Blastoid Mantle Cell Variant of Richter’s Transformation
Received: 18-Mar-2019 / Accepted Date: 25-Mar-2019 / Published Date: 30-Mar-2019 DOI: 10.4172/2161-0681.1000365
Abstract
An unusual occurrence of blastoid variant of mantle cell lymphoma arising in a patient with untreated chronic lymphocytic leukemia/small lymphocytic lymphoma is described. A localized site of intense uptake on positron emission tomography suggested Richter's transformation. Biopsy of the suspect area showed the blastoid variant of mantle cell lymphoma. Immunohistochemistry and fluorescence in situ hybridization confirmed the identity of both tumors. Molecular studies established that the mantle cell lymphoma and chronic lymphocytic leukemia/small lymphocytic lymphoma were clonally related.
Keywords: Chronic lymphocytic leukemia; Richter transformation; Mantle cell lymphoma; Blastoid variant mantle cell lymphoma; Positron emission tomography
Introduction
Richter's transformation (RT) is usually defined as the occurrence of diffuse large B-cell lymphoma (DLBCL) arising in patients with chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/ SLL). Transformation to the Hodgkin lymphoma variant is a considerably less frequent event. We report the unusual occurrence of blastoid mantle cell lymphoma (MCL) variant of Richter's transformation in a previously untreated patient with CLL/SLL. Transformation was signaled by interval changes in fluorodeoxyglucose (18FDG) positron emission tomography.
Case Report
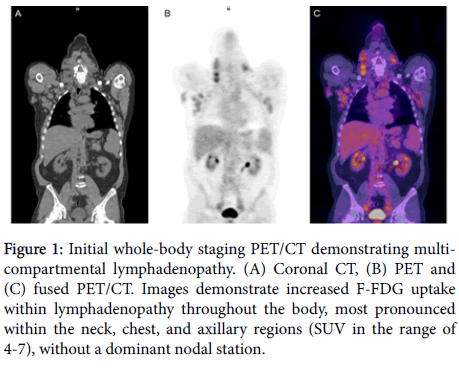
A sixty-six year old man was referred because of persistent lymphadenopathy. He was without constitutional symptoms. Examination revealed extensive adenopathy. The absolute lymphocyte count was 184,000/mm³, hemoglobin 12.6 gms/dL, and the platelet count was 254,000 mm³. The lactate dehydrogenase (LDH) was 836 IU/L (normal range 87-241). A PET-CT (Figure 1) was obtained, owing to the elevated LDH, and showed adenopathy in the neck, chest, upper abdomen, pelvis, mesentery, retroperitoneum, axillae and inguinal regions. There was no dominant area of FDG uptake and all standard uptake values were in the range of 3-7. A bone marrow biopsy and aspirate disclosed sheets of small lymphocytes. Flow cytometry showed B cells, CD5/19 positive, and trisomy 12 was documented by FISH. The IGVH gene was mutated. A diagnosis of CLL/SLL was made. The patient preferred continued observation rather than treatment.
Figure 1: Initial whole-body staging PET/CT demonstrating multicompartmental lymphadenopathy. (A) Coronal CT, (B) PET and (C) fused PET/CT. Images demonstrate increased F-FDG uptake within lymphadenopathy throughout the body, most pronounced within the neck, chest, and axillary regions (SUV in the range of 4-7), without a dominant nodal station.
Eighteen months later he presented with weight loss and bulky adenopathy. The spleen and liver were enlarged. The absolute lymphocyte count was 299,000/mm³, hemoglobin 9.3 gms/dL and platelets were 248,000/mm³. Studies performed on bone marrow aspirate showed mutated IgHV genes. NOTCH1 and SF3B1 were detected by sequencing. Trisomy 12 was the only cytogenetic abnormality detected. Mutation in TP53 or MYD88 was not detected.
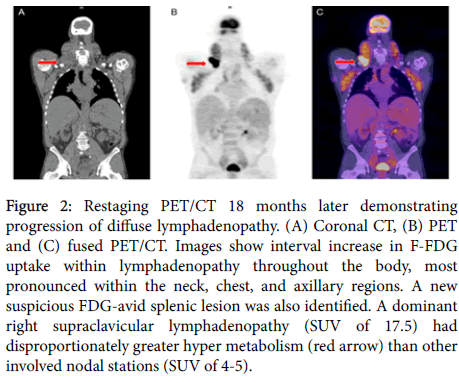
A repeat PET-CT (Figure 2) disclosed progressive disease throughout, with interval increase in the size of pre-existing nodal masses. Of particular note, there was a dominant area of disproportionately intense FDG uptake (SUV 17.5) in right supraclavicular adenopathy seen at the root of the neck (Figures 2B and 2C). Other sites of adenopathy had shown hyper metabolism similar to the previous PET/CT in the range of 3-7. Because of a concern for Richter's transformation, an incisional biopsy of the mass was performed as well as a repeat bone marrow examination.
Figure 2: Restaging PET/CT 18 months later demonstrating progression of diffuse lymphadenopathy. (A) Coronal CT, (B) PET and (C) fused PET/CT. Images show interval increase in F-FDG uptake within lymphadenopathy throughout the body, most pronounced within the neck, chest, and axillary regions. A new suspicious FDG-avid splenic lesion was also identified. A dominant right supraclavicular lymphadenopathy (SUV of 17.5) had disproportionately greater hyper metabolism (red arrow) than other involved nodal stations (SUV of 4-5).
Pathology
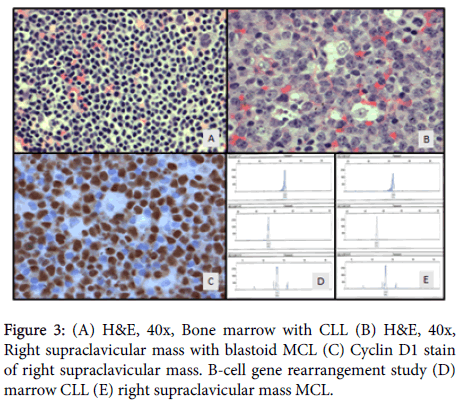
The bone marrow was markedly hypercellular and diffusely (>90%) involved by small lymphoid cells (Figure 3A) that were positive by immunohistochemistry (IHC) for CD5, CD20, PAX-5 and LEF-1 and negative for Cyclin D1 (BCL-1) and SOX-11. There was no significant increase in prolymphocytes or morphologic evidence of transformation to large cell lymphoma. Flow cytometry analysis performed on bone marrow and peripheral blood showed CD5 positive B-cell lymphoma with coexpression of CD5/CD23 immunophenotype consistent with CLL/SLL. CLL FISH analysis demonstrated trisomy 12, however, t (11:14) IgH/BCL1 fusion was not detected on either the bone marrow or peripheral blood.
Sections of the more intensely FDG avid (SUV 17.5) right supraclavicular lymph node revealed diffusely arranged, neoplastic, medium to large lymphoid cells with a blastic appearance resembling centroblasts with some showing prominent nucleoli. Neoplastic blastoid cells showed no significant nuclear pleomorphism. There was no evidence of normal nodal architecture, well-formed follicles, granulomas or necrosis. Foci of scattered epithelioid histiocytes mimicking starry sky pattern (Figure 3B) were present. IHC stains were performed to further characterize the lymph proliferative process. The large neoplastic blastoid cells were positive for CD20, PAX-5, CD5, Cyclin D1/BCL-1 (Figure 3C), LEF1, SOX11, MUM1, BCL2, and a Ki-67 proliferation rate of 90-95%. The large lymphoid cells were negative CD10, BCL6 and p53. cMYC was positive by FISH in 60-70% of B cells. FISH results were positive for t (11:14) IgH/BCL1 fusion but negative for BCL6, MYC, and DUSP22/IRF4 gene rearrangements. MYC amplification and IgH/BCL2 fusion were also negative by IHC. Based upon these findings, a diagnosis of blastoid variant of mantle cell lymphoma was made on the supraclavicular lymph node specimen.
To assess the relatedness of the two lymphomas, DNA was isolated from both the marrow core and lymph node paraffin-embedded tissue blocks, purified and subjected to PCR amplification using BIOMED-2 oligonucleotide consensus primers specific for the IgH gene Framework Regions 1, 2, 3 and joining regions. In addition, Ig Kappa light chain gene rearrangement analysis was performed using specific oligonucleotides recognizing the Vκ intragenic and Jκ regions (data not shown). PCR products were separated by capillary gel electrophoresis and compared with band pattern and sizes. Both marrow and right supraclavicular lymph node showed identical gene rearrangement band pattern and sizes, indicating CLL/SLL cells from the marrow and those of blastoid variant of mantle cell lymphoma are clonally related (Figures 3D and 3E).
Discussion
Richter's transformation occurs in 2-10% of patients with CLL/SLL [1]. It may occur early in the course of disease, although patients having had multiple treatment courses, and certain tumor genetic characteristics, are at increased risk [2,3]. Such transformation is associated with resistance to chemotherapy and, thus far, a guarded outlook [4].
DLBCL is the most common type of Richter's transformation. Such tumors may, or may not, be clonally related to the CLL [5]. The other recognized pathologic variant of RT is Hodgkin lymphoma, which is clonally related to CLL in roughly half the cases [6]. Puckrin et al. recently described a second patient with intravascular large B-cell lymphoma which was clonally related to the CLL [7].
Both MCL and CLL are thought to arise from CD5+ mature B-cells. They share similar phenotypic characteristics and clinical features, including involvement of lymphoid and non-lymphoid organs, blood and bone marrow. While both lymphomas may evolve in an indolent manner, either can be aggressive depending upon genomic and epigenomic alterations.
To some degree, both tumors share driver mutations and signaling pathways [8].
The blastoid variant of MCL has been recognized as a particularly aggressive histological subtype of MCL that is often refractory to chemoimmunotherapy [9]. That variant is usually recognized at the time of presentation, but may evolve from the classic cytologic form of MCL [10].
MCL and CLL have previously been recognized as composite lymphomas, demarcated within the same anatomic site [11]. Despite some shared clinical features and the same putative cells of origin, transformation of CLL to MCL is a rare event. Kourelis et al. described a patient with simultaneously diagnosed MCL and CLL [12]. In that instance it appeared to be a synchronous presentation of the two tumor types. Klener et al. described a clonally related MCL that evolved over four years in a patient with CLL [13]. In that study, transformation to MCL occurred in a step wise manner with the acquisition of mutations in multiple genes, and was not simply due to the emergence of t (11:14) IgH/BCL-1 Fusion.
In the present case, separate analysis of bone marrow and peripheral blood confirmed the diagnosis of CLL and disclosed no evidence of MCL. FISH analysis of peripheral blood and bone marrow were negative for t (11:14). IHC stains with high specificity towards mantle cell lymphoma, SOX11 and Cyclin D1, were positive on the FDG avid lymph node as well as positive by FISH t (11:14) IgH/BCL1 Fusion. The evolution of the MCL at a discrete site, indicated by serial FDG/PET imaging, suggested that the MCL represented a transformation. Molecular analysis confirmed that both tumors were clonally related.
Both clinical and genetic features influence the risk of Richter's transformation. Mutations in NOTCH1, present in the patient described, emerge with, or without, selection by exposure to treatment, and has a deleterious effect on disease progression [14]. NOTCH1 mutations are associated with trisomy 12 [15] and markedly increase the risk of transformation to clonally related DLBCL [16,17]. How NOTCH1 contributes to transformation in uncertain [18].
The utility of PET/CT in the detection of Richter's transformation is well documented [19,20]. The intensity of FDG avidity at a potential site of transformation typically exceeds an SUV of 5. In the present case, the conspicuous site of FDG uptake demonstrated an SUV of 17.5, which prompted biopsy.
Conclusion
Richter's transformation of CLL/SLL to the blastoid variant of mantle cell lymphoma has not been previously reported. That unusual event evolved in the setting of untreated CLL/SLL with high risk features; advanced Rai stage, elevated LDH, trisomy 12, and mutated NOTCH1. PET imaging detected the area of transformation, which then led to incisional biopsy and repeat staging. Molecular studies confirmed the clonal relatedness of the MCL and CLL/SLL.
References
- Parikh SA, Kay NE, Shanafelt TD (2014) How we treat Richter syndrome. Blood 123: 1647-1657.
- Robertson LE, Pugh W, O'Brien S, Kantarjian H, Hirsch Ginsberg C, et al. (1993) Richter's Syndrome: A report on 39 patients. J Clin Oncol 11: 1985-1989.
- Parikh SA, Shanafelt TD (2014) Risk factors for Richter syndrome in chronic lymphocytic leukemia. Curr Hematol Malig Rep 9: 294-299.
- Khan M, Siddiqi R, Thompson PA (2018) Approach to Richer transformation of chronic lymphocytic leukemia in the era of novel therapies. Ann Hematol 97: 1-15.
- Rossi D, Spina V, Gaidano G (2018) Biology and treatment of Richter syndrome. Blood 131: 2761-2772.
- Xiao W, Chen WW, Sorbara L, Davies-Hill T, Pittaluga S, et al. (2016) Hodgkin lymphoma variant of Richter transformation: Morphology, EBV status, clonality and survival analysis-with comparison to Hodgkin-like lesion. Hum Pathol 55: 108-116.
- Puckrin R, Pop P, Ghorab Z, Keith J, Chodirker L, et al. (2017) Intravascular large B-cell lymphoma presenting as Richter's syndrome with cerebral involvement in a patient with chronic lymphocytic leukemia. Clin Case Rep 9: 1444-1449.
- Puente X, Jares P, Campo E (2018) Chronic lymphocytic leukemia and mantle cell lymphoma: crossroads of genetic and microenvironmental interactions. Blood 131; 2283-2296.
- Bernard M, Gressin R, Lefrère F, Drénou B, Branger B, et al. (2001) Blastic variant of mantle cell lymphoma: A rare but highly aggressive subtype. Leukemia 15: 1785-1791.
- Laszlo T, Matolcsy A (1999) Blastic transformation of mantle cell lymphoma: Genetic evidence for a clonal link between the two stages of the tumor. Histopathology 35: 355-359.
- Sun Y, Wang SA, Sun T (2018) Composite mantle cell lymphoma and chronic lymphocytic leukemia/small lymphocytic lymphoma. Cytometry B Clin Cytom 94: 148-150.
- Kourelis TV, Kahl BS, Benn P, Delach JA, Bilgrami SF (2011) Treatment of synchronous mantle cell lymphoma and small lymphocytic lymphoma with bendamustine and rituximab. Acta Haematol 126: 40-43.
- Klener P, Fronkova E, Berkova A, Jaksa R, Lhotska H, et al. (2016) Mantle cell lymphoma-variant Richter syndrome: Detailed molecular-cytogenetic and backtracking analysis reveals slow evolution of a pre-MCL clone in parallel with CLL over several years. Int J Cancer 139: 2252-60.
- Nadeu F, Delgado J, Royo C, Baumann T, Stankovic T, et al. (2016) Clinical impact of clonal and subclonal TP53, SF3B1, BIRC3, NOTCH1 and ATM mutations in chronic lymphocytic leukemia. Blood 127: 2122-2130.
- Balatti V, Bottoni A, Palamarchuk A, Alder H, Rassenti LZ, et al. (2012) NOTCH1 mutations in CLL associated with trisomy 12. Blood 119: 329-331.
- Rossi D, Rasi S, Spina, V, Fangazio M, Monti S, et al. (2012) Different impact of NOTCH1 and SF3B1 mutations on the risk of chronic lymphocytic leukemia transformation to Richter syndrome. Brit Journ Haematol 158: 415-429.
- Villamor N, Conde L, Martinez Trillos A, Cazorla M, Navarro A, et al. (2013) NOTCH1 mutations identify a genetic subgroup of chronic lymphocytic leukemia patients with high risk of transformation and poor outcome. Leukemia 27: 1100-1106.
- Fabbri G, Rasi S, Rossi D, Trifonov V, Khiabanian H, et al. (2011) Analysis of the chronic lymphocytic leukemia coding genome: Role of NOTCH1 mutational activation. J Exp Med 208: 1389-1401.
- Bruzzi JF, Macapinlac H, Tsimberidou AM, Truong MT, Keating MJ, et al. (2006) Detection of Richter's transformation of chronic lymphocytic leukemia by PET/CT. J Nucl Med 47: 1267-1273.
- Shaikh F, Janjua A, Gestel FV, Adeel A (2017) Richter transformation of chronic lymphocytic leukemia: A review of fluorodeoxyglucose positron emission tomography-computed tomography and molecular diagnostics. Cureus 9: e96.
Citation: Prichard JG, Chen WW, Shah MC, Tobar DE, Wong EH (2019) Blastoid Mantle Cell Variant of Richter's Transformation. J Clin Exp Pathol 9: 365. DOI: 10.4172/2161-0681.1000365
Copyright: © 2019 Prichard JG, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 3610
- [From(publication date): 0-2019 - Nov 15, 2025]
- Breakdown by view type
- HTML page views: 2745
- PDF downloads: 865