PMC/PubMed Indexed Articles
Indexed In
- Academic Journals Database
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Ulrich's Periodicals Directory
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Abstract
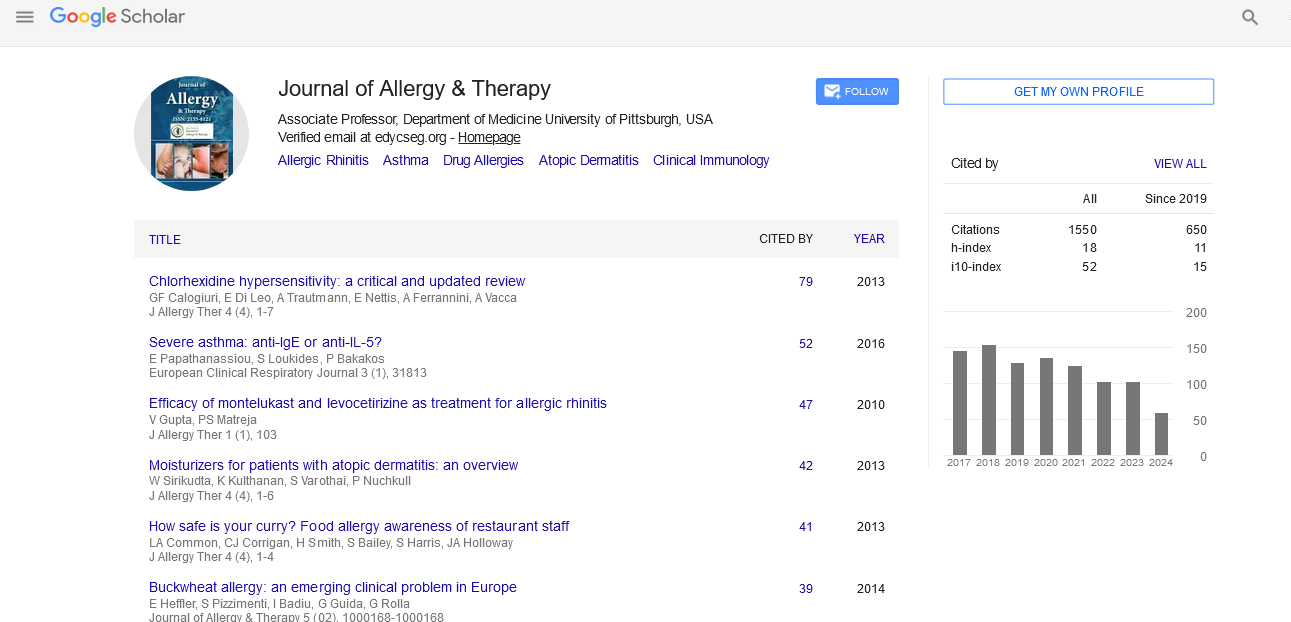
Safety of Modified Ultra-Rush Venom Immunotherapy in Children
Steiß JO, Lindemann H and Zimmer KP
Background: As many as 5% of the population in Central Europe suffer from insect venom allergy. The protective effect of conventional specific immunotherapy is quite convincing, as it is associated with a success rate up to 95%. We report our experience concerning ultra-rush dose titration in children and adolescents.
Objective: The aim of this study was to examine the safety and tolerability of a shortened insect venom immunotherapy in children.
Patients and methods: A modified version of the ultra-rush procedure was initiated for 38 bee venom and 54 wasp venom allergies in 90 patients (56 boys and 34 girls) aged 4 to 17 years. Consolidation therapy consisted of 100 μg boost injections administered after seven and 21 days while maintenance therapy was administered every four to six weeks.
Results: All patients developed local reactions under VIT, of whom 20 had extensive reddening (>5 cm-20 cm), 15 patients developed significant wheals (5 cm-15 cm), and two patients with a bee venom allergy had systemic reactions which could be controlled well. Maintenance therapy was tolerated well by all patients.
Conclusion: Ultra-rush titration is safe, tolerable and effective in children and adolescents, in addition to being associated with greater compliance and shorter hospital stays when compared to conventional therapy regimens. No serious side effects were registered. All patients could be discharged from the hospital at the latest after 48 hours.