Review Article Open Access
Analytical Method Development and Validation: A Concise Review
Ashish Chauhan1*, Bharti Mittu1 and Priyanka Chauhan2
1SMPIC, NIPER, Mohali, Punjab, India
2Shikhar S. Sadan Sr. Sec. School, Dhampur, Bijnor, India
- *Corresponding Author:
- Ashish Chauhan
SMPIC, NIPER, Mohali
Punjab, India
E-mail: aashishchauhan26@gmail.com
Received Date: January 28, 2015; Accepted Date: February 14, 2015; Published Date: February 23, 2015
Citation: Chauhan A, harti Mittu B, Chauhan P (2015) Analytical Method Development and Validation: A Concise Review. J Anal Bioanal Tech 6: 233. doi: 10.4172/2155-9872.1000233
Copyright: ©2015 Chauhan A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
Analytical method development and validation are the continuous and inter-dependent task associated with the research and development, quality control and quality assurance departments. Analytical procedures play a critical role in equivalence and risk assessment, management. It helps in establishment of product-specific acceptance criteria and stability of results.Validation should demonstrate that the analytical procedure is suitable for its intented purpose. Design of experiment is a powerful tool for the method characterization and validation. Analytical professionals should be comfortable to use it to characterize and optimize the analytical method. An effective analytical method development and its validation can provide significant improvements in precision and a reduction in bias errors. It can further help to avoid costly and time consuming exercises.
Keywords
Validation; Stability; Precision; Accuracy; SOP
Introduction
Analytical method development
Analytical Chemistry is the branch of Science that uses advance technologies in determining the composition by analytical technique. We can achieve both qualitative as well as quantitative results. Analytical instruments play a major role in the process to achieve high quality and reliable analytical data. Thus everyone in the analytical laboratory should be concerned about the quality assurance of equipment.
Analytical method could be spectral, chromatographic, electrochemical, hyphenated or miscellaneous. Analytical method development is the process of selecting an accurate assay procedure to determine the composition of a formulation. It is the process of proving that an analytical method is acceptable for use in laboratory to measure the concentration of subsequent samples Analytical methods should be used within GMP and GLP environments and must be developed using the protocols and acceptance criteria set out in the ICH guidelines Q2(R1). The prerequisite for method development are as follows [1-4]:
1. Qualified and calibrated instruments
2. Documented methods
3. Reliable reference standards
4. Qualified analysts
5. Sample selection and integrity
6. Change control
An analytical procedure is developed to test a defined characteristic of the substance against established acceptance criteria for that characteristic. In the development of a new analytical procedure, the choice of analytical instrumentation and methodology should be based on the intended purpose and scope of the analytical method. The important parameters that may be evaluated during method development are specificity, linearity, limits of detection (LOD) and quantitation limits (LOQ), range, accuracy and precision (Table 1). During early stages of method development, the robustness of methods should be evaluated because this characteristic ultimately helps to decide which method will be approved. Analytical procedures development are primarily based on a combination of mechanistic understanding of the basic methodology and prior experiences. Experimental data from early procedures can be used to guide further development.
| Parameters | Identification | Impurities QuantitativeLimit | Assay | |
|---|---|---|---|---|
| Accuracy | - | + | - | + |
| Precision | - | + | - | + |
| Specificity | + | + | + | + |
| Detection limit | - | - | + | - |
| Quantitation limit | - | + | - | - |
| Linearity | - | + | - | + |
| Range | - | + | - | + |
| Robustness | + | + | + | + |
Table 1: The parameters of an analytical procedure.
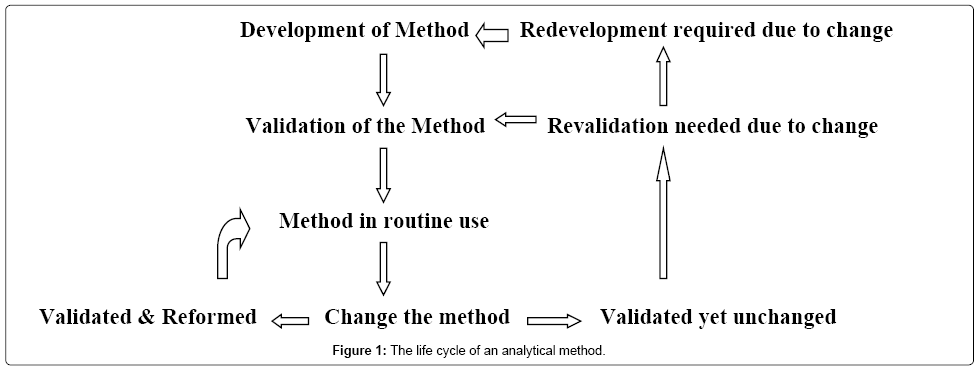
The life cycle of an analytical method is brief as shown in Figure 1. The common steps followed in the method development are as follows:
1. Standard analyte characterization
2. Method requirements
3. Literature search
4. Selecting the method
5. Instrumental setup and preliminary studies
6. Optimization of parameters
7. Documentation of analytical figure
8. Evaluation of the method development with the sample
9. Determination of percent recovery of the sample
10. Demonstration of quantitative sample analysis
The ability to provide accurate, reliable and consistent data is the motive of the analytical chemist. Method development procedures are complex, extended and expensive endeavors. An analytical method details the steps and techniques necessary to perform an analysis. This may include: preparation of samples, standards and reagents; use of apparatus; generation of the calibration curve, use of the formulae for the calculation etc. Analytical Method Development is required for [1-4]:
1. Herbal products and their potency
2. New process and reactions
3. New molecules development
4. Active ingredients (Macro analysis)
5. Residues (Micro analysis)
6. Impurity profiling
7. Component of interest in different proportion
8. Degradation studies
Need of analytical method development and validation
The need of validation of the analytical method development and validation emerged due to international competition, maintaining the standard of products in high commercial & market value and ethical reasons. Various International Regulatory Agencies have set the standard and fixed the protocol to match the reference for granting approval, authentication and registration. Some of the famous organizations governing the quality standards are:
1. United States Food and Drug Administration (US FDA)
2. Current Good Manufacturing Practice (cGMP) regulations
3. Good Laboratory Practice (GLP) regulations.
4. The Pharmaceutical Inspection Cooperation Scheme’s (PIC/S)
5. Pharmaceutical Inspection Cooperation Scheme (PIC/S)
6. The International Conference for Harmonization (ICH)
7. ISO/IEC 17025
8. World Health Organization (WHO)
When some changes are made in the validated nonstandard methods, the influence of such changes should be documented and a new validation should be carried out. If standard methods are available for a specific sample test, the most recent edition should be used. Validation includes specification of requirements, determination of method characteristics, a check that the requirements can be fulfilled by using the method, a statement on validity [5-8].
To fully understand the effect of changes in method parameters on an analytical procedure, adopt a systematic approach for method robustness study (design of experiments with method parameters) followed by an initial risk assessment and multivariate experiments. Such approaches allow us to understand parameter effects on method performance. Evaluation of a method’s performance may include analyses of samples obtained from in-process manufacturing stages to the finished product. The information obtained during these studies on the sources of method variation can help to assess the method’s performance.
Validation of the method
Data quality is assured by the combination of four components: analytical instrument qualification (AIQ); analytical method validation; system suitability tests and quality control checks. Validationof an analytical method is intended to demonstrate that it is suitable for its intended use. We generally validate the method under following conditions:
1. During method development
2. Checking the system suitability
3. Change of application, environment, analyst
4. While using after a prolonged period of time
5. Checking reliability and consistency
The type of method and analytical technique used will determine the nature and extent of the validation studies required. The most common methods for validation are identification, assay and impurities determination [5-8].
The validation report details the results of the validation study. Its purpose is to provide the information on the characteristics on the basis of which they were tested during the study, the results obtained and the interpretation of those results. Typical information in a validation report includes:
1. Validation protocol.
2. Analytical method
3. The validation parameters
4. The results
5. Interpretation of the results
6. Relevant validation information
7. Details of the reference materials
8. Details of batch number
9. Details of the equipment used for the study
10. References to the laboratory details
Typical validation parameters recommended by FDA, USP, and ICH are as follows:
1. Specificity
2. Linearity and Range
3. Precision
(A) Method precision (Repeatability)
(B) Intermediate precision (Ruggedness)
4. Accuracy
5. Solution stability
6. Limit of Detection (LOD)
7. Limit of Quantification (LOQ)
8. Robustness
Method validation is a vast area which includes many validation parameters with different approaches for different level of requirements based on intended use of analytical method. Validated method elucidates the unpredicted or unknown problem during the course of routine usage. Validated method has limited level of confidence. After method development it needs to be validated as per requirement that gives certain level of confidence for its intended use.
Criteria of Validation
The validation of an analytic method demonstrates the scientific soundness of the measurement or characterization. It is required to varying extents throughout the regulatory submission process. The validation practice demonstrates that an analytic method measures the correct substance, in the correct amount and in the appropriate range for the samples. It allows the analyst to understand the behavior of the method and to establish the performance limits of the method [9-11].
In order to perform method validation, the laboratory should follow a written standard operating procedure (SOP) that describes the process of conducting method validation. The laboratory should use qualified and calibrated instrumentation. There should be a welldeveloped and documented test method and an approved protocol prior to validation. The protocol is a systematic plan that describes which method performance parameters should be tested, how the parameters will be assessed with its acceptance criteria. Like in case of Pharmaceuticals, an API or drug product, placebos and reference standards are needed to perform the validation experiments.
Accuracy is the closeness of agreement between the values found. The value accepted as a conventional true value or the accepted reference value. Several methods of determining accuracy are available. It can be screened by the use of an analytical procedure to an analyte of known purity, by comparison of the results of the proposed analytical procedure with those of a second accepted procedure, the accuracy of which is stated and defined. It can also be inferred once precision, linearity and specificity have been established [3-9].
Precision of an analytical procedure expresses the closeness of agreement between a series of measurements obtained from multiple sampling of the same homogeneous sample under the prescribed conditions. It can be sub divided into repeatability, intermediate precision and reproducibility. The standard deviation, relative standard deviation like coefficient of variation and confidence interval should be reported for each type of precision investigated.
1. Repeatability should be assessed using a minimum of 9 determinations covering the specified range for the procedure by 3 replicates or 6 determinations at 100% of the test concentration.
2. Immediate precision depends upon the circumstances under which the procedure is intended to be used. The specific day, analyst performing, equipment are the random events that cast effect on the precision of the analytical procedure. It is not considered necessary to study these effects individually. The use of an experimental design should be encouraged.
3. Reproducibility is assessed by means of an inter-laboratory trial. Reproducibility should be considered in case of the standardization of an analytical procedure.
4. Specificity is the ability to assess the analyte for the presence of various components that may be present. It can be established by a number of approaches, depending on the intended purpose of the method. The ability of the method to assess the analyte of interest in a drug product is determined by a check for interference by placebo. Specificity can be assessed by measurement of the API in samples that are spiked with impurities or degradants. If API-related compounds are not available, drug can be stressed or force-degraded in order to produce degradation products. In chromatographic separations, apparent separation of degradants may be confirmed by peak purity determinations by photodiode array, mass purity determinations by mass spectroscopy (MS) or by confirming separation efficiency using alternate column chemistry. During forced degradation experiments, degradation is targeted at 5 to 20% degradation of the API, in order to avoid concerns about secondary degradation. Lack of specificity of an individual analytical procedure may be compensated by other supporting analytical procedures.
5. The detection limit of an individual analytical procedure is the lowest amount of analyte in a sample that can be detected. It can be determined visually, by signal to noise ratio, standard deviation of the response and the slope. Detection limit signal to noise approach can only be applied to analytical procedures which exhibit baseline noise. Comparing measured signals from samples with known concentrations of analyte with those of blank samples and establishing the minimum concentration at which the analyte can be reliably detected. A signalto- noise ratio between 3 or 2:1 is generally considered acceptable for estimating the detection limit. The detection limit (DL) may be expressed as: DL=3.3 σ/ S where, σ is the standard deviation of the response, S is the slope of the calibration curve. The slope S may be estimated from the calibration curve of the analyte. The estimate of σ may be carried out in a variety of ways, based on the standard deviation of the blank and the calibration curve [2-9]
6. The linearity of an analytical procedure is its ability to obtain test results that are directly proportional to the concentration of analyte in the sample. Test results should be evaluated by appropriate statistical methods, by calculation of a regression line like by the method of least squares. correlation coefficient, y-intercept, slope of the regression line and residual sum of squares for which a minimum of five concentrations are recommended.
7. The range of an analytical procedure is the interval between the upper and lower concentration of analyte in the sample for which it has been demonstrated that the analytical procedure has a suitable level of precision, accuracy and linearity.
8. Robustness is typically assessed by the effect of small changes in chromatographic methods on system suitability parameters such as peak retention, resolution and efficiency. Experimental factors that are typically varied during method robustness evaluations include: (i) age of standards and sample preparations (ii) sample analysis time (iii) variations to pH of mobile phase (iv) variation in mobile phase composition (v) analysis temperature (vi) flow rate (vii) column manufacturer (viii) type and use of filter against centrifugation. Robustness experiments are an ideal opportunity to utilize statistical design of experiments, providing data-driven method control.
The ICH guidance on validation distinguishes the types of methods according to the purpose of the method and lists suitable evaluation type. The ICH guidlines suggests detailed validation schemes relative to the purpose of the methods. It lists recommended data to report for each validation parameter. Acceptance criteria for validation must be based on the previous performances of the method, the product specifications and the phase of development.
As previously mentioned, the path to validation forms a continuum. It begins in the early phases of development as a set of informal experiments that establishes the soundness of the method for its intended purpose. It is expanded throughout the regulatory submission process into a fully-documented report that is required for commercial production. It is repeated whenever there is a significant change in instrumentation, method, specifications and process [1-11]
Issues and Challenges
For a method development and validation program to be successful, a holistic approach is recommended. A common challenge encountered during methods development and validation is that methods are typically developed by the research and development department whereas validation is typically the responsibility of a quality assurance and quality control. It’s important that all the groups work as one team. Various groups may be responsible for ensuring the suitability of the methods to support various phases and commercial manufacturing. The transfer of analytical methods from one group to another then becomes an important step for ensuring that the proper validation is in place to justify its intended use. Because the method will be run by several groups during its progress from development to validation but the method must be robust. A common weakness in development and validation of methods is that the methods are not robust enough. If robustness is not built into methods early in development then the results are likely to lack efficiency in quality testing and encounter lengthy and complicated validation process. It is achieved by conducting forced-degradation studies. The design and execution of these studies requires thorough knowledge of the product being tested as well as a good understanding of the analysis technique. New regulatory guidelines are being published that govern the expectations of regulatory agencies throughout the world for methods development and validation. There is need to meet current regulatory standards. From a simple method improvement to a complete redevelopment and subsequent implementation is tedious task. For this reason, one must be alert to current trends in regulatory guidelines and to adopt a proactive approach to changes that may affect development and validation programs. Finally, one of the key requirements for methods validation is that only well-characterized reference materials with proper documented purities should be used during method validation [1-15].
Conclusion
Analytical method development helps to understand the critical process parameters and to minimize their influence on accuracy and precision. Analytical methods should be used by following GMP and GLP guidelines and must be developed using the protocols and acceptance criteria set out in the ICH guidelines Q2(R1). Method validation helps to validate the analytical method for a range of concentrations so that the change in formulation or concentration do not require additional validation. Once the methods have been developed, qualified and validated the impact they have on out-ofspecification rates and process capability needs to be quantified and evaluated to determine its effectiveness for future use.
References
- (2000) International Conference on Harmonization (ICH) of Technical Requirements for Registration of Pharmaceuticals for Human Use, Topic Q7: Good Manufacturing Practices for Pharmaceutical Ingredients.
- Current Good Manufacturing Practices for finished Pharmaceuticals, 21 CFR, Parts 210 and 211, US Food and Drug Administration.
- European Commission (2001) Final Version of Annex 15 to the EU Guide toGood Manufacturing Practice: Qualification and validation: 4 1-10.
- McDowall RD (2005) Effective and Practical risk management options for computerized system validation, Quality Assurance Journal 9 (3): 196-227
- Bansal KS, Layloff T, Bush ED, Hamilton M, Hankinson EA, et al. (2004) Qualification of Analytical Instruments for Use in the Pharmaceutical Industry: a Scientific Approach. AAPS Pharm Sci Tech 5: 1-8.
- Bedson P, Sargent M (1996)The development and application of guidance on equipment qualification of analytical instruments.AccredQual. Assurance 1: 265-274.
- OonaMcPolin (2009) Validation of Analytical Methods for Pharmaceutical Analysis, Mourne Training Services, 14Burren Road, Warren point Co. Down BT34 3SA.
- FDA (2000) Guidance for Industry: Analytical Procedures and Method Validation, Chemistry, Manufacturing, and Controls Documentation, U.S. Department of Health and Human Services.
- International Conference on Harmonization (1994) Validation of analytical procedures: text and methodology Q2(R1).
- USP (2009) Chromatography System Suitability. United States Pharmacopeial Convention.
- Chan CC,Lam H, Lee YC, Zhang XM (2004) Analytical Method Validation and Instrument Performance Verification, Hoboken, John Wiley & Sons (Wiley Inter science),New Jersey.
- Robert W. Lee,Laurie Goldman The Central Role Of Analytic Method Development And Validation In Pharmaceutical Development. Life Science Connect 1-3
- Burdick RK, LeBlond D, Sandell D, Yang H. (2013) Statistical methods for validation of procedure accuracy and precision. PharmacopeialForum 39:(3)
- NethercoteP, Ermer J. (2013) Quality by design for analytical methods: implications for method validation and transfer. Pharm Technol 36(10): 74-79.
- Weitzel MLJ (2012)The estimation and use of measurement uncertainty for a drug substance test procedure validated according to USP 1225 Accreditation Quality Assurance 17(2): 139-146.
--
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 81374
- [From(publication date):
February-2015 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 71075
- PDF downloads : 10299