 |
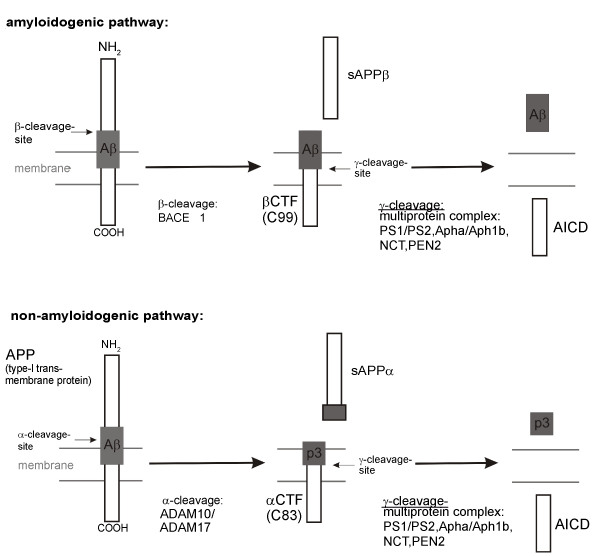
| Figure 1: Proteolytic processing of APP. The amyloid precursor protein (APP), a type-I transmembrane protein, can be cleaved in an amyloidogenic and a non-amyloidogenic pathway. The amyloidogenic pathway is initiated byβ-secretase (BACE1) cleavage of APP within its extracellular/luminal domain, generating soluble β-secreted APP (sAPPβ) and a C-terminal membrane-bound fragment of 99 amino acid residues, βCTF/C99. βCTF/C99 is further cleaved by β-secretase, a heterotetrameric protein complex consisting of at least four proteins, PS1 or PS2, Aph1a or Aph1b, nicastrin (Nct) and PEN2. The initial cut by β-secretase BACE1 generates the N-terminus of Aβ, whereas the final cut ofβCTF/C99 by γ-secretase generates the C-terminus of Aβ, thus releasing Aβ peptides. In the non-amyloidogenic pathway APP is first cleaved by α-secretases, identified as members of the ADAM family (a disintegrin and metalloproteinase) (ADAM10/ADAM17). Processing by α-secretase generates soluble α-secreted APP (sAPPα) and a C-terminal membrane-bound fragment of 83 amino acid residues, αCTF/C83, which is further cleaved by the γ-secretase complex to generate the peptide p3. The α-secretase cleaves APP within the Aβ domain, thus precluding the formation of Aβ peptides. The γ-secretase cleavage of βCTF/C99 and αCTF/C83 liberates the APP intracellular domain (AICD), which translocates to the nucleus and regulates gene expression [306-310]. |