PMC/PubMed Indexed Articles
Indexed In
- Open J Gate
- Genamics JournalSeek
- JournalTOCs
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
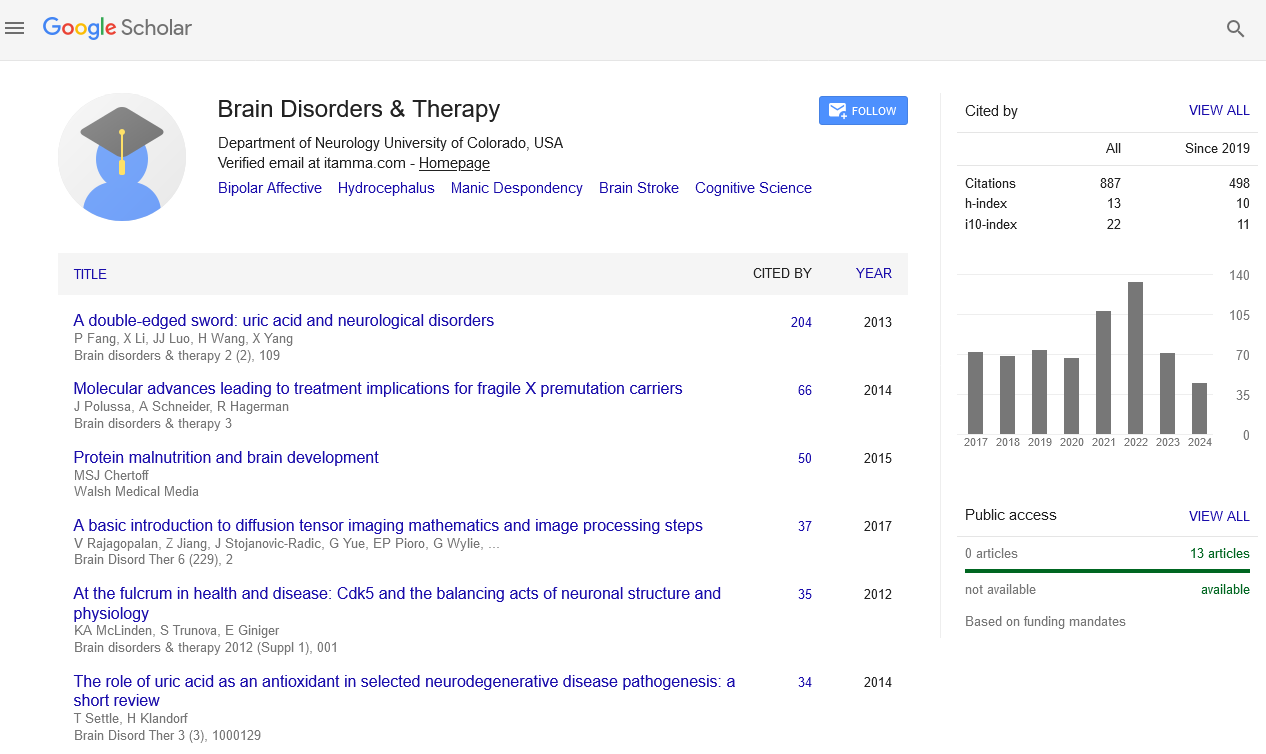
Abstract
Effects of Intracerebroventricular Sugammadex Administration on Central Nervous System in Rats
Hale Aksu Erdost, Elvan Ocmen, Seden Duru, Burc Aydin and Ali Necati Gokmen
Introduction: Sugammadex (Bridon ®) (SUG) is a recently developed neuromuscular block reversing agent. SUG can reverse also deep neuromuscular blockages in a short time period unlike other existing agents. SUG passes across blood brain barrier (BBB) in a very low ratio in normal patients. However SUG may pass the BBB in a higher ratio in patients whom BBB integrity is decreased. Since SUG passes BBB in a low ratio in normal patients there are only a small amount of studies investigating effects of this agent on central nervous system (CNS). In this study we aimed to assess the effects of SUG administered directly to intracerebroventricular space on CNS system of rats.
Materials and Method: A total of 36 Wistar-Albino rats with normal motor activity weighting between 250-280 g were included in this study. Anesthesia was achieved with intraperitoneal 50 mg/kg sodium thiopental. The rats were divided into 6 equal groups randomly as one group being the control group. The experiment groups were received 2,4,8,16 and 32 mg/kg sugammadex via intracerebroventricular cannula. Effects of the SUG on CNS were assessed based on a 5 point scale.
Results: Intracerebroventricular SUG administration did not result in any changes in behavioral status, locomotor activity or posture at any doses (2,4,8,16 and 32 mg/kg). There was no tonic clonic convulsion or seizure development following the sugammadex administration.
Discussion: SUG barely passes the BBB in normal patients. However it was stated that this drug can pass BBB in higher ratios in certain patients. Therefore investigating the effects of SUG on CNS is an emerging subject of experiments. In our study we could not find any adverse effect of SUG on CNS even at high doses administered directly to intracerebroventricular space. However presence of a study indicating an increase in apoptotic cell death in cell cultures in presence of SUG makes it difficult to make a statement that SUG does not have any adverse effect on CNS. The authors of the aforementioned study stated a connection between decrease in cholesterol levels and apoptosis. It can be speculated that some mechanisms in live animals may restore this decrease in cholesterol levels occurring in presence of SUG therefore prevents the cells from apoptosis.
Conclusion: In our study SUG did not cause any adverse effect on CNS in rats. Further studies assessing the relationship between SUG and cholesterol control mechanisms in neurons are necessary in order to make a certain statement.