PMC/PubMed Indexed Articles
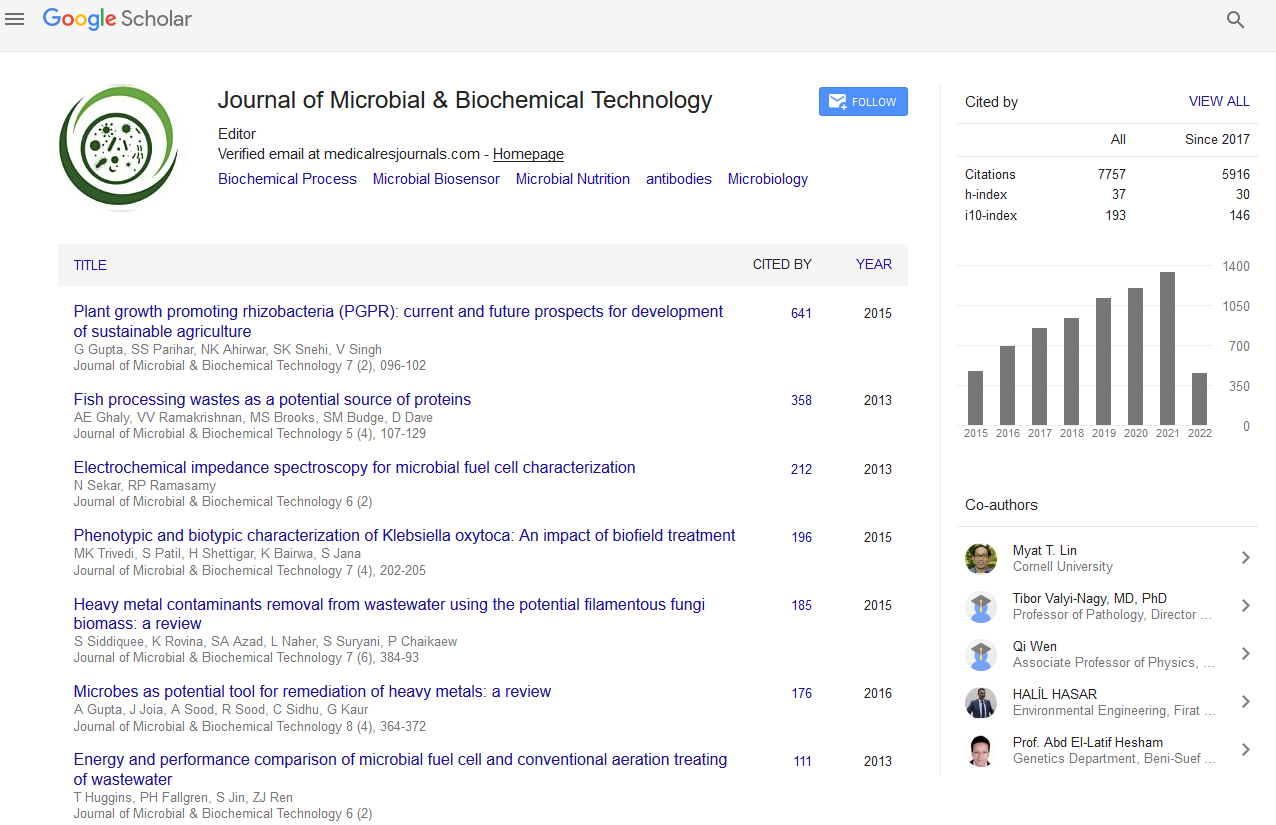
Indexed In
- Academic Journals Database
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Scimago
- Access to Global Online Research in Agriculture (AGORA)
- Electronic Journals Library
- RefSeek
- Directory of Research Journal Indexing (DRJI)
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- MIAR
- University Grants Commission
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Abstract
Expression and Purification of SAK-fused Human Interferon Alpha in Escherichia coli
Shardul Salunkhe, Bhaskarjyoti Prasad, Ketaki Sabnis-Prasad, Anjali Apte-Deshpande and Sriram Padmanabhan
A method for improved refolding and purification of E. coli derived human Interferon -? (rhIFN ?2b) from inclusion bodies as a Staphylokinase (SAK) fusion protein is described. Such a fusion protein did not require the supplementation of rare codons for expression and was found to be stable at 37?C. The optimal conditions of refolding involved the use of a mild denaturating agent without the need for any other agents to prevent aggregation. The SAKrhIFN ?2b fusion protein was successfully purified using two steps of purification and was cleaved using enterokinase into two fragments namely SAK and IFN. Both the proteins were found to be biologically active showing proper folding of both the fusion partners. The cleaved IFN showed similar retention time on RP-HPLC as the bacterial derived untagged purified IFN as well as similar molecular weight on Agilent 2100 Bioanalyzer indicating the right processing of the IFN after enterokinase cleavage. The expression levels of SAK-IFN were found to be two folds higher than that observed with untagged IFN under similar experimental conditions.