Research Article Open Access
Assessment of the Importance of Non-Stage Related Factors in the Prognosis of Resected Colorectal Cancer
Jamish Gandhi1*, Fraser Welsh1 and Keating JP2
1Surgical Registrar, Wellington Hospital, Wellington Hospital, New Zealand
2Colon and Rectal Surgeon, Wellington Hospital, New Zealand
- *Corresponding Author:
- Jamish Gandhi
Department of General Surgery, Wellington Hospital
PO Box 7343, Wellington Southm, New Zealand
Tel: 0064 212667892
E-mail: j.gandhi@xtra.co.nz
Received date: November 08, 2012; Accepted date: December 12, 2014; Published date: December 15, 2014
Citation: Gandhi J, Welsh F, Keating JP (2014) Assessment of the Importance of Non-Stage Related Factors in the Prognosis of Resected Colorectal Cancer. J Gastrointest Dig Syst 4:244. doi:10.4172/2161-069X.1000244
Copyright: © 2014 Gandhi J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Objective: To assess the importance of clinical and histopathological features in the risk stratification of resected colorectal cancer and their utility as indicators of the need for adjuvant therapy.
Methods: Prospectively collected data was analysed from 524 consecutive cases of colon and rectal cancer resected by a single surgical team with a median follow up of 49 months. Multivariate analysis was used to determine clinical and pathological features that independently predicted cancer related death and their relative importance.
Results: Pathological features associated with an increased risk of cancer related death were the presence of metastatic disease, penetration of the bowel wall, lymph node status, perineural invasion and vascular invasion. Emergent surgery was the only clinical feature to predict cancer related death. Adjuvant chemotherapy significantly improved the survival of colorectal cancer patients whose tumours demonstrated either vascular or perineural invasion.
Conclusions: Vascular and perineural invasion and emergent surgery are independent and significant predictors of death from colorectal cancer. Adjuvant chemotherapy improved the survival of patients whose tumours demonstrated vascular or perineural invasion. The components of TNM stage alone do not provide enough information on which to make decisions on the utility of adjuvant chemotherapy.
Keywords
Colorectal cancer; Colon cancer; Rectal cancer; Prognostic factors
Introduction
Histopathological assessment of the surgical specimen in colorectal cancer (CRC) remains fundamental to risk stratification and assessment of the relative merits of the use of adjuvant chemotherapy. Tumour stage, as assessed by depth of penetration of the bowel wall, and the status of the draining lymph nodes form the basis of stage definition in all bowel cancer staging systems with the exception of the Jass system [1-4].
TNM stage alone however provides at best an inexact guide to prognosis [5,6]. Fifty percent of Stage III cases are cured by surgery alone while up to twenty percent of stage II cases will die of colorectal cancer [7,8]. TNM Stage by itself is clearly an imprecise prognostic tool and decision making with regard to the use of adjuvant therapy may be improved by incorporation of other clinical and pathological factors of quantified importance.
In addition to tumour stage a number of other clinical and pathological variables have been shown to have prognostic significance. Emergent surgery, perioperative blood transfusion and male gender are clinical factors that have been variably reported to adversely affect prognosis [9-12] whereas, number of nodes examined, vascular invasion (VI), poor differentiation, perineural invasion (PNI), mucinous tumour type and tumour extension outside involved lymph nodes are pathological features reported by some authors to worsen prognosis in any given stage [13-18]. The reported incidence of these variables and their relative importance varies widely in the surgical literature.
In order to test the contribution of the clinical and pathological variables in resected colon and rectal cancer a comprehensive range of variables were prospectively recorded for bowel cancer resections in a colorectal unit over a thirteen year period. Their influence on cancer specific survival and the utility of adjuvant chemotherapy was assessed.
Methods
A prospective electronic database (EPI INFOTM, CDC Atlanta, Georgia, USA) was used to record the details of all patients treated with colorectal cancer. The clinical and pathological variables, treatment received and the short and long term outcome of patients treated for colorectal cancer in a tertiary unit from January 1997 to June 2011 were prospectively collected and analysed.
Pathology reporting of resected colorectal cancers incorporated a synoptic reporting system to reliably capture pathological variables. In a small number of cases in which the original pathology report omitted PNI and VI status the pathologist was requested to submit a supplementary report incorporating these features.
Patients were operated on by, or under the supervision of, the senior author. The clinical outcome of all patients was determined from ongoing clinical review, by telephone interview with the patient or their general practitioner, from the death certificate or national patient database after discharge from surgical follow up.
Cancers were recorded as being colon or rectal, with tumours at the rectosigmoid junction being grouped with colon cancers. Operations performed outside an elective list were classified as emergent. Total mesorectal excision (TME) was performed for all mid and lower third rectal cancers. Latterly all treatment decisions have taken place in a multidisciplinary team (MDT) setting. Perioperative blood transfusion was defined as any transfusion within two weeks of the operation date.
Patients referred with recurrent tumours, or those who did not undergo a resection of the primary tumour were excluded from analysis. Patients were staged according to the TNM and Australian Clinicopathological Staging Systems (ACPS). In patients with synchronous tumours the more pathologically advanced lesion was used to classify the tumour. Patients subsequently treated for metachronous colorectal cancers were analysed on the clinical and pathological criteria of the index cancer.
Postoperative surveillance consisted of three to six monthly clinical review, three monthly CEA estimation, colonoscopy at one and subsequently three year intervals. Regular imaging was reserved earlier in the study for high risk cases, or to investigate a rising CEA and more recently as a routine at one year and two and a half years following resection.
In order to determine the relative importance of each clinical and pathological variable the individual components of TNM stage; penetration of the bowel wall, lymph node involvement and the presence or absence of metastatic disease were assessed alongside the non-stage related pathological factors and clinical factors.
A Cox proportional hazard model (SAS, SAS Institute Inc, Cary, North Carolina, USA) was used to identify independent risk factors and the strength of their association (Hazard ratio with 95% confidence intervals) with cancer related death. The log rank test was used to compare survival probabilities.
Results
Over a fourteen year period, from January 1997 to June 201, 554 patients had surgery for a new primary colorectal malignancy and of these 524 patients, 95%, had the primary tumour resected. Six patients died within 30 days of operation and were excluded from further analysis. Thus 288 patients who underwent resection of a colon cancer and 230 of a rectal cancer form the basis for this study. The median length of follow up from time of operation to time of death or last contact was 49 months with a mean of 62 months.
The stage distribution of resected tumours reflects a predominantly unscreened population (TNM stage I 21%, II 33%, III 33%, IV 13%). There was no difference in either overall (p=0.10) or the disease free (p=0.31) survival of patients with colon cancer compared to patients with rectal cancer.
Perineural invasion (PNI) was reported in 12.4% of the resected specimens, and was more commonly reported in rectal cancer (16.3%) than in colon cancer (9.3%). The incidence of PNI steadily increased with TNM stage (Table 1).
| TNM Stage | PNI | VI |
|---|---|---|
| I | 2.0% | 4.9% |
| II | 7.3% | 14.4% |
| III | 19.8% | 36.7% |
| IV | 23.3% | 54.7% |
Table 1: Frequency of perineural invasion and vascular invasion to TNM stage (N= 518).
Vascular invasion (VI) was in 24.6% and in contradistinction to PNI was recognised more commonly in colon cancer (28.5%) than in rectal cancer (19.8%) but showed a similar association to TNM stage as did PNI (Table 1).
The presence of metastatic disease when analysed in the proportional hazards model was associated with an over six fold risk of cancer related death (Table 2). Penetration of the bowel wall, that is tumour that had breached the full thickness of the muscularis propria, and the presence of lymph node involvement by tumour were associated with hazard rates (HR) of 2.8 and 2.7 respectively.
| Analysis of Maximum Likelihood Estimates | |||
|---|---|---|---|
| Parameter | Hazard | 95% Hazard Ratio Confidence Limits | |
| Ratio | |||
| METASTATIC DISEASE AT DIAGNOSIS | 6.13 | 4.01 | 9.37 |
| PENETRATION OF THE BOWEL WALL | 2.78 | 1.09 | 7.12 |
| LYMPH NODE INVOLVEMENT | 2.65 | 1.6 | 4.38 |
| PERINEURAL INVASION | 2.29 | 1.45 | 3.6 |
| EMERGENT OPERATION | 1.79 | 1.12 | 2.87 |
| VASCULAR INVASION | 1.78 | 1.18 | 2.68 |
| INVOLVED SURGICAL MARGIN | 2.09 | 0.95 | 4.6 |
| HIGH GRADE DIFFERENTIATION | 1.16 | 0.73 | 1.85 |
| PERIOPERATIVE BLOOD TRANSFUSION | 1.14 | 0.73 | 1.78 |
Table 2: Hazard ratio of death from colorectal cancer by clinical and pathological variables
In the proportional hazards model the presence of perineural invasion conferred a HR of 2.3 and vascular invasion a HR of 1.8 in respect to colorectal cancer death. Likewise an emergent operation was associated with a HR of 1.8. An involved surgical margin was associated with a doubling of the risk of cancer related death but fell short of statistical significance. Tumour differentiation (low grade versus high grade) and the administration of a perioperative blood transfusion were not associated with an increased risk of cancer related death. Patient gender and mucinous histology showed no association with cancer outcome on univariate analysis and were not considered further.
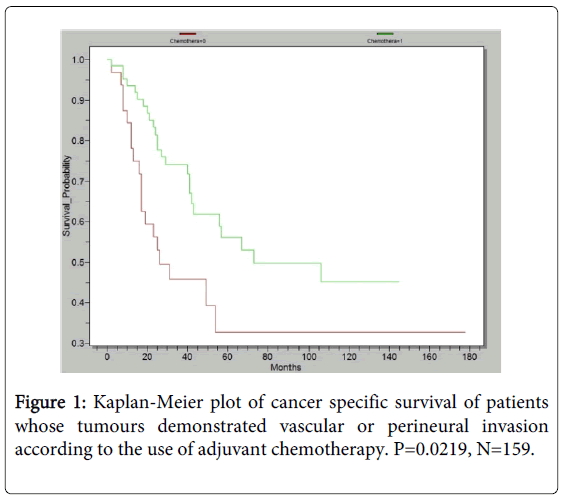
Although not specifically powered to look at the effect of adjuvant chemotherapy we were able to demonstrate that patients whose tumours demonstrated PNI, VI or both who received adjuvant chemotherapy demonstrated an improved cancer specific survival compared to those who did not (p=0.0219, Figure 1). Likewise patients with node positive tumours who received adjuvant chemotherapy had an improved cancer specific survival but this failed to reach statistical significance (N=17, p=0.123).
Twelve per cent of cases in this study had an emergent operation for obstruction, perforation or bleeding. Emergent surgery conferred a HR of 1.8 of cancer related death compared to patients receiving a resection on an elective list.
Discussion
It is now over eighty years since Dukes demonstrated that the depth penetration of the bowel wall and the status of the draining lymph nodes could be used as the basis for a staging system in rectal cancer that provided a useful guide to prognosis [1]. Various permutations of these two factors have remained the sole basis of nearly all colorectal cancer staging systems with the exception of the Jass system [4].
Accurate histopathological assessment remains central to the postoperative management of patients with CRC. In addition important clinical variables are known to affect cancer specific outcomes. Adjuvant chemotherapy of increasing efficacy is available to patients deemed to be at increased risk of recurrent disease after surgery [19]. Accurate risk stratification of patients following potentially curative surgery is essential to the rational use of the adjuvant chemotherapy regimens currently available [20]. The TNM system however remains a “blunt tool” with which to prognosticate as to the outcome of a given patient and as a guide to the utility of adjuvant chemotherapy.
This study confirms that penetration of the bowel wall and the status of draining lymph nodes are the two most important histological features in determining risk following resection of a CRC. However the hazard ratio (HR) of death from CRC associated with the identification of perineural invasion in the resected specimen is not numerically dissimilar to the presence of lymph node involvement (2.3 versus 2.7). The presence of vascular invasion and the requirement for an emergent operation carry a numerically smaller but clinically and statistically significant increase in risk.
Although the first serious investigation of perineural invasion in rectal cancer was reported by Seefeld in 1943 [21] the significance of PNI in CRC has been overlooked or underreported in the surgical and pathological literature until recently [7,22]. PNI does not feature in the current Royal College of Pathologists minimum dataset for colorectal cancer [23] but is included in the Royal College of Pathologists of Australasia’s minimum dataset [24]. Perineural invasion has been reported to be an independent prognostic factor in resected CRC in the great majority of studies in which it has been assessed [16,25-29]. In this study PNI was present in 12% of resected colorectal cancers, lower than the 22% recorded by Liebig and 24% by Peng [26,28], and had a highly significant independent effect on cancer specific survival.
In series where CRC slides have been reread specifically to look for PNI the incidence rose from 2 % to 24% in some series, likewise where synoptic reporting of CRC is in use the incidence of reported PNI and VI increases dramatically [28,30].
In this study we identified the presence of PNI twice as commonly in rectal cancer specimens as in colon cancers (16.3% versus 9.3%).
Ueno has further refined the reporting of PNI in rectal cancer and shown the extent of PNI correlates with both local recurrence and survival in a stepwise fashion [29].
Vascular invasion has long been recognised to be of prognostic importance in colorectal cancer [31] although some authors report that only extramural venous invasion to be of prognostic value [15,32]. The incidence of VI in this study is similar to other reported studies in which it has been looked for [15]. As is the case for PNI synoptic reporting of CRC specimens significantly increases the rate of VI reported by pathologists [30]. Although VI worsens the prognosis of CRC regardless of stage it’s identification in early stage disease is arguably more important as it indicates the need for adjuvant chemotherapy in node negative cases and a similar arguement can be made for node negative tumours displaying PNI [33-35]. The rate of PNI and VI in early stage disease is however correspondingly low.
Emergent surgery was an independent risk factor for cancer related death, presumably due to a more rapidly dividing tumour being more likely to result in an emergency presentation. The size of this effect was significant and of a similar importance to the finding of vascular invasion in the resected specimen. Both colonic perforation and obstruction have previously been shown to be associated with a worse cancer specific survival than elective cases [9,36-38].
Tumour grade has been shown in older studies to be an independent prognostic factor in CRC however in studies that include perineural invasion it tends to drop out on multivariate analysis as an independent risk factor for death [28,34,35,39,40]. There is however an association between PNI and differentiation with PNI positive tumours being more likely to be high grade than PNI negative tumours [29].
Perioperative blood transfusion has been shown in some studies to be associated with a deleterious effect on colorectal cancer recurrence and survival in patients undergoing curative resection [1,3,41]. We found that the transfusion of blood within two weeks of the tumour resection was not independently associated with an increased risk of cancer related death.
Conclusion
The use of the TNM staging system alone gives an incomplete picture of a patient’s risk of disease relapse and death after potentially curative surgery for colorectal cancer. The ideal staging system would allow patients to be divided into those cured by surgery alone and those that remain to be cured. Until we are able to individualise a patient’s treatment according to a comprehensive genetic profile for their individual tumour a post-surgical prognostic index, that incorporates the known clinical and pathological factors discussed above and their validated importance in risk assessment, may provide a more accurate estimate of the chance of surviving colorectal cancer after surgical resection and of the likely benefit of adjuvant chemotherapy. The PNI and VI status should be reported in all colorectal resection specimens, preferably in a synoptic report, as these tumour features are crucial in deciding on the merits of adjuvant therapy especially in early stage disease. Emergent surgery should be considered as a clinical factor suggesting the need for adjuvant therapy in the absence of other pathological indications.
At a research level further work now needs to centre on identification of the molecular basis of PNI in colorectal cancer and the identification of targets for therapeutic intervention.
References
- Dukes CE (1932) The classification of cancer of the rectum. Journal of pathology and bacteriology 35: 323-332.
- Edge SB, Byrd DD, Compton CC, Fritz AG, Greene FL, et al. (2010) AJCC Cancer Staging Manual 7th edition. editor: Springer.
- Davis NC, Newland RC (1983) Terminology and classification of colorectal adenocarcinoma: the Australian clinico-pathological staging system. Aust N Z J Surg 53: 211-221.
- Jass JR, Love SB, Northover JM (1987) A new prognostic classification of rectal cancer. Lancet 1: 1303-1306.
- Puppa G , Sonzogni A, Colombari R, Pelosi G (2010) TNM staging system of colorectal carcinoma: a critical appraisal of challenging issues. Arch Pathol Lab Med 134: 837-852.
- Compton CC , Greene FL (2004) The staging of colorectal cancer: 2004 and beyond. CA Cancer J Clin 54: 295-308.
- Newland RC , Dent OF, Lyttle MN, Chapuis PH, Bokey EL (1994) Pathologic determinants of survival associated with colorectal cancer with lymph node metastases. A multivariate analysis of 579 patients. Cancer 73: 2076-2082.
- Newland RC , Dent OF, Chapuis PH, Bokey L (1995) Survival after curative resection of lymph node negative colorectal carcinoma. A prospective study of 910 patients. Cancer 76: 564-571.
- Katoh H , Yamashita K, Wang G, Sato T, Nakamura T, et al. (2011) Prognostic significance of preoperative bowel obstruction in stage III colorectal cancer. Ann SurgOncol 18: 2432-2441.
- Cheynel N , Cortet M, Lepage C, Ortega-Debalon P, Faivre J, et al. (2009) Incidence, patterns of failure, and prognosis of perforated colorectal cancers in a well-defined population. Dis Colon Rectum 52: 406-411.
- Busch OR , Hop WC, Hoynck van Papendrecht MA, Marquet RL, Jeekel J (1993) Blood transfusions and prognosis in colorectal cancer. N Engl J Med 328: 1372-1376.
- Paulson EC , Wirtalla C, Armstrong K, Mahmoud NN (2009) Gender influences treatment and survival in colorectal cancer surgery. Dis Colon Rectum 52: 1982-1991.
- Ratto C , Sofo L, Ippoliti M, Merico M, Bossola M, et al. (1999) Accurate lymph-node detection in colorectal specimens resected for cancer is of prognostic significance. Dis Colon Rectum 42: 143-154.
- Tsai HL, Cheng KI, Lu CY, Kuo CH, Ma CJ, Wu JY, et al. (2008) Prognostic significance of depth of invasion, vascular invasion and numbers of lymph node retrievals in combination for patients with stage II colorectal cancer undergoing radical resection. J SurgOncol 97: 383-387.
- Courtney ED , West NJ, Kaur C, Ho J, Kalber B, et al. (2009) Extramural vascular invasion is an adverse prognostic indicator of survival in patients with colorectal cancer. Colorectal Dis 11: 150-156.
- Shirouzu K , Isomoto H, Kakegawa T (1993) Prognostic evaluation of perineural invasion in rectal cancer. Am J Surg 165: 233-237.
- Glasgow SC , Yu J, Carvalho LP, Shannon WD, Fleshman JW, et al. (2005) Unfavourable expression of pharmacologic markers in mucinous colorectal cancer. Br J Cancer 92: 259-264.
- Mulcahy HE , Toner M, Patchett SE, Daly L, O'Donoghue DP (1997) Identifying stage B colorectal cancer patients at high risk of tumor recurrence and death. Dis Colon Rectum 40: 326-331.
- Meyerhardt JA , Mayer RJ (2005) Systemic therapy for colorectal cancer. N Engl J Med 352: 476-487.
- Ueno H , Mochizuki H, Hashiguchi Y, Shimazaki H, Aida S, et al. (2004) Risk factors for an adverse outcome in early invasive colorectal carcinoma. Gastroenterology 127: 385-394.
- Seefeld PH, Bargen JA (1943) The Spread of Carcinoma of the Rectum: Invasion of Lymphatics, Veins and Nerves. Ann Surg 118: 76-90.
- Quirke P , Morris E (2007) Reporting colorectal cancer. Histopathology 50: 103-112.
- Standards and Datasets for reporting; Dataset for Colorectal Cancer (2nd edition). 2nd ed: The Royal College of Pathologists; 2007.
- Colorectal Cancer structured reporting protocol (1st edition). Royal College of Pathologists of Australasia; 2010.
- Krasna MJ , Flancbaum L, Cody RP, Shneibaum S, Ben Ari G (1988) Vascular and neural invasion in colorectal carcinoma. Incidence and prognostic significance. Cancer 61: 1018-1023.
- Liebig C , Ayala G, Wilks J, Verstovsek G, Liu H, et al. (2009) Perineural invasion is an independent predictor of outcome in colorectal cancer. J ClinOncol 27: 5131-5137.
- Poeschl EM, Pollheimer MJ, Kornprat P, Lindtner RA, Schlemmer A, et al. (2010) Perineural invasion: correlation with aggressive phenotype and independent prognostic variable in both colon and rectum cancer. J ClinOncol 28: e358-360.
- Peng J , Sheng W, Huang D, Venook AP, Xu Y, et al. (2011) Perineural invasion in pT3N0 rectal cancer: the incidence and its prognostic effect. Cancer 117: 1415-1421.
- Ueno H , Hase K, Mochizuki H (2001) Criteria for extramural perineural invasion as a prognostic factor in rectal cancer. Br J Surg 88: 994-1000.
- Messenger DE, McLeod RS, Kirsch R(2011) What impact has the introduction of a synoptic report for rectal cancer had on reporting outcomes for specialist gastrointestinal and nongastrointestinal pathologists? Arch Pathol Lab Med 135: 1471-1475.
- Amato A , Pescatori M (2006) Perioperative blood transfusions for the recurrence of colorectal cancer. Cochrane Database Syst Rev : CD005033.
- Talbot IC, Ritchie S, Leighton MH, Hughes AO, Bussey HJ, et al. (1980) The clinical significance of invasion of veins by rectal cancer. Br J Surg 67: 439-442.
- Stewart CJ , Morris M, de Boer B, Iacopetta B (2007) Identification of serosal invasion and extramural venous invasion on review of Dukes' stage B colonic carcinomas and correlation with survival. Histopathology 51: 372-378.
- Losi L , Ponti G, Gregorio CD, Marino M, Rossi G, et al. (2006) Prognostic significance of histological features and biological parameters in stage I (pT1 and pT2) colorectal adenocarcinoma. Pathol Res Pract 202: 663-670.
- Huh JW , Kim HR, Kim YJ (2010) Lymphovascular or perineural invasion may predict lymph node metastasis in patients with T1 and T2 colorectal cancer. J GastrointestSurg 14: 1074-1080.
- Guerra A , Borda F, Javier Jiménez F, Martinez-Peñuela JM, Larrínaga B (1998) Multivariate analysis of prognostic factors in resected colorectal cancer: a new prognostic index. Eur J GastroenterolHepatol 10: 51-58.
- Khan S , Pawlak SE, Eggenberger JC, Lee CS, Szilagy EJ, et al. (2001) Acute colonic perforation associated with colorectal cancer. Am Surg 67: 261-264.
- Mitchell AD , Inglis KM, Murdoch JM, Porter GA (2007) Emergency room presentation of colorectal cancer: a consecutive cohort study. Ann SurgOncol 14: 1099-1104.
- Desolneux G , Burtin P, Lermite E, Bergamaschi R, Hamy A, et al. (2010) Prognostic factors in node-negative colorectal cancer: a retrospective study from a prospective database. Int J Colorectal Dis 25: 829-834.
- Fujita S , Shimoda T, Yoshimura K, Yamamoto S, Akasu T, et al. (2003) Prospective evaluation of prognostic factors in patients with colorectal cancer undergoing curative resection. J SurgOncol 84: 127-131.
- Jadallah F , McCall JL, van Rij AM (1999) Recurrence and survival after potentially curative surgery for colorectal cancer. N Z Med J 112: 248-250.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 13568
- [From(publication date):
December-2014 - Sep 22, 2024] - Breakdown by view type
- HTML page views : 9218
- PDF downloads : 4350