 |
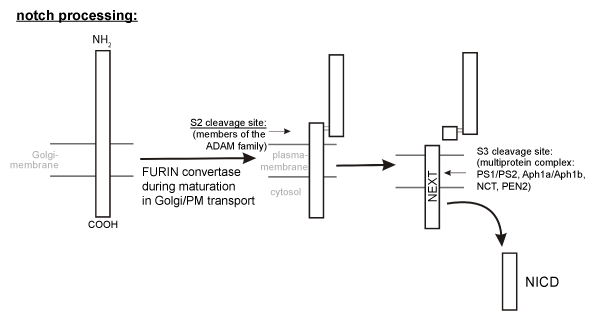
| Figure 3: Proteolytic processing of Notch. During its maturation in the secretory pathway, the Notch receptor is constitutively cleaved at the S1 site by a furinlike protease, generating a non-covalently associated heterodimeric molecule. Upon activation of the Notch signalling pathway by binding of DSL ligands to cell surface localized Notch receptors, the heterodimeric Notch molecule is cleavaed at the S2 site. In analogy to β-secretase cleavage of APP, S2 cleavage of Notch is mediated by proteases of the ADAM family. The membrane-embedded remaining Notch fragment NEXT (Notch extracellular truncation) is further processed at the S3 site by the γ-secretase complex, generating the Notch intracellular fragment (NICD), which translocates to the nucleus, where it participates directly in the transcriptional regulation of target genes involved in the generation, differentiation and survival of neuronal cells. |