over the course
of 8 weeks, with a corresponding increase in strength. In the active
group, the severity of nail softness decreased by 43.2% (p<.001), while those in the placebo group experienced only 23.6% (p=.03)
improvement (Table 1). |
| |
| Hair dryness and dullness at baseline statistically and significantly
decreased in each group over the course of the study, with a
corresponding increase in softness and luster. The severity of dullness
decreased by 48.4% (p<.001) in those taking active product, but only
22.7% (p=.006) in those taking placebo. The active group experienced
a 47.3% (p<.001) decrease in the severity of dryness, while those in the
placebo group experienced only a 17.8% (p=.013) improvement over
the same 8-week time period. |
| |
| Skin dryness at baseline decreased in each group over 8 weeks, with
a corresponding increase in softness. In the active group, the severity of
dryness decreased by 21.7% (p=.004) while those in the placebo group
experienced a 19.5% (p=.009) improvement over the same 8-week time
period. |
| |
| In participants taking active who started with moderately-severe to
severe symptoms, 95% (p<.001) of those saw significant improvement
in hair dryness and dullness within 8 weeks, 85% (p=.001) in nail
splitting and peeling, 65% (p<.001) in nail hardness, and 60% (p<.004)
in skin dryness. When starting with mild to moderate symptoms, those
taking active also saw significant results in hair dryness and dullness
and nail splitting, peeling and brittleness, and skin dryness, but to a
lesser degree. While improvement was often seen with the placebo,
it was to a significantly lesser degree than those improvements seen
with the active. We believe that there was a design error in creating the placebo in that it was not completely inactive. In fact, the carbohydrate
vehicle in the placebo likely provided unexpected nutritional benefits
(Table 2). |
| |
| Double-blinded 60-week cross-over study |
| |
| Tables 3 and 4 pertain to the double-blinded 60-week cross-over
study. The participants in the active group received 52 weeks of active
and then were switched to placebo for an additional 8 weeks. The
participants in the placebo group received 52 weeks of placebo and then
were switched to active for an additional 8 weeks. |
| |
| Table 3 shows that, when starting with moderately-severe to
severe symptoms, participants taking active experienced significant
improvement in hair dryness and dullness, nail hardness and peeling
at various times throughout the course of the 12-month study. After
the first 8 weeks of treatment, 80% of participants saw improvement in
hair dryness, and 90% saw improvement in both hair dullness and nail
peeling. After 6 months of treatment, 88% saw improvement in hair
dryness and nail peeling, while 100% saw improvement in hair dullness.
After 9 months, 100% of participants saw improvement in hair dryness,
hair dullness, and nail peeling. |
| |
| During the course of the 12-month study, at least 75% of participants
experienced significant improvement in nail splitting, and at least 50-
75% experienced significant improvement in skin dryness. |
| |
| The effect of the cross-over is noted in Table 4. Those in the active
group experienced a reversal of effects in hair dryness and dullness, nail
hardness, splitting and peeling when switched to placebo, and those in
the former placebo group showed improvement in those areas when
switched to active. Since participants were unwilling to refrain from
using sunblock during the summer months of the study, evaluation of
their skin was only performed for the first nine months. As a result, the
effects of cross-over on skin dryness could not be made. |
| |
| While participants in the 60-week cross-over study clearly
experienced significant benefits related to hair dryness and dullness, nail
hardness, splitting and peeling, and skin dryness when taking active,
the amount of improvement represented in Table 4 reflects a cohort
with a mixed baseline severity of symptoms. Because inclusion criteria
only required participants to have a severe or moderately-severe skin,
hair or nail problem in one or more areas (dullness, dryness, brittleness,
unmanageability of the hair; dryness, cracking, breaking and splitting
of the nails; and, dryness, roughness, cracking, peeling and scaling of
the skin), or mild to moderate problems in two or more areas, some participants may have had no problem in one or more areas initially. |
| |
| Discussion |
| |
| Participants who ingested 20 grams of nutritional supplement
daily experienced a significant improvement in their nails, hair and
skin. Statistically significant benefits, albeit to a much lesser degree,
were also often noted with the placebo. We believe this was because
the placebo was not inactive: there were unexpected nutritional benefits
from the carbohydrate-containing vehicle in the placebo. Overall, the
greatest improvements were seen in participants in the active group,
who started out with more severe problems. |
| |
| The authors believe that nutritional supplementation with a
special combination of dosage bioavailability and amount (20 grams
of powder), dosing interval (once daily) and consumption period (30
minutes or less) provides benefit over a healthy diet and is critical for
a supplement’s rapid and intense results. Earlier studies by the authors
showed that decreasing the dosage interval (i.e. 10 grams twice/day) or
increasing the consumption time (more than 30 minutes) reduced the
effectiveness both in terms of magnitude of results and onset of change.
The authors theorize that as the dosing interval is increased and/or the
consumption time is prolonged, the supplement’s absorption is delayed
and begins to mimic that which occurs with consumption of a typical
diet. Note that pills, tablets and bars may have binders and additives
that may delay and/or block absorption, while powder formulations
lead to immediate bioavailability. With consumption of a typical
diet, nutrients become bioavailable over a 4-6 hour digestion period,
and may be “rationed” with only some allotted for synthesis, and the
remainder set aside for repair. The authors postulate that nutritional
supplementation provides additional bioavailable nutrients over a short
interval, allowing the body to jumpstart its own biochemical synthesis
pathway to increase moisture retention in the nails (decreasing splitting,
dryness, breaking and peeling); to strengthen bonds in hair (reducing
breakage and split ends, and increasing luster); and, to enrich the lipid
content of the skin (enhancing softness and smoothness). |
| |
| The implications of the study findings are clear: nutritional
supplements with peptides, lipids, collagen and hyaluronic acid provide
measurable and significant benefits and are an effective, important and
perhaps even necessary component of a comprehensive nail care, hair
care and skin care regimen. |
| |
| The cosmetic and dermatological literature has a great many
reports, using qualitative assessments, on the effects of topically applied
agents on cosmetic properties of hair skin or nails. But this study is
one of the first to report on the effects of orally ingested agents (not
topically applied agents) on the important physical properties of the
hair, skin and nails. Of further value, is the use here of quantitative
scoring, rather than qualitative measures or opinions, to assess the
validity of these changes. This report demonstrates, perhaps for the first
time qualitatively, that a properly formulated nutritional supplement
can have significant, positive, measurable effects upon the physical
properties of hair, skin and nails. |
| |
| Limitations of the study were the small study size, and the likelihood
that the carbohydrate vehicle in the placebo may have rendered it active
and provided unanticipated nutritional benefits. In addition, it was not
possible to fully evaluate the effect of the nutritional supplement on
the skin during the summer months of the 60-week crossover study
because it proved to be unreasonable to expect participants to refrain
from using sunblock during this time. |
| |
| Possibilities for future research include the evaluation of the effects of the carbohydrate vehicle alone, and the investigation of
supplementation as an effective treatment for those with diseases
of the skin, hair and nails. It would be worthwhile to investigate the
bioavailability of peptides and lipids in the bloodstream after the
ingestion of supplement in attempt to correlate benefits on a biochemical
basis, in attempt to uncover the supplement’s mechanism of action. |
| |
| Conclusion |
| |
| Beauty nutritional supplementation with peptides and lipids-the
building blocks necessary for healthy skin, hair and nails —as well
as hyaluronic acid and collagen, all in highly bioavailable powder
formulation provides rapid, measurably significant, and long-lasting
results. As a result, it should be considered a critical component of a
comprehensive hair, nail and skin care regimen. |
| |
| Acknowledgements |
| |
| Dr. Eugene Gans wishes to acknowledge the support of Dr. Elias Packman,
Philadelphia, PA, and others, in developing the objective techniques used to
assess changes in the physical characteristics of hair, skin and nails. |
| |
| Author Disclosure Statement |
| |
| Michelle R. Yagoda, M.D, and Eugene H. Gans, Ph.D., authors of this
manuscript, are co-creators of this nutritional supplement formula, and owners of
Opus Skincare, LLC, and the company that manufactures this patented beauty
nutritional supplement. No agency was used to assist in scientific writing. No grant
support was received. |
| |
| References |
| |
- Hashizume H (2004) Skin aging and dry skin. J Derm 31: 603-609.
- Misery L, Sibaud V, Merial-Kieny C, Taieb C (2011) Sensitive skin in the American population: prevalence, clinical data, and the role of the dermatologist. Int J Dermatol 50: 961-967.
- Misery L, Boussetta S, Nocera T, Perez-Cullell N, Taieb C (2009) Sensitive skin in Europe. J EurAcadDermatolVenereol 23: 376-381.
- Paul C, Maumus-Robert S, Mazereeuw-Hautier J, Guyen CN, Saudez X, et al.(2011) Prevalence and risk factors for xerosis in the elderly:a cross-sectional epidemiological study in primary care. Dermatol 223: 260-265.
- Halvorsen JA, Braae OA, Thoresen M, Holm JO, Bjertness E (2008) Comparison of self-reported skin complaints with objective skin signs among adolescents. ActaDermVenereol 88: 573-577.
- U.S. Centers for Disease Control and Prevention (2012) Second National Report on Biochemical Indicators of Diet and Nutrition in the U.S. Population. Atlanta, GA; National Center for Environmental Health.
- Bender AD (1968) Effect of age on intestinal absorption: implications for drug absorption in the elderly. J Am GeriatrSoc 16: 1331-1339.
- Pennings B, Groen B, de Lange A, Gijsen AP, Zorenc AH, et al. (2012) Amino acid absorption and subsequent muscle protein accretion following graded intake of whey protein in elderly men. Am J PhysioEndocrinolMetabol 30: 992-999.
- Chenoff R (2005) Micronutrient requirements in older women.Am J ClinNutr 8: 1240S- 1245S.
- Jahan-Mihan A, Luhovyy B, El Khoury H (2011) Dietary proteins as determinants of metabolic and physiologic functions of the gastrointestinal tract. Nutrients 3: 574-603.
- Imokawa G, Akasaki S, Minematsu Y, Kawai M (1989) Importance of intercellular lipids in water-retention properties of the stratum corneum: induction and recovery study of surfactant dry skin. Arch Dermatol Res 281:45-51.
- Oesser S, Seifert J (2003) Stimulation of type II collagen biosynthesis and secretion in bovine chondrocytes cultured with degraded collagen. Cell Tissue Res 311: 393-399.
- Iwai K, Hasegawa T, Taguchi Y, Morimatsu F, Sato K, et al.(2005) Identification of the food-derived collagen peptides in human blood after oral ingestion of gelatin hydrolysates. J Agric Food Chem 53: 6531-6536.
- Matsuda N, Koyama Y, Hosaka Y, Ueda H, Watanabe T, et al. (2006) Effects of ingestion of collagen peptide on collagen fibrils and glycosaminoglycans in the dermis.J Nutri Sci Vitaminol 52: 211-215.
- Ohara H, Iida H, Ito K, Takeuchi Y, Nomura Y (2010) Effects of Pro-Hyp, a collagen hydrolysate-derived peptide, on hyaluronic acid synthesis using in vitro cultured synovium cells and oral ingestion of collagen hydrolysates in a guinea pig model.Biosci, BiotechnolBiochem 74: 2096-2099.
- Zhuang Y, Hou H, Zhao X, Zhang Z, Li B (2009) Effects of collagen and collagen hydrolysate from jellyfish (Rhopilemaesculentum) on mice skin photoaging induced by UV radiation. J Food Sci 74: 183-188.
- Sato T, Sakamoto W, Odanaka W, Yoshida K, Urishibata O (2002) Clinical effects of dietary hyaluronic acid on dry, rough skin. Aesthetic Dermatol 12: 109-120.
- Akazaki S, Nakagawa H, Kazama H, Osanai O, Kawai M, et al. (2002) Age-related changes in skin wrinkles assessed by a novel three dimensional morphometric analysis. Br J Dermatol 147: 689-698.
- Packman EW, Gans EH (1978) The panel study as a scientifically controlled investigation: moisturizers and superficial facial lines. J Society Cosmet Chemists 29: 91-98.
- Packman EW, Gans EH (1978) Topical moisturizers: quantification of their effect of superficial facial lines. J Society Cosmet Chemists 29: 79-90.
|
| |
References
|
- Hashizume H (2004) Skin aging and dry skin. J Derm 31: 603-609.
- Misery L, Sibaud V, Merial-Kieny C, Taieb C (2011) Sensitive skin in the American population: prevalence, clinical data, and the role of the dermatologist. Int J Dermatol 50: 961-967.
- Misery L, Boussetta S, Nocera T, Perez-Cullell N, Taieb C (2009) Sensitive skin in Europe. J EurAcadDermatolVenereol 23: 376-381.
- Paul C, Maumus-Robert S, Mazereeuw-Hautier J, Guyen CN, Saudez X, et al.(2011) Prevalence and risk factors for xerosis in the elderly:a cross-sectional epidemiological study in primary care. Dermatol 223: 260-265.
- Halvorsen JA, Braae OA, Thoresen M, Holm JO, Bjertness E (2008) Comparison of self-reported skin complaints with objective skin signs among adolescents. ActaDermVenereol 88: 573-577.
- U.S. Centers for Disease Control and Prevention (2012) Second National Report on Biochemical Indicators of Diet and Nutrition in the U.S. Population. Atlanta, GA; National Center for Environmental Health.
- Bender AD (1968) Effect of age on intestinal absorption: implications for drug absorption in the elderly. J Am GeriatrSoc 16: 1331-1339.
- Pennings B, Groen B, de Lange A, Gijsen AP, Zorenc AH, et al. (2012) Amino acid absorption and subsequent muscle protein accretion following graded intake of whey protein in elderly men. Am J PhysioEndocrinolMetabol 30: 992-999.
- Chenoff R (2005) Micronutrient requirements in older women.Am J ClinNutr 8: 1240S- 1245S.
- Jahan-Mihan A, Luhovyy B, El Khoury H (2011) Dietary proteins as determinants of metabolic and physiologic functions of the gastrointestinal tract. Nutrients 3: 574-603.
- Imokawa G, Akasaki S, Minematsu Y, Kawai M (1989) Importance of intercellular lipids in water-retention properties of the stratum corneum: induction and recovery study of surfactant dry skin. Arch Dermatol Res 281:45-51.
- Oesser S, Seifert J (2003) Stimulation of type II collagen biosynthesis and secretion in bovine chondrocytes cultured with degraded collagen. Cell Tissue Res 311: 393-399.
- Iwai K, Hasegawa T, Taguchi Y, Morimatsu F, Sato K, et al.(2005) Identification of the food-derived collagen peptides in human blood after oral ingestion of gelatin hydrolysates. J Agric Food Chem 53: 6531-6536.
- Matsuda N, Koyama Y, Hosaka Y, Ueda H, Watanabe T, et al. (2006) Effects of ingestion of collagen peptide on collagen fibrils and glycosaminoglycans in the dermis.J Nutri Sci Vitaminol 52: 211-215.
- Ohara H, Iida H, Ito K, Takeuchi Y, Nomura Y (2010) Effects of Pro-Hyp, a collagen hydrolysate-derived peptide, on hyaluronic acid synthesis using in vitro cultured synovium cells and oral ingestion of collagen hydrolysates in a guinea pig model.Biosci, BiotechnolBiochem 74: 2096-2099.
- Zhuang Y, Hou H, Zhao X, Zhang Z, Li B (2009) Effects of collagen and collagen hydrolysate from jellyfish (Rhopilemaesculentum) on mice skin photoaging induced by UV radiation. J Food Sci 74: 183-188.
- Sato T, Sakamoto W, Odanaka W, Yoshida K, Urishibata O (2002) Clinical effects of dietary hyaluronic acid on dry, rough skin. Aesthetic Dermatol 12: 109-120.
- Akazaki S, Nakagawa H, Kazama H, Osanai O, Kawai M, et al. (2002) Age-related changes in skin wrinkles assessed by a novel three dimensional morphometric analysis. Br J Dermatol 147: 689-698.
- Packman EW, Gans EH (1978) The panel study as a scientifically controlled investigation: moisturizers and superficial facial lines. J Society Cosmet Chemists 29: 91-98.
- Packman EW, Gans EH (1978) Topical moisturizers: quantification of their effect of superficial facial lines. J Society Cosmet Chemists 29: 79-90.
|
Tables at a glance
 | |
 | |
 | |
 |
| Table 1 | |
Table 2 | |
Table 3 | |
Table 4 |
Figures at a glance
 |
| Figure 1 |
|
|
|