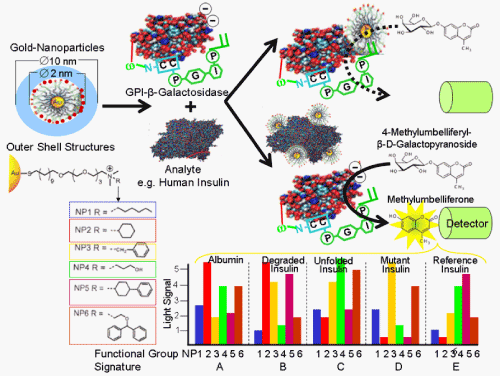
Gold-NPs exposing cationic hydrophobic structures at the outer shell compete for binding to the protein analyte, human insulin and the negatively charged GPI-modified reporter enzyme, GPI-β-galactosidase. As a consequence, GPI-ß-galactosidase may or may not be relieved from inhibition by the NPs, respectively. The light signal generated upon deinhibition of GPI-β-galactosidase and the resulting cleavage of the 4-methylumbelliferyl-ß-D-galactopyranoside dye is characteristic for each protein (surface) and each type of the NPs differing in their functional groups, NP1-6. On the basis of their differential interaction with the protein analyte surfaces, the combination of NP1-6 will result in unique signatures, A-E, that differentiate the reference insulin from the insulin variants and serum albumin.