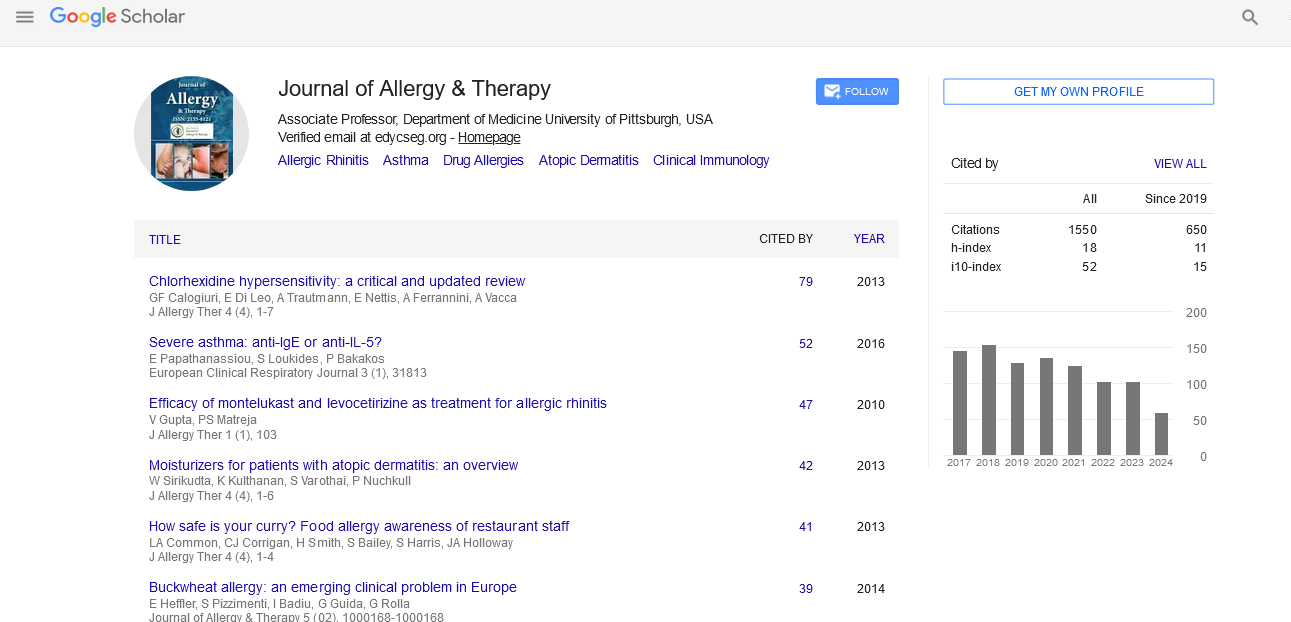
PMC/PubMed Indexed Articles
Indexed In
- Academic Journals Database
- Open J Gate
- Genamics JournalSeek
- Academic Keys
- JournalTOCs
- China National Knowledge Infrastructure (CNKI)
- Ulrich's Periodicals Directory
- Electronic Journals Library
- RefSeek
- Hamdard University
- EBSCO A-Z
- OCLC- WorldCat
- SWB online catalog
- Virtual Library of Biology (vifabio)
- Publons
- Geneva Foundation for Medical Education and Research
- Euro Pub
- Google Scholar
Useful Links
Share This Page
Journal Flyer

Open Access Journals
- Agri and Aquaculture
- Biochemistry
- Bioinformatics & Systems Biology
- Business & Management
- Chemistry
- Clinical Sciences
- Engineering
- Food & Nutrition
- General Science
- Genetics & Molecular Biology
- Immunology & Microbiology
- Medical Sciences
- Neuroscience & Psychology
- Nursing & Health Care
- Pharmaceutical Sciences
Abstract
Treatment with the SQ-Standardised Grass Allergy Immunotherapy Tablet is well Tolerated in Children, Adolescents and Adults in Real Life Application-A Non-Interventional Observational Study
Christine Gronke, Hendrik Wolf, Jörg Schnitker and Eike Wüstenberg
Background: Efficacy and safety of the SQ-standardised grass allergy immunotherapy tablet (GRAZAX®) has been reported in a large number of randomised, controlled clinical trials in children and adults performed in Europe and the US. GRAZAX® became available for routine treatment in children from 5 years of age in Germany and Austria in 2008.To examine the safety and tolerability of GRAZAX® in patients that were less highly selected as in the controlled trials we performed an open label, uncontrolled, non-interventional study in children and adults who were routinely treated in allergists´ offices.
Materials and methods: Patients with allergic rhinoconjunctivitis with or without asthma were treated with GRAZAX® and observed for 3-4 visits every 3 months with the last visit after the first grass pollen season. Adverse seasons before and during therapy.
Results: Treatment was documented in 1,761 patients (797 <18 years; 964 ≥18 years) treated by 373 allergists in Germany and Austria between November 2008 and January 2010. Adverse drug reactions were reported in 31.8% of patients (27.3% ≥18 years, 37.3% <18 years). The higher number of patients <18 years with reactions was due to a higher frequency of local oral reactions. The overall tolerability profile was similar in children and adults. Nasal symptoms improved in 82.7% of patients and no or less symptomatic medication was used by 89.7%.The compliance was rated >75% in 89.8% of patients, >95% of patients and physicians were satisfied with treatment.
Conclusion: The results of our study confirm the safety and tolerability profile observed in controlled clinical trials with GRAZAX®. Treatment satisfaction during routine application was rated high and was combined with a high compliance.