Atypical DRESS Syndrome Induced by Lenalidomide in Chronic Hemodialysis
Received: 15-Apr-2016 / Accepted Date: 07-Jun-2016 / Published Date: 09-Jun-2016 DOI: 10.4172/2161-0681.1000277
Keywords: DRESS syndrome; Lenalidomide; Hemodialysis; Hypersensitivity syndrome; Eosinophilia
314959Introduction
Drug adverse reactions manifested with cutaneous eruptions are not uncommon in chronic hemodialysis. Drug Reaction with Eosinophilia and Systematic Symptoms (DRESS) syndrome is a prime example of a rare, potentially fatal cutaneous reaction attributed to drug hypersensitivity. It is typically characterized by a long latency period of two weeks to two months from drug exposure to rash emergence. The term was introduced by Bocquet et al. in 1996 [1]. Due to the variability in clinical presentation and the lack of clear diagnostic criteria imperfect definitions of this syndrome had been initially used such as “anticonvulsant hypersensitivity syndrome” and “drug induced hypersensitivity syndrome”. DRESS frequency has been reported to be 1 case among 1,000 to 10,000 drug exposures. In a prospective sevenyear study, the annual incidence of DRESS syndrome in the general population was estimated at 0.9/100,000 [2]. Numerous drugs have been associated with DRESS syndrome. In a comprehensive literature review, 44 culprit drugs related to 172 case reports over a 12-year period were detected. The majority of cases were found to be related to aromatic anticonvulsants (carbamazepine, phenytoin, phenobarbital), allopurinol and sulfasalazine administration [3]. Clinical manifestations of DRESS syndrome include fever, lymphadenopathy, cutaneous eruption, hematologic abnormalities (eosinophilia, atypical lymphocytosis) and internal organ involvement, primarily of the liver. Data regarding DRESS syndrome in chronic hemodialysis patients are limited and the possible influence of uremia on the natural history of the syndrome has not been explored [4]. Two sets of diagnostic criteria have been independently introduced by the Registry of Severe Cutaneous Adverse Reactions (RegiSCAR) study group and the Japanese consensus group to aid in the diagnosis and classification of suspected cases [5,6].
Lenalidomide, a derivative of thalidomide, is an immunomodulatory drug, which is indicated for treatment of multiple myeloma (MM) and myelodysplastic syndromes. The use of lenalidomide has been consistently associated with the development of skin rash. In a systematic review including 10 studies with 737 patients, the incidence of lenalidomide-associated all-grade rash was 27.2% [7]. Although Stevens-Johnson syndrome (SJS) is the most common cutaneous adverse reaction caused by lenalidomide [8], recent case reports have also linked this medication with DRESS syndrome [9,10]. To our knowledge, this is the first reported case of DRESS syndrome in a chronic hemodialysis patient five days after the lenalidomide commencement.
Case Report
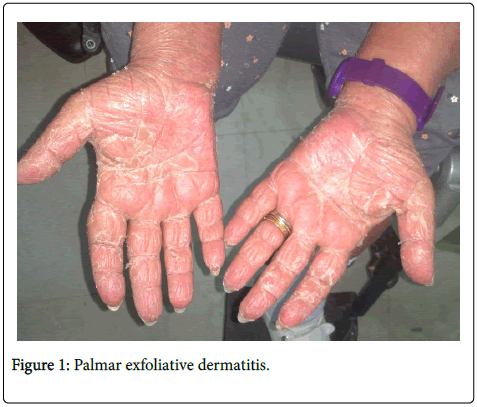
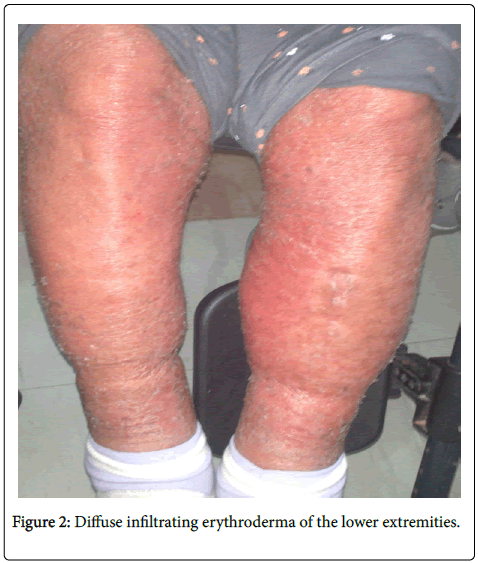
A 62-year-old female on chronic hemodialysis (HD) was admitted for fever (38.5°C) and acute cutaneous eruption. She had been simultaneously diagnosed with end-stage renal disease and MM fifteen months ago for which she commenced treatment with bortezomib and dexamethasone. Five months ago, she developed progressive paralysis of the lower extremities due to spinal cord compression. Bortezomib and dexamethasone were discontinued and she underwent radiotherapy. Radiotherapy was complicated by bowel perforation treated with partial colectomy and palliative colostomy formation. Her latest medications consisted of oral furosemide, allopurinol, calcium carbonate, quetiapine, paroxetine, alprazolam and intravenous erythropoietin during HD sessions. Five days before admission the patient had been started on lenalidomide at a reduced renal dose of 5 mg per day to treat relapsed MM. The skin rash was characterized by diffuse, infiltrating erythema affecting equally the face, trunk and limbs. The patient also exhibited facial edema, pruritus, as well as mild cervical and axillary lymphadenopathy (Figures 1 and 2).
The rash soon progressed to extensive exfoliative erythroderma. Laboratory test results on admission were: Hemoglobin: 9.4 gr/dl (12.2-18.1), hematocrit: 32.4% (37.7-53.7), white blood cells: 12.300/mm3 (4.600-10.200) [neutrophils 54%, lymphocytes 13%, eosinophils 19%, monocytes 12%, basophils 2%], C-reactive protein: 37.1 mg/l (<5.0), urea: 123 mg/dl (10-50), creatinine: 9.9 mg/dl (0.5-1.5), sodium: 140 mmol/l (136-145), potassium: 5.3 mmol/l (3.5-5.2), calcium: 9.0 mg/dl (8.2-10.4), phosphate: 4.2 mg/dl (3.5-5.2), AST: 11 IU/l (8-40), ALT: 10 IU/l (8-45), total bilirubin: 0.2 mg/dl (0-1.5), alkaline phosphatase: 79 IU/l (25-130), gamma-GT: 9 IU/l (5-40), CK: 36 IU/l (26-135) and LDH: 426 IU/l (230-460). Blood cultures taken on admission came back negative. Serology for hepatotropic viruses and antinuclear antibodies was negative. Application of the RegiSCAR scoring system in this clinical setting yielded a score of 5 and led to the identification of a “probable” case of DRESS syndrome due to lenalidomide. The drug was promptly discontinued. Furosemide and allopurinol were also discontinued although they had been administered for many years and therefore their causative role seemed unlikely. The patient was treated with antihistamines and a brief course of oral corticosteroids. However, she failed to improve and the absolute eosinophil count remained over 2.500/mm3. Apart from the persistent skin rash, the patient also developed non-erosive oropharyngeal mucositis with accompanying dysphagia. The overall condition of the patient deteriorated rapidly mainly because of relapsed MM and comorbidities. She progressed to denying medical care and food intake and passed away eight weeks after disease onset.
Discussion
DRESS syndrome is a severe, immune-mediated idiosyncratic reaction to medications marked by the delayed onset after offending agent commencement. The pathogenesis of DRESS syndrome is still unclear. A strong association of DRESS syndrome with human herpes virus 6 (HHV-6) reactivation has been proposed [11]. This has led the Japanese consensus group to the inclusion of HHV-6 reactivation in the diagnostic criteria of DRESS syndrome. Defective drug detoxification process resulting in accumulation of toxic metabolites has also been suggested [12]. Furthermore, drug specific CD4+ and CD8+ T-cells have been shown to expand during the acute phase of the disease leading to the release of pro-inflammatoryinterleukin 5 [13]. Intriguingly, the expansion of regulatory T-cells has also been associated to HHV-6 reactivation and DRESS syndrome development [14]. The histologic pattern of skin rash in DRESS syndrome usually displays spongiosis, acanthosis and lymphocytic infiltration in the superficial dermis with few eosinophils [15]. Timely recognition of this entity is of utmost importance since it is associated with a significant mortality rate of 10%-30%. Fulminant hepatic failure has been reported to be the leading cause of death in DRESS syndrome [16].
The interesting features of the present case are the short latency period of 5 days (between lenalidomide commencement and rash onset) and the mild systemic symptoms - fever and lymphadenopathy. No signs of liver or other vital organ involvement were detected. In a recent review, Cacoub et al. reported that the latency period can be as short as half week whereas 12% of the patients have no internal organ involvement [3]. It is, therefore, obvious that atypical manifestations of DRESS syndrome may well occur and this should not impede clinicians from prompt diagnosis and removal of the putative culprit drug. Moreover, overlapping manifestations between DRESS syndrome and SJS may be observed, further complicating the diagnostic process. Severe mucosal involvement with erosions and bleeding occurs in over 90% of cases of SJS, whereas mucosal involvement without erosions occurs in approximately 50% of patients with DRESS syndrome. Most importantly, eosinophilia and atypical lymphocytosis are not observed in SJS. The exact mode of action of lenalidomide is incompletely understood. However, MM patients on lenalidomide exhibit regulatory T-cell activation [17] which has also been described as a pathogenetic mechanism for DRESS syndrome.
The mainstay of therapeutic management o DRESS syndrome is the withdrawal of the offending agent along with supportive care and topical or systemic corticosteroids according to severity of internal organ involvement. Systemic corticosteroids can be administered via the oral or intravenous route. Cyclosporine and intravenous immunoglobulin (IVIG) have been used in steroid-resistant patients in isolated case reports [18]. Persistence or aggravation of clinical condition despite removal of the offending agent is advocated as a unique feature of DRESS syndrome. Our patient developed dysphagia due to oropharyngeal mucositis, despite treatment. We propose that early nutritional support should be considered even in seemingly mild cases without internal organ involvement. Concomitant administration of antibiotics and anti-inflammatory drugs should generally be avoided as they may complicate clinical presentation with superimposed drug adverse reactions. Moreover, cross-reacting drugs should also be strictly avoided. Cross-reactivity between chemically similar anticonvulsants has been documented in association with skin rash, especially involving carbamazepine and phenytoin [19].
Conclusion
We reported a rare, atypical case of DRESS syndrome in a chronic hemodialysis patient and recognized lenalidomide as the culprit medication. DRESS syndrome diagnosis necessitates a high index of suspicion because of the extended variability in clinical presentations. Immediate discontinuation of the suspected offending agent is a sine qua non for proper management. A multidisciplinary approach potentially involving dermatologists and clinical pharmacists apart from the attending physicians is often needed to attain successful outcomes.
References
- Bocquet H, Bagot M, Roujeau JC (1996) Drug-induced pseudolymphoma and drug hypersensitivity syndrome (Drug Rash with Eosinophilia and Systemic Symptoms: DRESS). SeminCutan Med Surg 15: 250-257.
- Muller P, Dubreil P, Mahé A, Lamaury I, Salzer B, et al. (2003) Drug Hypersensitivity Syndrome in a West-Indian population. Eur J Dermatol 13: 478-481.
- Cacoub P, Musette P, Descamps V, Meyer O, Speirs C, et al. (2011) The DRESS syndrome: a literature review. Am J Med 124: 588-597.
- Vlachopanos G (2012) Phenytoin-Related DRESS Syndrome in a Hemodialysis Patient. BANTAO Journal 10: 43-45.
- Kardaun SH, Sidoroff A, Valeyrie-Allanore L, Halevy S, Davidovici BB, et al. (2007) Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: does a DRESS syndrome really exist? Br J Dermatol 156: 609-611.
- Shiohara T, Iijima M, Ikezawa Z, Hashimoto K (2007) The diagnosis of a DRESS syndrome has been sufficiently established on the basis of typical clinical features and viral reactivations. Br J Dermatol 156: 1083-1084.
- Nardone B, Wu S, Garden BC, West DP, Reich LM, et al. (2013) Risk of rash associated with lenalidomide in cancer patients: a systematic review of the literature and meta-analysis. Clin Lymphoma Myeloma Leuk 13: 424-429.
- Castaneda CP, Brandenburg NA, Bwire R, Burton GH, Zeldis JB (2009) Erythema multiforme/Stevens-Johnson syndrome/toxic epidermal necrolysis in lenalidomide-treated patients. J ClinOncol 27: 156-157.
- Foti C, Antelmi A, Mazzocca A, Vella SF, Romita P, et al. (2012) Drug reaction with eosinophilia and systemic symptoms caused by lenalidomide. Eur J Dermatol 22: 799-800.
- Shaaban H, Layne T, Guron G (2013) A case of DRESS (drug reaction with eosinophilia and systemic symptoms) with acute interstitial nephritis secondary to lenalidomide. J Oncol Pharm Pract 20: 302-304.
- Descamps V, Valance A, Edlinger C, Fillet AM, Grossin M, et al. (2001) Association of human herpesvirus 6 infection with drug reaction with eosinophilia and systemic symptoms. Arch Dermatol 137: 301-304.
- Shear NH, Spielberg SP, Grant DM, Tang BK, Kalow W (1986) Differences in metabolism of sulfonamides predisposing to idiosyncratic toxicity. Ann Intern Med 105: 179-184.
- Choquet-Kastylevsky G, Intrator L, Chenal C, Bocquet H, Revuz J, et al. (1998) Increased levels of IL5 are associated with generation of eosinophilia in drug-induced hypersensitivity syndrome. Br J Dermatol 139: 1026-1032.
- Takahashi R, Kano Y, Yamazaki Y, Kimishima M, Mizukawa Y, et al. (2009) Defective regulatory T cells in patients with severe drug eruptions: timing of the dysfunction is associated with the pathological phenotype and outcome. J Immunol 182: 8071-8079.
- Chi MH, Hui RC, Yang CH, Lin JY, Lin YT, et al. (2014) Histopathological analysis and clinical correlation of drug reaction with eosinophilia and systemic symptoms (DRESS). Br J Dermatol 170: 866-873.
- Zaccara G, Franciotta D, Perucca E (2007) Idiosyncratic adverse reactions to antiepileptic drugs. Epilepsia 48: 1223-1244.
- Galustian C, Meyer B, Labarthe MC, Dredge K, Klaschka D, et al. (2009) The anti-cancer agents lenalidomide and pomalidomide inhibit the proliferation and function of T regulatory cells. Cancer ImmunolImmunother 58: 1033-1045.
- Zuliani E, Zwahlen H, Gilliet F, Marone C (2005) Vancomycin-induced hypersensitivity reaction with acute renal failure: resolution following cyclosporine treatment. ClinNephrol 64: 155-158.
- Hirsch LJ, Arif H, Nahm EA, Buchsbaum R, Resor SR Jr, et al. (2008) Cross-sensitivity of skin rashes with antiepileptic drug use. Neurology 71: 1527-1534.
Citation: Vlachopanos G, Kokkona A, Zerva A, Stavroulaki E, Zacharogiannis C, et al. (2016) Atypical DRESS Syndrome Induced by Lenalidomide in Chronic Hemodialysis. J Clin Exp Pathol 6:277. DOI: 10.4172/2161-0681.1000277
Copyright: © Vlachopanos G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 15488
- [From(publication date): 6-2016 - Jul 16, 2025]
- Breakdown by view type
- HTML page views: 14430
- PDF downloads: 1058