PMC/PubMed Indexed Articles
Indexed In
- Academic Journals Database
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Ulrich's Periodicals Directory
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Abstract
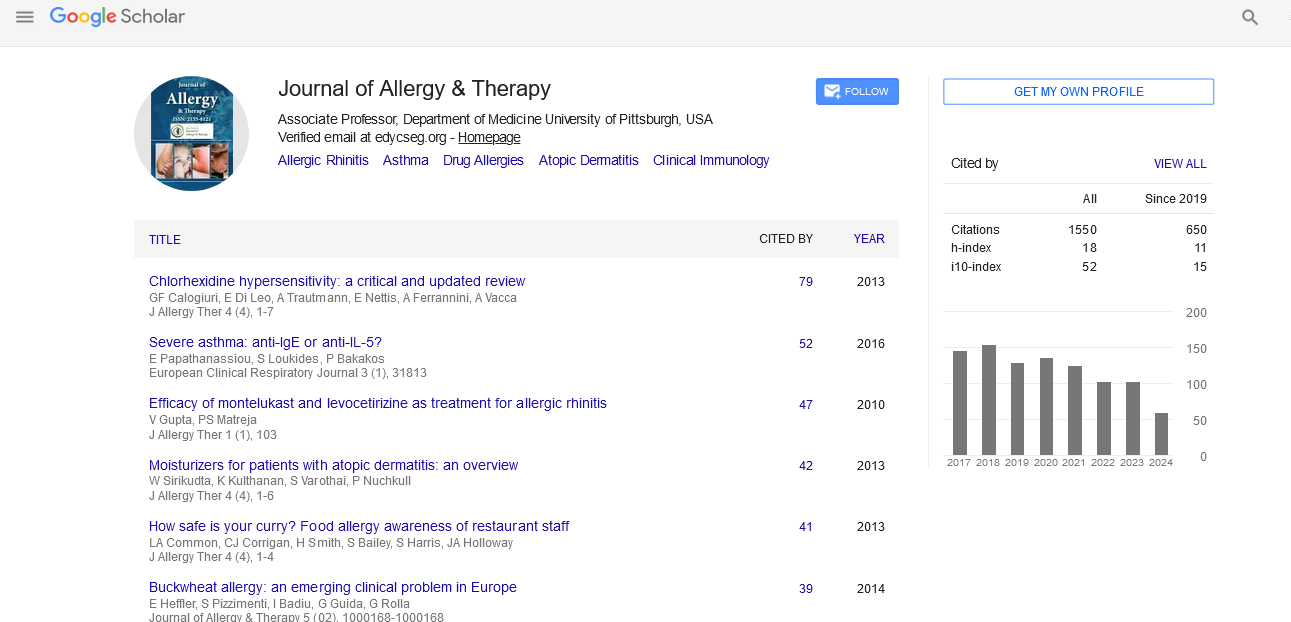
The Prevention of New Sensitizations by Specific Immunotherapy: A Long-Term Observational Case Control Study
Andrzej Bozek, Radoslaw Gawlik and Jerzy Jarzab
Some studies have indicated that allergen specific immunotherapy (SIT) may prevent new sensitizations to other inhalant allergens; however, there are only a few longitudinal observations that have explored this event. The aim of this study was to perform a 20 year post-SIT observational analysis to assess the appearance of new sensitizations in SIT patients compared with non-SIT patients.
Material and methods: In total, 1,420 atopic bronchial asthma or/and allergic rhinitis patients (701 women and 719 men) with a mean age of 21.2 ± 9.2 years (at the time when the SIT concluded) were evaluated twenty years after their immunotherapies. New sensitization cases were determined by evaluating skin prick tests, allergen specific IgE and the clinical symptoms that were evaluated prior to and 5, 10, 15 and 20 years after SIT. The SIT group was compared with a control group consisting of 1,254 allergic patients who had never received SIT and had only received symptomatic treatments.
Results: After 20 years, 301 (21.2%) patients in the 4-5 year SIT group showed a new sensitization compared with 509 (40.6%) control group patients (p=0.004). In monosensitized SIT patients (n=886), there were significantly more new sensitizations in the control group (n=624): 69 (7.8%) vs. 195 (31.3%) (p=0.001). The odds ratio of the post-SIT new sensitization incidence in the whole group was 0.76 (95% CI: 0.55-0.92), whereas it was 1.32 in the control group (95% CI: 1.22-1.45).
Conclusion: The obtained data suggests a preventive role for specific immunotherapy in new sensitizations, especially in monosensitized patients.