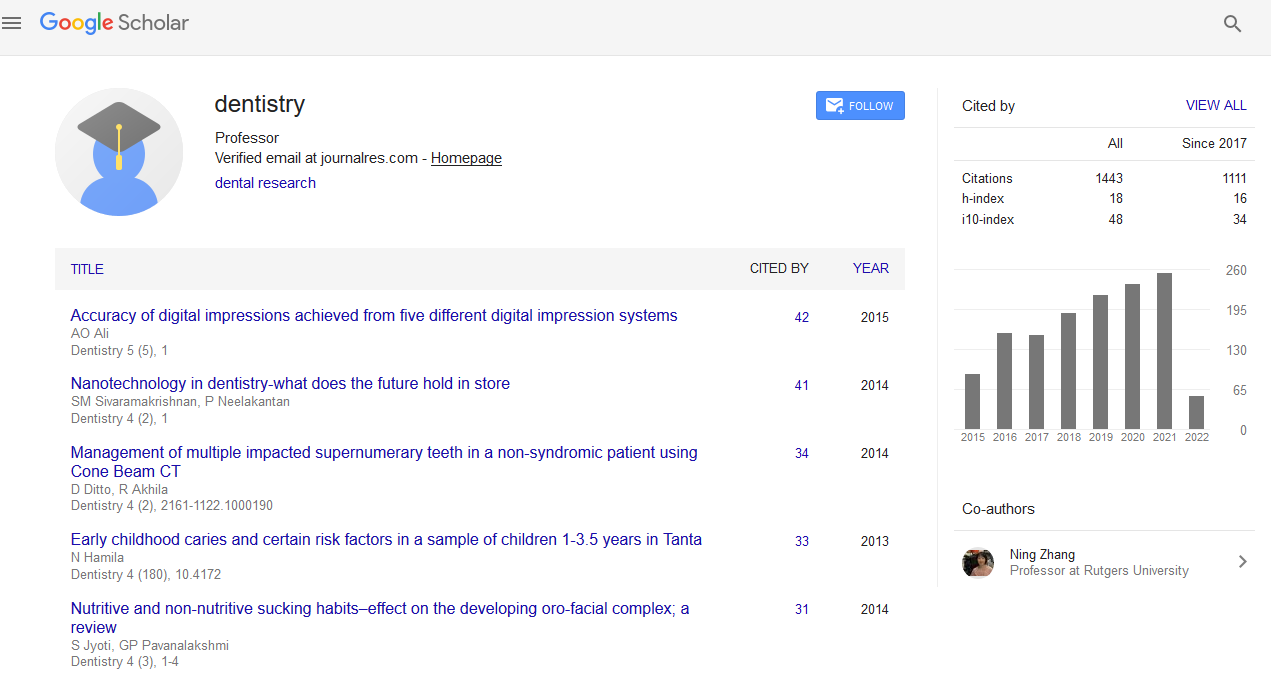
Citations : 1817
Dentistry received 1817 citations as per Google Scholar report
Indexed In
- Genamics JournalSeek
- JournalTOCs
- CiteFactor
- Ulrich's Periodicals Directory
- RefSeek
- Hamdard University
- EBSCO A-Z
- Directory of Abstract Indexing for Journals
- OCLC- WorldCat
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer
Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Abstract
The Erosivity Potential Of Common Pediatric Over-The-Counter Medications And Its Reduction By Remineralising Agents
Aim: The exposure of children’s teeth to acidic medications poses a risk for dental erosion, yet the erosivity of paediatric medications has scarcely been reported. The present study investigated the erosive potential of paediatric over-the-counter (OTC) medications, and assessed the change of the erosive potential in the presence of remineralising agents.
Methods: A range of paediatric OTC medications and commercially available drinks was examined for pH and titratable acidity (TA). Detailed testings of pH and TA were investigated on representative drinks after addition of remineralising agents: Tooth Mousse® (TM), Tooth Mousse Plus® (TMP), ClinproTM, 1.23% neutral sodium fluoride (NaF), and artificial saliva (AS).
Results: Paediatric OTC medications exhibited pH values of less than 5.5, which were comparable to commercial fruit juices and carbonated soft drinks. The TA values of OTC medications were similar to commerical fruit juices and carbonated soft drinks and ranged between 0.9 mL to 25.9 mL/ 20 mL of 0.1 M sodium hydroxide. In contrast, most dairy products, baby formulas, and bottled water had near-neutral pH (6.3-7.4) and low TA values of less than 1.3 mL/ 20 mL. The addition of TM, TMP, neutral NaF, and AS to the paediatric OTC medication increased the pH significantly compared to the control (Phosphate-buffered saline, PBS; p<0.001). The addition of TM, TMP, neutral NaF, and AS decreased the TA for paediatric OTC medication compared to the control (PBS; p<0.001-p<0.05), but not consistently.
Conclusions: Paediatric OTC medications have erosive potential comparable to commercially available drinks. The erosive parameters (pH and TA) of paediatric OTC medications can be modified by the addition of TM, TMP, neutral NaF and artificial saliva, but not ClinproTM.