| Research Article |
Open Access |
|
| Allwyn Sundar Raj A*, Rubila S, Jayabalan R and Ranganathan T. V |
| Department of Food Processing and Engineering, School of Food Sciences and Technology, Karunya University, Coimbatore-641 114, India |
| *Corresponding author: |
Allwyn Sundar Raj A
Department of Food Processing and Engineering
School of Food Sciences and Technology
Karunya University
Coimbatore-641 114, India
E-mail: asrthegreat@gmail.com |
|
| Â |
| Received September 26, 2012; Published December 21, 2012 |
| Â |
| Citation: Sundar Raj AA, Rubila S, Jayabalan R, Ranganathan TV (2012) A Review on Pectin: Chemistry due to General Properties of Pectin and its Pharmaceutical Uses. 1:550 doi:10.4172/scientificreports.550 |
| Â |
| Copyright: © 2012 Sundar Raj AA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. |
| Â |
| Abstract |
| Â |
| Pectin, a naturally occurring polysaccharide, has in recent years gained increasingly in importance. The benefits of natural pectin are also more and more appreciated by scientists and consumer due to its biodegradability. Pectin is the methylated ester of polygalacturonic acid. It is commercially extracted from citrus peels and apple pomace under mildly acidic conditions. Pectin’s are divided into two major groups on the basis of their degree of esterification. The association of pectin chains leads to the formation of the three dimensional networks that is to gel formation. The pectin, by itself or by its gelling properties, was employed in pharmaceutical industry, health promotion and treatment. Pectin’s have been utilized for their functionality in foods for many years. Ubiquitous in the conserves and preserves industries, development of pectin’s has centred on its use to impart texture in high sugar systems. This review will discuss the importance of pectin and its chemistry, general properties of pectin, and its gel formation mechanism and properties. |
| Â |
| Keywords |
| Â |
| Pectin; Polysaccharide; Polygalacturonic Acid; Esterification; Gel formation; Pharmaceuticals |
| Â |
| Introduction |
| Â |
| Pectin is a naturally occurring biopolymer that is finding increasing applications in the pharmaceutical and biotechnology industry. It has been used successfully for many years in the food and beverage industry as a thickening agent, a gelling agent and a colloidal stabilizer. Pectin has been widely studied and published but is difficult to characterize as a model system due to the heterogeneous nature of the polymer. This review will first describe the source and production, chemical structure and general properties of pectin. The methods of gel formation and properties of the gels will then be discussed. Finally, some examples of the pharmaceutical uses of pectin will be given. |
| Â |
| Chemistry of pectin |
| Â |
| Source and production: Pectin is a complex mixture of polysaccharides that makes up about one third of the cell wall dry substance of higher plants. The highest concentrations of pectin are found in the middle lamella of cell wall, with a gradual decrease as one passes through the primary wall toward the plasma membrane (Kertesz, 1951). Because the ability of pectins to form gel depends on the molecular size and degree of esterification (DE), the pectin from different sources does not have the same gelling ability due to variations in these parameters. Therefore, detection of a large quantity of pectin in a fruit alone is not in itself enough to qualify that fruit as a source of commercial pectin [1]. At present, commercial pectins are almost exclusively derived from citrus peel or apple pomace, both by-products from juice (or cider) manufacturing. Apple pomace contains 10-15% of pectin on a dry matter basis. Citrus peel contains of 20-30% [2]. From an application point of view, citrus and apple pectins are largely equivalent. Citrus pectins are light cream or light tan in colour; apple pectins are often darker. Alternative sources include sugarbeet waste from sugar manufacturing, sunflower heads (seeds used for edible oil), and mango waste [3]. |
| Â |
| Commercially, pectin is extracted by treating the raw material with hot dilute mineral acid at pH about 2. The hot pectin extract is separated from the solid residue as efficiently as possible. This is not easy since the solids are by now soft and the liquid phase are viscous. The viscosity increases with pectin concentration and molecular weight. There is a compromise between efficient extraction and solids separation and operating cost. The pectin extract may be further clarified by filtration through a filter aid. The clarified extract is then concentrated under vacuum. Powdered pectin can be produced by mixing the concentrated liquid from either apple or citrus with an alcohol. The pectin is separated as a stringy gelatinous mass, which is pressed and washed to remove the mother liquor, dried and ground. |
| Â |
| This process yields pectin of around 70% esterification. To produce other types, some of the ester groups must be hydrolysed. This is commonly carried out by the action of acid, either before or during a prolonged extraction, in the concentrated liquid, or in alcoholic slurry before separation and drying. This process can produce a range of calcium reactive low methoxyl pectins. Hydrolysis using ammonia results in the conversion of some of the ester groups into amide groups, producing ‘amidated low methoxyl pectins’ [2]. |
| Â |
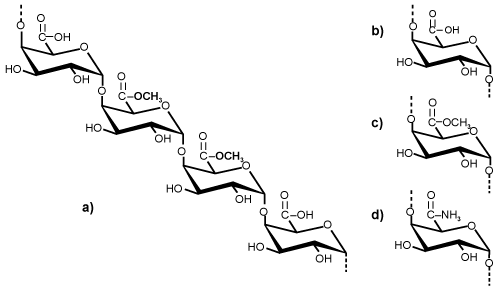
| Chemical structure: Pectin is an essentially linear polysaccharide. Like most other plant polysaccharides, it is both polydisperse and polymolecular and its composition varies with the source and the conditions applied during isolation. The composition and structure of pectin are still not completely understood although pectin was discovered over 200 years ago. The structure of pectin is very difficult to determine because pectin can change during isolation from plants, storage, and processing of plant material [4]. At present, pectin is thought to consist mainly of D-galacturonic acid (GalA) units [5], joined in chains by means of a-(1-4) glycosidic linkage. These uronic acids have carboxyl groups, some of which are naturally present as methyl esters and others which are commercially treated with ammonia to produce carboxamide groups (Figure 1). |
| Â |
|
|
Figure 1: (a) A repeating segment of pectin molecule and functional groups: (b) carboxyl; (c) ester; (d) amide in pectin chain. |
|
| Â |
| Pectin contains from a few hundred to about 1000 saccharide units in a chain-like configuration; this corresponds to average molecular weights from about 50,000 to 150,000 daltons. Large differences may exist between samples and between molecules within a sample and estimates may differ between methods of measurement. |
| Â |
| Degree of esterification: The polygalacturonic acid chain is partly esterified with methyl groups and the free acid groups may be partly or fully neutralized with sodium, potassium or ammonium ions. Pectin might be formed initially in a highly esterified form, undergoing some deesterification after they have been inserted into the cell wall or middle lamella. There can be a wide range of DEs dependent on species, tissue, and maturity. In general, tissue pectins range from 60 to 90% DE. It seems that the distribution of free carboxyl groups along the pectin chains is somewhat regular, and the free carboxyl groups are largely isolated from one another [6]. The pectin classes based on the DE are high methoxyl (HM) pectins, and the low methoxyl (LM) pectins which are either the conventionally demethylated or the amidated molecule. DEs values for commercial HM-pectins typically range from 60 to 75% and those for LM-pectins range from 20 to 40%. These two groups of pectin gel by different mechanisms. HMpectin requires a minimum amount of soluble solids and a pH within a narrow range, around 3.0, in order to form gels. HM-pectin gels are thermally reversible. In general, HM-pectins are hot water soluble and often contain a dispersion agent such as dextrose to prevent lumping. LM-pectins produce gels independent of sugar content. They also are not as sensitive to pH as the HM-pectins are. LM-pectins require the presence of a controlled amount of calcium or other divalent cations for gelation. |
| Â |
| General properties of pectin: Pectins are soluble in pure water. Monovalent cation salts of pectinic and pectic acids are usually soluble in water; di- and trivalent cations salts are weakly soluble or insoluble. Dry powdered pectin, when added to water, has a tendency to hydrate very rapidly, forming clumps. These clumps consist of semidry packets of pectin contained in an envelope of highly hydrated outer coating. Clump formation can be prevented by dry mixing pectin powder with water-soluble carrier material or by the use of pectin having improved dispersibility through special treatment during manufacturing [3,7]. Dilute pectin solutions are Newtonian but at a moderate concentration, they exhibit the non-Newtonian, pseudoplastic behaviour characteristics. Viscosity, solubility, and gelation are generally related. For example, factors that increase gel strength will increase the tendency to gel, decrease solubility, and increase viscosity, and vice versa. These properties of pectins are a function of their structure, which is that of a linear polyanion. As such, monovalent cation salts of pectins are highly ionised in solution, and the distribution of ionic charges along the molecule tends to keep it in an extended form by reason of coulombic repulsion [8]. |
| Â |
| Furthermore, this same coulombic repulsion between the carboxylate anions prevents aggregation of the polymer chains. In addition, each polysaccharide chain, and especially each carboxylate group, will be highly hydrated. Solutions of monovalent salts of pectins exhibit stable viscosity because each polymer chain is hydrated, extended, and independent. As the pH is lowered, ionisation of the carboxylate groups is suppressed, and this results in a reduction in hydration of the carboxylic acid groups. As a result of reduced ionisation, the polysaccharide molecules no longer repel each other over their entire length, and as a result, they can associate and form a gel. Apparent pK values vary with the DE of the pectin [9]; a 65% DE pectin has an apparent pK of 3.55, while a 0% DE pectic acid has an apparent pK of 4.10. However, pectins with increasingly greater degrees of methylation will gel at somewhat higher pH, because they have fewer carboxylate anions at any given pH. In general, maximum stability is found at pH 4. The presence of sugar in the pectin solution has a certain protective effect while elevated temperatures increase the rate of degradation. At low pH-values and elevated temperatures degradation due to hydrolysis of glycosidic linkages is observed. Desertification is also favoured by low pH. By deesterification a HM-pectin becomes slower setting or gradually adapts LM-pectin characteristics. At nearto- neutral pH (5-6), HM-pectin is stable at room temperature only. As the temperature (or pH) increases, a so-called elimination starts which results in chain cleavage and very rapid loss of viscosity and gelling properties. LM-pectins show a somewhat better stability at these conditions. At alkaline pH-values pectin is rapidly deesterified and degraded even at room temperature [3]. Powdered HM-pectins slowly lose their ability to form gels if stored under humid or warm conditions while LM-pectins are more stable and loss should not be significant after one year storage at room temperature [7]. |
| Â |
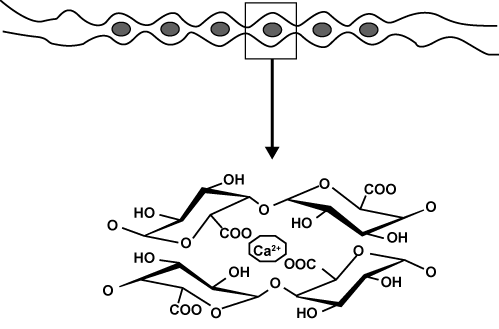
| Gel formation properties of pectin: The most important use of pectin is based on its ability to form gels. HM-pectin forms gels with sugar and acid. The particular structure of pectin imposes some specific constraints. HM-pectin, unlike LM-pectin, does not contain sufficient acid groups to gel or precipitate with calcium ions, although other ions such as aluminium or copper cause precipitation under certain conditions. It has been suggested by Oakenfull [10] that hydrogen bonding and hydrophobic interactions are important forces in the aggregation of pectin molecules. Gel formation is caused by hydrogen bonding between free carboxyl groups on the pectin molecules and also between the hydroxyl groups of neighbouring molecules. In a neutral or only slightly acid dispersion of pectin molecules, most of the unesterified carboxyl groups are present as partially ionised salts. When acid is added, the carboxyl ions are converted to mostly unionised carboxylic acid groups. This decrease in the number of negative charges not only lowers the attraction between pectin and water molecules, but also lowers the repulsive forces between pectin molecules. Sugar further decreases hydration of the pectin by competing for water. These conditions decrease the ability of pectin to stay in a dispersed state. When cooled, the unstable dispersing of less hydrated pectin forms a gel, a continuous network of pectin holding the aqueous solution. The rate at which gel formation takes place is also affected by the degree of esterification. A higher DE causes more rapid setting. Rapid-set pectins (i.e. pectin with a DE of above 72%) also gel at lower soluble solids and higher levels than slow-set pectins (i.e. pectin with a DE of 58- 65%). LM-pectins require the presence of divalent cations for proper gel formation. The mechanism of LM-pectin gelation relies mainly on the well-known ‘egg-box’ model [11]. The mechanism involves junction zones created by the ordered, side-by-side associations of galacturonans, whereby specific sequences of GalA monomer in parallel or adjacent chains are linked intermolecularly through electrostatic and ionic bonding of carboxyl groups (Figure 2). |
| Â |
|
|
Figure 2: Schematic representation of calcium binding to polygalactoronate sequences: ‘egg box’ dimer and ‘egg-box’ cavity (adapted from Axelos and Thibault, 1991). |
|
| Â |
| It is generally accepted that the junctions consist of dimers in 21 helical symmetry, similar to the 21 model proposed for alginates [12]. The oxygen atoms of the hydroxyl groups, the ring oxygen atoms, and the bridging oxygen atoms of the component sugar units participate in the bonding process through their free-electron pairs [13]. The life of the junction depends on the strength of the electrostatic bonds. The bonds are stable when there are at least seven consecutive carboxyl groups on the interior of each participating chain [14]. Nevertheless, all LM-pectin gels seem to develop similar, if not identical, junction zones [15]. Furthermore, amidation increases or improves the gelling ability of LM-pectin: amidated pectins need less calcium to gel and are less prone to precipitation at high calcium levels [2]. |
| Â |
| The amount of LM-pectin required for the formation of such gel decreases with the DE. The strengths of such ionic bonded gels are strongly dependent on the DE. Monovalent ions such as sodium, which can also react with free carboxyl groups, can affect gel formation because they decrease the cross-linking reaction of calcium and improve the solubility of LM-pectin in the presence of calcium [16]. Although sugar is not essential for gel formation with LM-pectins, the presence of small amounts (10-20%) of sugar tends to decrease syneresis and adds desirable firmness of these gels [17]. When some sugar is present, the amount of calcium required to form gel is reduced. High concentrations of sugar (60% or higher) interfere with gel formation because the dehydration of the sugar favours hydrogen bonding and decreases cross-linking by divalent ion forces. |
| Â |
| Pharmaceutical uses of pectin: Pectin has applications in the pharmaceutical industry. Pectin favaorably influences cholesterol levels in blood. It has been reported to help reduce blood cholesterol in a wide variety of subjects and experimental conditions as comprehensively reviewed [18]. Consumption of at least 6 g/day of pectin is necessary to have a significant effect in cholesterol reduction. Amounts less than 6 g/day of pectin are not effective [19]. |
| Â |
| Mietinnen and Tarplia [20] reported a 13% reduction in serum cholesterol within 2 weeks of treatment. Pectin acts as a natural prophylactic substance against poisoning with toxic cations. It has been shown to be effective in removing lead and mercury from the gastrointestinal tract and respiratory organs [21]. When injected intravenously, pectin shortens the coagulation time of drawn blood, thus being useful in controlling hemorrhage or local bleeding [22]. Pectin and combinations of pectin with other colloids have been used extensively to treat diarrheal diseases, especially in infants and children. Although a bactericidal action of pectin has been proposed to explain the effectiveness of pectin treating diarrhea, most experimental results do not support this theory. However, some evidence suggests that under certain in-vitro conditions, pectin may have a light antimicrobial action toward Echerichia coli [1]. |
| Â |
| Pectin reduces rate of digestion by immobilising food components in the intestine. This results in less absorption of food. The thickness of the pectin layer influences the absorption by prohibiting contact between the intestinal enzyme and the food, thus reducing the latter’s availability [23-25]. Due to its large waterbinding capacity, pectin gives a feeling of satiety, thus reducing food consumption. Experiments showed a prolongation of the gastric emptying half-time from 23 to 50 minutes of a meal fortified with pectin [26]. These attributes of pectin are used in the treatment of disorders related to overeating [27]. Recently, Sungthongjeen [28] have investigated HM-pectins for their potential value in controlled release matrix formulations. Pectin beads prepared by the ionotropic gelation method [29] were used as a sustained release drug delivery system. However, the use of these beads has some drawbacks due to their rapid in-vitro release. By changing the DE of LM-pectin, Sriamornsak and Nunthanid [30] modified the drug release pattern from calcium pectinate gel beads. The spherical pellets, which contain calcium acetate, were prepared using an extrusionspheronisation method and then coated in a pectin solution. An insoluble and uniform coating of calcium pectinate gel was formed around the pellets. The potential of pectin or its salt as a carrier for colonic drug delivery was first demonstrated by two studies, i.e. Ashford and Rubinstein, [31,32]. The rationale for this is that pectin and calcium pectinate will be degraded by colonic pectinolytic enzymes [33], but will retard drug release in the upper gastrointestinal tract due to its insolubility and because it is not degraded by gastric or intestinal enzymes [34]. Rubinstein, [35] demonstrated that pectindegrading bacteria, Klebsiella oxytoca, could adhere to a film casted of low methoxylated pectin. The ability of the bacteria to adhere to the films, however, was not correlated with their ability to degrade pectin. |
| Â |
| Pectin is an interesting candidate for pharmaceutical use, e.g. as a carrier of a variety of drugs for controlled release applications. Many techniques have been used to manufacture the pectin-based delivery systems, especially ionotropic gelation and gel coating. These simple techniques, together with the very safe toxicity profile, make pectin an exciting and promising excipient for the pharmaceutical industry for present and future applications. |
| Â |
| Conclusion |
| Â |
| The chemistry and gel-forming characteristics of pectin have enabled this naturally occurring biopolymer to be used in pharmaceutical industry, health promotion and treatment. It has also been used potentially in pharmaceutical preparation and drug formulation as a carrier of a wide variety of biologically active agents, not only for sustained release applications but also as a carrier for targeting drugs to the colon for either local treatment or systemic action. By selection of the appropriate type of pectin, gelation conditions, added excipients, and coating agents, the dosage forms of various morphology and characteristics can be fabricated. As research and development continues with delivery system using pectin, we expect to see many innovative and exciting applications in the future. |
| Â |
| References |
| Â |
- Thakur BR, Singh RK, Handa AK (1997) Chemistry and uses of pectin--a review. Crit Rev Food Sci Nutr 37: 47-73.
- May CD (1990) Industrial pectins: Sources, production and applications. Carbohyd Polym 12: 79-99.
- Rolin C (1993) Pectin. In Industrial gums. Whistler RL, BeMiller JN (eds), (3rdedn), New York: Academic Press.
- NovoselÂ’skaya IL, Voropaeva NL, Semenova S, Rashidova SSh (2000) Trends in the science and applications of pectins. Chem Nat Compd36: 1-10.
- Mukhiddinov ZK, Khalikov DK, Abdusamiev FT, Avloev CC (2000) Isolation and structural characterization of a pectin homo and ramnogalacturonan. Talanta53: 171-176.
- DeVries JA, Hansen M, Søderberg J, Glahn PE, Pedersen JK (1986) Distribution of methoxyl groups in pectins. Carbohyd Polym 6: 165-176.
- Hercules Incorporated (1999) Food gum products description: General description of pectin.
- Paoletti S (1986) In Chemistry and function of pectins, eds. M.L. Fishman and J.J. Jen. (Washigton DC: American Chemical Society)
- Plashchina IG, Braudo EE, Tolstoguzov VB (1978) Circular-dichroism studies of pectin solutions. Carbohyd Res60: 1-8.
- Oakenfull DG (1991) The chemistry of high-methoxyl pectins. The chemistry and technology of pectin 87-108.
- Grant GT, Morris ER, Rees DA, Smith PJC, Thom D (1973) Biological interactions between polysaccharides and divalent cations: The egg-box model. FEBS Lett32: 195-198.
- Axelos MAV, Thibault JF (1991) The Chemistry of Low-Methoxyl Pectin Gelation. The chemistry and technology of pectin 109-118.
- Kohn R (1987) Binding of divalent cations to oligomeric fragments of pectin. Carbohyd Res160: 343-353.
- Powell DA, Morris ER, Gidley MJ, Rees DA (1982) Conformations and interactions of pectins: II. Influence of residue sequence on chain association in calcium pectate gels. J Mol Biol155: 517-531.
- Filippov MP, Komissarenko MS (1988) Investigation of ion exchange on films of pectic substances by infrared spectroscopy. Carbohyd Polym8: 131-135.
- Axelos MAV (1990) Ion complexation of biopolymers: Macromolecular structure and viscoelastic properties of gels. Macromol Symp39: 323-328.
- Christensen SH (1986) Pectins. In Food hydrocolloids III. Glicksman M (ed), CRC Press, Florida.
- Sriamornsak P (2001) Pectin: The role in health. Journal of Silpakorn University21: 60-77.
- Ginter E, Kubec FJ, Vozár J, Bobek P (1979) Natural hypocholesterolemic agent: pectin plus ascorbic acid. Int JVitam Nutr Res49: 406-412.
- Miettinen TA, Tarpila S (1977) Effect of pectin on serum cholesterol, fecal bile acids and biliary lipids in normolipidemic and hyperlipidemic individuals. Clin Chim Acta 79: 471-477.
- Kohn R (1982) Binding of toxic cations to pectin, its oligomeric fragments and plant tissues. Carbohyd Polym2: 273-275.
- Joseph GH (1956) Pectin: Bibliography of pharmaceutical literature. (Ontario: Sunkist Growers)
- Wilson FA, Dietschy JM (1974) The intestinal unstirred layer: Its surface area and effect on active transport kinetics. Biochimica et Biophysica Acta 363: 112-126.
- Dunaif G, Schneeman BO (1981) The effect of dietary fiber on human pancreatic enzyme activity in vitro. Am J Clin Nutr 34: 1034-1035.
- Flourie B, Vidon N, Florent CH, Bernier JJ (1984) Effect of pectin on jejunal glucose absorption and unstirred layer thickness in normal man. Gut 25: 936-941.
- Holt S, Heading RC, Carter DC, Prescott LF, Tothill P (1979) Effect of gel fibre on gastric emptying and absorption of glucose and paracetamol. Lancet24: 636-639.
- Di Lorenzo C, Williams CM, Hajnal F, Valenzuela JE (1988) Pectin delays gastric emptying and increases satiety in obese subjects. Gastroenterology 95: 1211-1215.
- Sungthongjeen S, Pitaksuteepong T, Somsiri A, Sriamornsak P (1999). Studies on Pectins as Potential Hydrogel Matrices for Controlled-Release Drug Delivery. Drug Dev Ind Pharm 25: 1271-1276.
- Aydin Z, Akbuga J (1996) Preparation and evaluation of pectin beads. Int J Pharm137: 133-136.
- Sriamornsak P, Nunthanid J (1998) Calcium pectinate gel beads for controlled release drug delivery: I. Preparation and in vitro release studies. Int J Pharm160: 207-212.
- Ashford M, Fell J, Attwood D, Sharma H, Woodhead P (1993) An evaluation of pectin as a carrier for drug targeting to the colon. J Control Release 26: 213-220.
- Rubinstein A, Radai R, Ezra M, Pathak S, Rokem JS (1993) In vitro evaluation of calcium pectinate: a potential colon-specific drug delivery carrier. Pharm Res 10: 258-263.
- Englyst HN, Hay S, Macfarlane GT (1987) Polysaccharide breakdown by mixed populations of human faecal bacteria. Fems Microbiol Lett45: 163-171.
- Sandberg AS, Ahderinne R, Andersson H, Hallgren B, Hultén L (1983) The effect of citrus pectin on the absorption of nutrients in the small intestine. Hum Nutr Clin Nutr 37: 171-183.
- Rubinstein A, Ezra M, Rokem JS (1992) Adhesion of bacteria on pectin casted films. Microbios 70: 163-170.
|
| Â |
| Â |