Expression of Silent Information Regulator-1(Sirt1), CD163 and Tryptase; Implications for Immune Dys-regulation, Prognosis and Therapeutic Targeting in Classical Hodgkin Lymphoma
Received: 27-May-2017 / Accepted Date: 20-Jun-2017 / Published Date: 26-Jun-2017 DOI: 10.4172/2161-0681.1000313
Abstract
Background: Many studies have assessed the relation between cHL histology and its clinical outcome but the results are still controversial. The most recent issue was the discovery of the methods of interaction between the elements of benign reactive inflammatory cells and their effect on cHL metabolism. Silent information regulator-1 (Sirt1) is a class III HDACs family member, it was expressed in T-cell lymphoma and was considered a recently discovered therapeutic target, we tried to detect that the significance of Sirt1 expression in malignant Reed-Sternberg cells and surrounding inflammatory infiltrate in cHL. Cluster of Differentiation 163 (CD163) is a scavenger receptor cystein-rich (SRCR) family member which identifies monocytes and macrophages, Tryptase is the secretory granules that were serine proteinase derived and it has been used as a mast cell activation marker. The tumor-infiltrating macrophages and mast cells have been found to have different roles in malignancies of many organs but their role in cHL prognosis had not been sufficiently clarified.
Aim of the work: To assess the influence of Sirt1 expression in the malignant cells and the surrounding tumor microenvironment CD163 positive macrophages and tryptase positive mast cells on the pathological parameters and clinical outcome in cHL patients.
Methods: We evaluated clinicopathological and prognostic significance of Sirt1, CD163 and tryptase expressions in sections from fifty paraffin blocks of cHL using immunehistochemistry.
Results: The expression of Sirt1 in cHL was associated with advanced stage of the disease, presence of B symptoms, splenic affection, (p<0.001), bone marrow infiltration (p=0.002) and the presence of bulky mediastinal lymph node (p=0.004). The expression of CD163 and tryptase in cHL was associated with advanced stage of the disease and the presence of bulky mediastinal lymph node (p<0.001). High Sirt1, tryptase and CD163 expression was associated with poor response to therapy, high incidence of relapse after successful therapy and poor three-year overall survival rate (p<0.001).
Conclusion: High levels of expression of Sirt1, CD163 and tryptase were found to be markers of poor prognosis in cHL.
Keywords: Classical hodgkin lymphoma; Sirt1; CD163; Tryptase; Immunohistochemistry; Prognosis
316098Introduction
Classical Hodgkin lymphoma (cHL) forms ninety percent of all Hodgkin lymphomas and it is categorized into 4 subtypes; lymphocyte-rich (LR), nodular sclerosis (NS), mixed cellularity (MC) and lymphocyte-depletion (LD) subtype [1]. CHL is a treatable cancer [2], but still few patients are resistant to therapy or they may have lymphoma recurrence after successful therapy [3]. Novel researches had focused on recent therapeutic targets detection to improve the response to therapy and to decrease the incidence of lymphoma recurrence. CHL is composed of malignant giant cells i.e. Reed-Stenberg (RS) cells that are surrounded by reactive inflammatory infiltrate [4], e.g. lymphocytes, eosinophils, mast cells, macrophages which interact with RS cells via many cellular pathways and by production of cytokine by RS cells that affect the benign reactive inflammatory cells via autocrine and paracrine pathways [5,6]. Previous studies have focused on the cellular composition of cHL microenvironment to detect their clinicopathological and prognostic role, but conflicting results were detected. Silent information regulator-1 (Sirt1) is a class III HDACs family member that has a role in the gene silencing of yeast [7]. It was expressed in T-cell lymphoma and was considered a recent therapeutic target [8]. Our study attempted to acquire more understanding of the pathogenesis of cHL. As previously found that the RS cells and the surrounding reactive lymphocytes could express Sirt1 [9] we tried to detect the clinicopathological role of such expression in our patients. A more understanding of cHL microenvironment, may allow better management via restoration of the immune balance that could allow its better remission. Cluster of Differentiation 163 (CD163) is a protein that is encoded by the CD163 gene [10], it is a scavenger receptor cystein-rich (SRCR) family member [11,12], which has been found to mark macrophages [13]. Tryptase enzyme is detected in the secretory granules that were serine proteinase derived, were contained in mast cells and it has been used as a mast cell activation marker [14,15]. Increased numbers of tryptase positive mast cells had found to accompany lympho-plasmacytic lymphoma (LPL) [16]. Researchers had described the different roles of mast cells infiltration in cHL and the relations between increased mast cell infiltration and disease progression [17], but still conflicting results were found. Little information was proved regarding Sirt1 expression in cHL [18]. The tumor-infiltrating macrophages have been found to have different roles in malignancies of many organs, but their role in cHL prognosis had not been sufficiently clarified [19], and the mast cells infiltration in cHL has been found to have different conflicting roles in the prognosis in adults’ patients [20,21].
Aim of the work
To assess the influence of Sirt1 expression in the malignant cells and the surrounding tumor microenvironment CD163 positive macrophages and tryptase positive mast cells on the pathological parameters and clinical outcome in cHL patients.
Patients and Methods
We evaluated expressions of Sirt1, CD163 and tryptase in sections from fifty paraffin blocks retrieved from fifty cases of cHL using immunohistochemistry. We have collected the paraffin blocks from Pathology Department, Faculty of Medicine, Zagazig University in the period from December 2010 to December 2015. We used REAL/WHO and Cotswolds-modified Ann Arbor staging system for classification and staging of cHL [22,23]. We detected the data of patient as sex, age, lymphoma size, stage and the presence of B symptoms by retrospective examination of the patient’s and the slide files in pathology department.
Most patients had follow-up records for 5 years in both Medical Oncology and Clinical oncology & nuclear medicine departments, Faculty of Medicine, Zagazig University. We followed all the fifty patients till death or till the most recent follow-up contact with them and we have finished data collection in December 2016.
Immunohistochemical staining
We performed the immunohistochemical staining by using streptavidin-peroxidase method [24], where we incubated our slides with primary rabbit monoclonal anti- Sirt1 antibody (ab32441) (Abcam, Cambridge, MA, USA) at 1:150 dilution, anti-CD163 antibody (ab87099) (Abcam, Cambridge, MA, USA) at 1/500 dilution and primary mouse monoclonal anti-mast cell tryptase antibody (ab81703) at 1:2000 dilution. We used colon carcinoma, spleen and tonsil sections as positive controls for Sirt1, CD163 and tryptase respectively and we have replaced the primary antibodies by the non-immune serum for the negative controls.
Evaluation of immunohistochemical expression of Sirt1
We evaluated the staining extension & intensity independently in 5 random fields in our entire examined slides [25]. We graded the stain intensity as follows: negative, 1+, 2+, and 3+. Then we evaluated the extent of positively stained cells as follows: zero (0-1%); one (1–25%); two (25–50%); three (50–75%); and four (>75%). To reach the final immunopositivety score we multiplied both the intensity & extent of the stain that has given values from zero to twelve. We used a cutoff of 4 above which was considered as high Sirt1 expression and below which was low expression. Sirt1 expression either as high or low, was it based on the expression by both the malignant tumor cells (RS) and the small reactive lymphocytes.
Evaluation of immunohistochemical expression of CD163 and Tryptase
For the CD163 staining, we counted 3 variable areas of the tumor in high power field x 400 magnifications and we scored the percentage of CD163 positivity in the cell membrane and cytoplasm of macrophages [26]. We have counted mast cells positive for tryptase in ten randomly selected high power fields x 200 magnifications [6]. We used a cutoff of twenty five percent for CD163 positive macrophage and tryptase positive mast cells above which was considered high expression and below which was low expression of both markers and that was applied for all patients.
Statistical Analysis
The statistics were evaluated by using SPSS 22.0 for windows (SPSS Inc., Chicago, IL, USA) and MedCalc windows (MedCalc Software bvba 13, Ostend, Belgium). Continuous variables that were expressed as the mean ± SD & median (range), were checked for normality by using Shapiro-Wilk test. Mann Whitney U test was used to compare between two groups of non-normally distributed variables. Kruskal Wallis H test was used to compare between more than two groups of normally distributed variables. Stratification of OS and RFS was done according to all clinicopathological features and immunohistochemical markers. These time-to-death distributions were estimated using the method of Kaplan- Meier plot, and compared using two-sided exact log-rank test. All tests were two sided. A p-value <0.05 was considered significant.
Results
Patient data
The clinical data of our patients with cHL that were included in the current study were summarized (Table 1). We included 28 (56%) males and 22 (44%) females with age ranged from (20-80) years (Mean: 47.5 ± 20.7 years), 31 (62%) cases were having mixed cellularity subtype, 14 (28%) cases were of nodular sclerosis subtype and 5 (10%) cases were of lymphocyte rich subtype.
| Characteristics | All (N=50) |
Sirt1 | p-value | CD163 | p-value | Tryptase | p-value | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Low (N=21) |
High (N=29) |
Low (N=27) |
High (N=23) |
Low (N=28) |
High (N=22) |
||||||||||||||
| Age (years) | |||||||||||||||||||
| Mean ± SD | 47.5 ± 20.7 | 35.9 ± 17.6 | 55.8 ± 18.8 | 0.001 | 37.7 ± 17.8 | 59 ± 18 | <0.001 | 38.6 ± 18.2 | 58.7 ± 18.4 | 0.001 | |||||||||
| Median (Range) | 50 (20-80) | 29 (20-72) | 60 (20-80) | 30 (20-70) | 64 (20-80) | 31 (20-70) | 64 (20-80) | ||||||||||||
| ≤ 45 years | 24 (48%) | 16 (66.7%) | 8 (33.3%) | 0.001§ | 19 (79.2%) | 5 (20.8%) | 0.001§ | 19 (79.2%) | 5 (20%) | 0.002§ | |||||||||
| >45 years | 26 (52%) | 5 (19.2%) | 21 (80.8%) | 8 (30.8%) | 18 (69.2%) | 9 (34.6%) | 17 (65.4%) | ||||||||||||
| Sex | |||||||||||||||||||
| Male | 28 (56%) | 9 (32.1%) | 19 (67.9%) | 0.111§ | 13 (46.4%) | 15 (53.6%) | 0.226§ | 14 (50%) | 14 (50%) | 0.335§ | |||||||||
| Female | 22 (44%) | 12 (54.5%) | 10 (45.5%) | 14 (63.6%) | 8 (36.4%) | 14 (63.6%) | 8 (36.4%) | ||||||||||||
| B symptoms | |||||||||||||||||||
| Absent | 35 (70%) | 21 (60%) | 14 (40%) | <0.001§ | 27 (77.1%) | 8 (22.9%) | <0.001§ | 28 (80%) | 7 (20%) | <0.001§ | |||||||||
| Present | 15 (30%) | 0 (0%) | 15 (100%) | 0 (0%) | 15 (100%) | 0 (0%) | 15 (100%) | ||||||||||||
| Bulky Med. LN | |||||||||||||||||||
| Absent | 37 (74%) | 20 (58.8%) | 17 (45.9%) | 0.004§ | 26 (70.3%) | 11 (29.7%) | <0.001§ | 27 (73%) | 10 (27%) | <0.001§ | |||||||||
| Present | 13 (26%) | 1 (6.3%) | 12 (92.3%) | 1 (7.7%) | 12 (92.3%) | 1 (7.7%) | 12 (92.3%) | ||||||||||||
| Number of nodal sites | |||||||||||||||||||
| One site | 7 (14%) | 7 (100%) | 0 (0%) | <0.001‡ | 7 (100%) | 0 (0%) | <0.001‡ | 7 (100%) | 0 (0%) | <0.001‡ | |||||||||
| Two sites | 10 (20%) | 10 (100%) | 0 (0%) | 10 (100%) | 0 (0%) | 10 (100%) | 0 (0%) | ||||||||||||
| Three sites | 3 (6%) | 0 (0%) | 3 (100%) | 3 (100%) | 0 (0%) | 3 (100%) | 0 (0%) | ||||||||||||
| Four sites | 6 (12%) | 1 (16.7%) | 5 (83.3%) | 3 (50%) | 3 (50%) | 3 (50%) | 3 (50%) | ||||||||||||
| Five sites | 6 (12%) | 1 (16.7%) | 5 (83.3%) | 3 (50%) | 3 (50%) | 3 (50%) | 3 (50%) | ||||||||||||
| >Five sites | 18 (36%) | 2 (11.1%) | 16 (88.9%) | 1 (5.6%) | 17 (94.4%) | 2 (11.1%) | 16 (88.9%) | ||||||||||||
| Spleen involvement | |||||||||||||||||||
| Absent | 28 (56%) | 21 (75%) | 7 (25%) | <0.001§ | 23 (82.1%) | 5 (17.9%) | <0.001§ | 23 (82.1%) | 5 (17.9%) | <0.001§ | |||||||||
| Present | 22 (44%) | 0 (0%) | 22 (100%) | 4 (18.2%) | 18 (81.8%) | 5 (22.7%) | 17 (77.3%) | ||||||||||||
| BM infiltration | |||||||||||||||||||
| Absent | 36 (72%) | 20 (55.6%) | 16 (44.4%) | 0.002§ | 26 (72.2%) | 10 (27.8%) | <0.001§ | 27 (75%) | 9 (25%) | <0.001§ | |||||||||
| Present | 14 (28%) | 1 (7.1%) | 13 (92.9%) | 1 (7.1%) | 13 (92.9%) | 1 (7.1%) | 13 (92.9%) | ||||||||||||
| Extranodal lesions | |||||||||||||||||||
| Absent | 14 (28%) | 12 (85.7%) | 2 (14.3%) | <0.001§ | 13 (92.9%) | 1 (7.1%) | 0.001§ | 13 (92.9%) | 1 (7.1%) | 0.001§ | |||||||||
| Present | 36 (72%) | 9 (25%) | 27 (75%) | 14 (38.9%) | 22 (61.1%) | 15 (41.7%) | 21 (58.3%) | ||||||||||||
| Histopathological subtype | |||||||||||||||||||
| NS | 14 (28%) | 12 (85.7%) | 2 (14.3%) | <0.001§ | 14 (100%) | 0 (0%) | <0.001§ | 14 (100%) | 0 (0%) | <0.001§ | |||||||||
| MC | 31 (62%) | 8 (28.5%) | 23 (74.2%) | 10 (32.3%) | 21 (67.7%) | 11 (35.5%) | 20 (64.5%) | ||||||||||||
| LR | 5 (10%) | 1 (20%) | 4 (80%) | 3 (60%) | 2 (40%) | 3 (60%) | 2 (40%) | ||||||||||||
| Stage | |||||||||||||||||||
| Stage I | 7 (14%) | 7 (100%) | 0 (0%) | <0.001‡ | 7 (100%) | 0 (0%) | <0.001‡ | 7 (100%) | 0 (0%) | <0.001‡ | |||||||||
| Stage II | 13 (26%) | 10 (76.9%) | 3 (23.1%) | 13 (100%) | 0 (0%) | 13 (100%) | 0 (0%) | ||||||||||||
| Stage III | 12 (24%) | 2 (16.7%) | 10 (83.3%) | 6 (50%) | 6 (50%) | 6 (50%) | 6 (50%) | ||||||||||||
| Stage IV | 18 (36%) | 2 (11.1%) | 16 (88.9%) | 1 (5.6%) | 17 (94.4%) | 2 (11.1%) | 16 (88.9%) | ||||||||||||
| Prognostic group | |||||||||||||||||||
| Unfavorable | 30 (60%) | 4 (13.3%) | 26 (86.7%) | <0.001§ | 7 (23.3%) | 23 (76.7%) | <0.001 | 8 (26.7%) | 22 (73.3%) | <0.001§ | |||||||||
| Favorable | 20 (40%) | 17 (85%) | 3 (15%) | 20 (100%) | 0 (0%) | 20 (100%) | 0 (0%) | ||||||||||||
| Risk group | |||||||||||||||||||
| Low favorable | 16 (32%) | 16 (100%) | 0 (0%) | <0.001‡ | 16 (100%) | 0 (0%) | <0.001‡ | 16 (100%) | 0 (0%) | <0.001‡ | |||||||||
| Low unfavorable | 3 (6%) | 0 (0%) | 3 (100%) | 3 (100%) | 0 (0%) | 3 (100%) | 0 (0%) | ||||||||||||
| Intermediate | 18 (38%) | 5 (27.8%) | 13 (72.2%) | 8 (44.4%) | 10 (55.6%) | 9 (50%) | 9 (50%) | ||||||||||||
| High | 13 (26%) | 0 (0%) | 13 (100%) | 0 (0%) | 13 (100%) | 0 (0%) | 13 (100%) | ||||||||||||
| Categorical variables were expressed as number(percentage); continuous variables were expressed as mean ± SD & median (range) Ÿ Mann Whitney U test; §Chi-square test; ‡Chi-square test for trend; p<0.05 is significant. |
|||||||||||||||||||
Table 1: Correlation between clinicopathological criteria, Sirt1, CD163 & Tryptase expression in our patients.
Sirt1 expression and its correlation with clinicopathological criteria of our patients
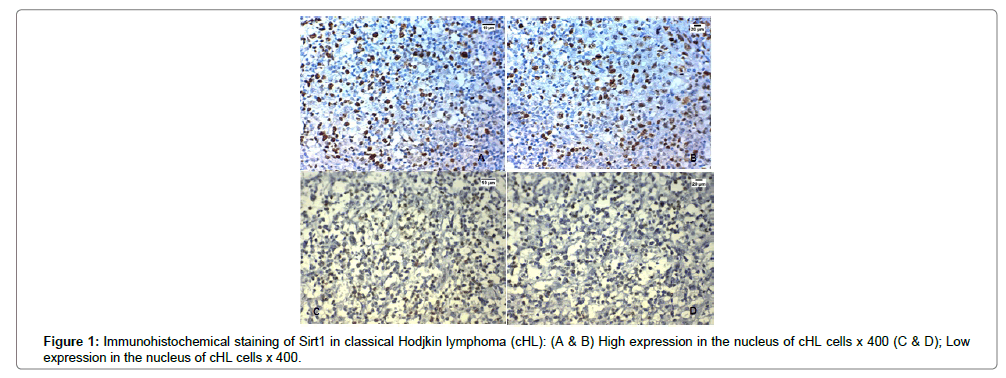
Sirt1 was overexpressed in the nuclei of both malignant cells and the background of tumor infiltrating lymphocytes in 29 cases out of 50 (58%) cases of cHL, and in the remaining 42% of cases there were no lymphocytes expressing Sirt1 at all. The expression of Sirt1 in was significantly positively correlated with advanced stage, presence of B symptoms, splenic affection, presence of extranodal spread, low serum albumin, low hemoglobin (p<0.001), presence of bone marrow infiltration (p=0.002) and the presence of bulky mediastinal lymph node involvement (p=0.004) (Table 1 and Figure 1A-D).
correlation clinicopathological features of our patients
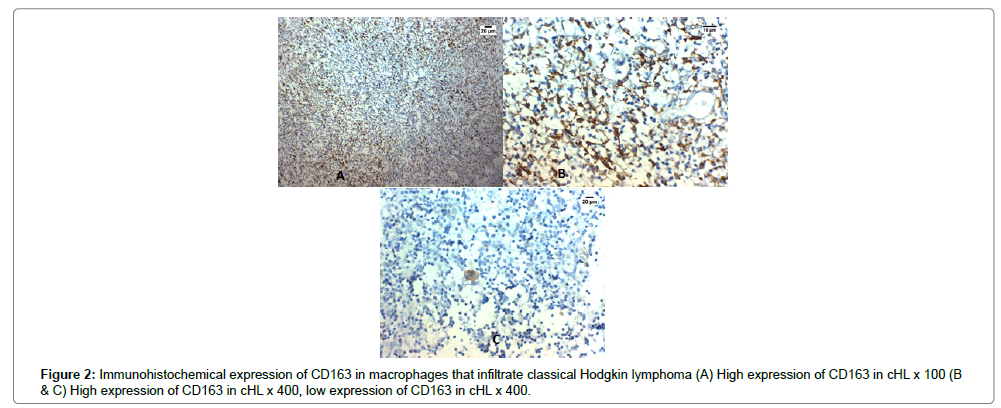
High expression of CD163 in tumor infiltrating macrophages was detected in in 23 out of 50 (46%) cases of cHL.
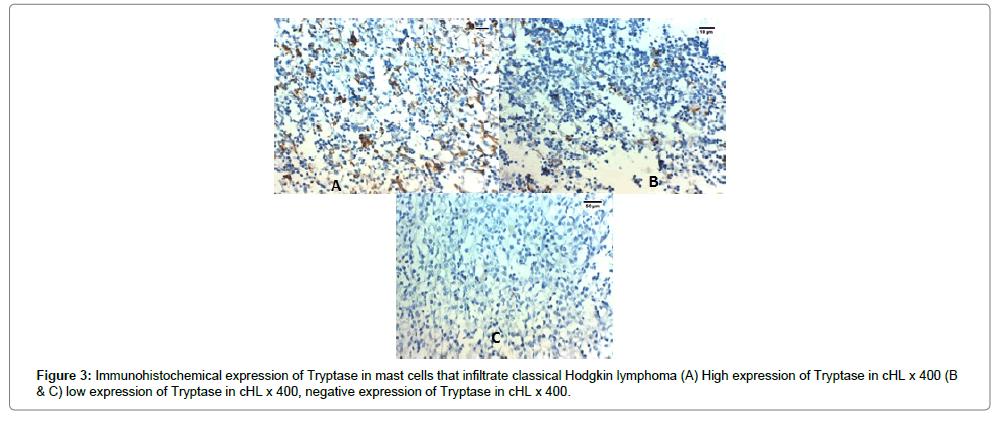
High expression of tryptase in tumor infiltrating mast cells was detected in 22 out of 50 (44%) cases of cHL. The expression of CD163 and tryptase in cHL was significantly positively correlated with advanced stage, presence of B symptoms, splenic involvement, presence of extra-nodal spread, low albumin, low hemoglobin, bone marrow infiltration and the presence of bulky mediastinal lymph node (p<0.001) (Table 1 and Figures 2A-C, 3A-C).
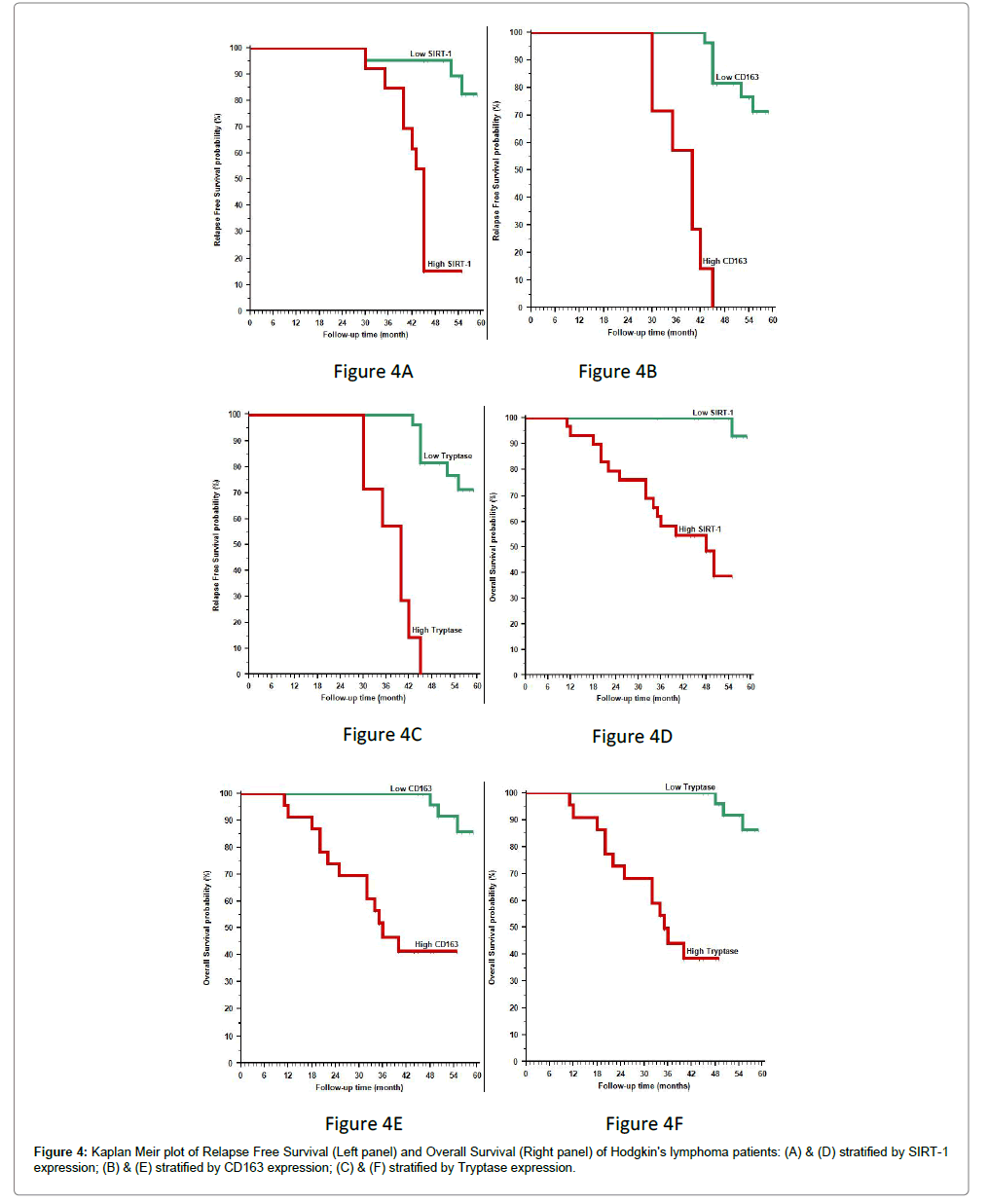
• The 3-year recurrence free survival rate of our patients was 91.2% for all cases, 95.2% and 84.6% in patients with low and high Sirt1 expression respectively, 100% and 57.1% in patients with low and high tryptase and CD163 expression respectively (p<0.001).
• The 3-year overall survival (OS) rate of our patients was 75.8% for all cases, 58.2% in patients with high Sirt1 expression (p=0.008), 46.6% in patients with high CD163 expression and 44.1% in patients with high tryptase expression (p<0.001).
High Sirt1, tryptase and CD163 expression was significantly positively correlated with poor response to therapy and high incidence of relapse after treatment and poor 3-year overall survival rate (OS) (p<0.001) (Tables 2-4 and Figure 4A-F).
| Characteristics | All (N=50) |
Response | p-value | Response | p-value | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PD (N=6) |
SD (N=6) |
PR (N=4) |
CR (N=34) |
NR (N=24) |
OAR (N=38) |
||||||||||||
| ESR | |||||||||||||||||
| <50 mm/h | 34 (68%) | 0 (0%) | 0 (0%) | 2 (5.9%) | 32 (94.1%) | <0.001§ | 0 (0%) | 34 (100%) | <0.001§ | ||||||||
| >50 mm/h | 16 (32%) | 6 (37.5%) | 6 (37.5%) | 2 (12.5%) | 2 (12.5%) | 12 (75%) | 4 (25%) | ||||||||||
| Albumin | |||||||||||||||||
| <4 g/dl | 30 (60%) | 6 (20%) | 6 (20%) | 4 (13.3%) | 14 (46.7%) | 0.001§ | 12 (40%) | 18 (60%) | 0.001§ | ||||||||
| >4 g/dl | 20 (40%) | 0 (0%) | 0 (0%) | 0 (0%) | 20 (100%) | 0 (0%) | 20 (100%) | ||||||||||
| Hemoglobin | |||||||||||||||||
| <10 g/dl | 30 (60%) | 6 (20%) | 6 (20%) | 4 (13.3%) | 14 (46.7%) | 0.001§ | 12 (40%) | 18 (60%) | 0.001§ | ||||||||
| >10 g/dl | 20 (40%) | 0 (0%) | 0 (0%) | 0 (0%) | 20 (100%) | 0 (0%) | 20 (100%) | ||||||||||
| WBCs | |||||||||||||||||
| <15x103/mm3 | 30 (60%) | 6 (20%) | 6 (20%) | 4 (13.3%) | 14 (46.7%) | 0.001§ | 12 (40%) | 18 (60%) | 0.001§ | ||||||||
| >15x103/mm3 | 20 (40%) | 0 (0%) | 0 (0%) | 0 (0%) | 20 (100%) | 0 (0%) | 20 (100%) | ||||||||||
| Stage | |||||||||||||||||
| Stage I | 7 (14%) | 0 (0%) | 0 (0%) | 0 (0%) | 7 (100%) | <0.001‡ | 0 (0%) | 7 (100%) | 0.002‡ | ||||||||
| Stage II | 13 (26%) | 0 (0%) | 0 (0%) | 0 (0%) | 13 (100%) | 0 (0%) | 13 (100%) | ||||||||||
| Stage III | 12 (24%) | 1 (8.3%) | 3 (25%) | 1 (8.3%) | 7 (58.3%) | 4 (33.3%) | 8 (66.7%) | ||||||||||
| Stage IV | 18 (36%) | 5 (27.8%) | 3 (16.7%) | 3 (16.7%) | 7 (39.9%) | 8 (44.4%) | 10 (55.6%) | ||||||||||
| Prognostic group | |||||||||||||||||
| Unfavorable | 30 (60%) | 6 (20%) | 6 (20%) | 4 (13.3%) | 14 (46.7%) | 0.001§ | 12 (40%) | 18 (60%) | 0.001§ | ||||||||
| Favorable | 20 (40%) | 0 (0%) | 0 (0%) | 0 (0%) | 20 (100%) | 0 (0%) | 20 (100%) | ||||||||||
| Risk group | |||||||||||||||||
| Low favorable | 16 (32%) | 0 (0%) | 0 (0%) | 0 (0%) | 16 (100%) | <0.001‡ | 0 (0%) | 16 (100%) | <0.001‡ | ||||||||
| Low unfavorable | 3 (6%) | 0 (0%) | 0 (0%) | 0 (0%) | 3 (100%) | 0 (0%) | 3 (100%) | ||||||||||
| Intermediate | 18 (38%) | 0 (0%) | 0 (0%) | 3 (16.7%) | 15 (83.3%) | 0 (0%) | 18 (100%) | ||||||||||
| High | 13 (26%) | 6 (46.2%) | 6 (46.2%) | 1 (7.7%) | 0 (0%) | 12 (92.3%) | 1 (7.7%) | ||||||||||
| Sirt1 | |||||||||||||||||
| Low | 21 (42%) | 0 (0%) | 0 (0%) | 0 (0%) | 21 (100%) | 0.001§ | 0 (0%) | 21 (100%) | 0.001§ | ||||||||
| High | 29 (58%) | 6 (20.7%) | 6 (20.7%) | 4 (13.8%) | 13 (44.8%) | 12 (41.4%) | 17 (58.6%) | ||||||||||
| CD163 | |||||||||||||||||
| Low | 27 (54%) | 0 (0%) | 0 (0%) | 0 (0%) | 27 (100%) | <0.001§ | 0 (0%) | 27 (100%) | <0.001 | ||||||||
| High | 23 (46%) | 6 (20.7%) | 6 (20.7%) | 4 (13.8%) | 7 (30.4%) | 12 (52.2%) | 11 (47.8%) | ||||||||||
| Tryptase | |||||||||||||||||
| Low | 28 (56%) | 0 (0%) | 0 (0%) | 1 (3.6%) | 27 (96.4%) | <0.001§ | 0 (0%) | 28 (100%) | <0.001§ | ||||||||
| High | 22 (44%) | 6 (27.3%) | 6 (27.3%) | 3 (13.6%) | 7 (31.8%) | 12 (54.5%) | 10 (45.5%) | ||||||||||
| Categorical variables were expressed as number(percentage); continuous variables were expressed as mean ± SD & median (range). Ÿ Mann Whitney U test; Ø Kraskall Wallis test for more than two groups; §Chi-square test; ‡Chi-square test for trend; p<0.05 is significant. |
|||||||||||||||||
Table 2: Correlation between clinicopathological criteria, Sirt1, CD163, Tryptase expression and response to treatment in our patients.
| Characteristics | All (N=34) |
Relapse | p-value | Median RFS | Relapse Free Survival (RFS) | HR (95%CI) |
p-value | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No (N=20) |
No (N=14) |
1 year RFS (%) |
3 year RFS (%) |
5 year RFS (%) |
||||||||||||||||
| All patients | NR | 100% | 91.2% | 56.5% | ||||||||||||||||
| Initial site | ||||||||||||||||||||
| Cervical | 14 (41.2%) | 9 (64.3%) | 5 (35.7%) | 0.474§ | NR | 100% | 85.7% | 62.9% | ---- | 0.242† | ||||||||||
| SC | 3 (8.8%) | 1 (33.3%) | 2 (66.7%) | 45 month | 100% | 100% | 33.3% | |||||||||||||
| Axilla | 8 (23.5%) | 4 (50%) | 4 (50%) | NR | 100% | 100% | 50% | |||||||||||||
| Mediastinum | 8 (23.5%) | 6 (75%) | 2 (25%) | NR | 100% | 100% | 75% | |||||||||||||
| Inguinal | 1 (2.9%) | 0 (0%) | 1 (100%) | 42 month | 100% | 100% | 0% | |||||||||||||
| Bulky LN | ||||||||||||||||||||
| Absent | 23 (67.6%) | 18 (78.3%) | 5 (21.7%) | 0.002§ | NR | 100% | 100% | 75.3% | 6.931 | <0.001† | ||||||||||
| Present | 11 (32.4%) | 2 (18.2%) | 9 (81.8%) | 42 month | 100% | 72.7% | 18.2% | (1.898-25.304) | ||||||||||||
| Bulky Med. LN | ||||||||||||||||||||
| Absent | 32 (94.1%) | 19 (59.4%) | 13 (40.6%) | 1.000§ | NR | 100% | 90.6% | 56.8% | 1.399 | 0.737† | ||||||||||
| Present | 2 (5.9%) | 1 (50%) | 1 (50%) | NR | 100% | 100% | 50% | (0.132-14.785) | ||||||||||||
| Number of nodal sites | ||||||||||||||||||||
| One site | 7 (20.6%) | 7 (100%) | 0 (0%) | 0.001‡ | NR | 100% | 100% | 100% | ---- | <0.001† | ||||||||||
| Two sites | 10 (29.4%) | 9 (90%) | 1 (10%) | NR | 100% | 100% | 85.7% | |||||||||||||
| Three sites | 3 (8.8%) | 0 (0%) | 3 (100%) | 45 month | 100% | 100% | 0% | |||||||||||||
| Four sites | 4 (11.8%) | 2 (50%) | 2 (50%) | 52 month | 100% | 100% | 37.5% | |||||||||||||
| Five sites | 3 (8.8%) | 1 (33.3%) | 2 (66.7%) | 45 month | 100% | 100% | 33.3% | |||||||||||||
| >Five sites | 7 (20.6%) | 1 (14.3%) | 6 (85.7%) | 40 month | 100% | 57.1% | 14.3% | |||||||||||||
| BM infiltration | ||||||||||||||||||||
| Absent | 33 (38.2%) | 19 (57.6%) | 14 (42.4%) | 1.000§ | NR | 100% | 90.9% | 55.1% | 0.000 | 0.438† | ||||||||||
| Present | 1 (2.9%) | 1 (100%) | 0 (0%) | NR | 100% | 100% | 100% | |||||||||||||
| Extranodal lesions | ||||||||||||||||||||
| Absent | 13 (38.2%) | 12 (92.3%) | 1 (7.7%) | 0.002§ | NR | 100% | 100% | 87.5% | 11.043 | 0.002† | ||||||||||
| Present | 21 (61.8%) | 8 (38.1%) | 13 (61.9%) | 45 month | 100% | 85.7% | 37.5% | (3.860-31.595) | ||||||||||||
| Histopathological subtype | ||||||||||||||||||||
| NS | 14 (41.2%) | 12 (85.7%) | 2 (14.3%) | 0.005§ | NR | 100% | 100% | 86.7% | ---- | 0.028† | ||||||||||
| MC | 16 (47.1%) | 8 (50%) | 8 (50%) | 52 month | 100% | 81.2% | 48.2% | |||||||||||||
| LR | 4 (11.8%) | 0 (0%) | 4 (100%) | 45 month | 100% | 100% | 0% | |||||||||||||
| Stage | ||||||||||||||||||||
| Stage I | 7 (20.6%) | 7 (100%) | 0 (0%) | 0.001‡ | NR | 100% | 100% | 100% | ---- | <0.001† | ||||||||||
| Stage II | 13 (38.2) | 9 (69.2%) | 4 (30.8%) | NR | 100% | 100% | 65.9% | |||||||||||||
| Stage III | 7 (20.6%) | 3 (42.9%) | 4 (57.1%) | 52 month | 100% | 100% | 38.1% | |||||||||||||
| Stage IV | 7 (20.6%) | 1 (14.3%) | 6 (85.7%) | 40 month | 100% | 57.1% | 14.3% | |||||||||||||
| Prognostic group | ||||||||||||||||||||
| Unfavorable | 14 (41.2%) | 4 (28.6%) | 10 (71.4%) | 0.003§ | 45 month | 100% | 78.6% | 26.8% | 0.181 | <0.001† | ||||||||||
| Favorable | 20 (58.8%) | 16 (80%) | 4 (20%) | NR | 100% | 100% | 77.9% | (0.058-0.561) | ||||||||||||
| Sirt1 | ||||||||||||||||||||
| Low | 21 (61.8%) | 18 (85.7%) | 3 (14.3%) | <0.001§ | NR | 100% | 95.2% | 82.7% | 9.590 | <0.001† | ||||||||||
| High | 13 (38.2%) | 2 (15.4%) | 11 (84.6%) | 45 month | 100% | 84.6% | ----- | (2.973-30.935) | ||||||||||||
| CD163 | ||||||||||||||||||||
| Low | 27 (79.4%) | 20 (74.1%) | 7 (25.9%) | 0.001§ | NR | 100% | 100% | 71.2% | 11.545 | <0.001† | ||||||||||
| High | 7 (20.6%) | 0 (0%) | 7 (100%) | 40 month | 100% | 57.1% | ----- | (1.669-79.866) | ||||||||||||
| Tryptase | ||||||||||||||||||||
| Low | 27 (79.4%) | 20 (74.1%) | 7 (25.9%) | 0.001§ | NR | 100% | 100% | 71.2% | 11.545 | <0.001† | ||||||||||
| High | 7 (20.5%) | 0 (0%) | 7 (100%) | 40 month | 100% | 57.1% | ----- | (1.669-79.866) | ||||||||||||
| Categorical variables were expressed as number(percentage); continuous variables were expressed as mean±SD & median (range). Ÿ Mann Whitney U test; §Chi-square test; ‡Chi-square test for trend; † Log rank test; NR denote not reached yet; HR Hazards Ratio; 95%CI: 95% confidence interval; p<0.05 is significant. |
||||||||||||||||||||
Table 3: Correlation between clinicopathological criteria, Sirt1, CD163, Tryptase expression and relapse of our patients.
| Characteristics | All (N=50) |
Death | p-value | Median OS | Overall Survival (OS) | HR (95%CI) |
p-value | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No (N=34) |
Yes (N=16) |
1 year OS (%) | 3 year OS (%) | 5 year OS (%) | ||||||||||||||
| All patients | NR | 96% | 75.8% | 63.9% | ||||||||||||||
| Spleen involvement | ||||||||||||||||||
| Absent | 28 (56%) | 24 (85.7%) | 4 (14.3%) | 0.002§ | NR | 100% | 96.4% | 80.8% | 6.091 | <0.001† | ||||||||
| Present | 22 (44%) | 10 (45.5%) | 12 (54.5%) | 36 month | 90.9% | 49.6% | 44.6% | (2.148-17.270) | ||||||||||
| Histopathological subtype | ||||||||||||||||||
| NS | 14 (28%) | 11 (78.6%) | 3 (21.4%) | 0.429§ | NR | 100% | 100% | 74% | ----- | 0.216† | ||||||||
| MC | 31 (62%) | 19 (61.3%) | 12 (38.7%) | NR | 93.5% | 64% | 60.5% | |||||||||||
| LR | 5 (10%) | 4 (80%) | 1 (20%) | NR | 100% | 80% | 80% | |||||||||||
| Stage | ||||||||||||||||||
| Stage I | 7 (14%) | 6 (85.7%) | 1 (14.3%) | 0.054‡ | NR | 100% | 100% | 80% | ----- | 0.056† | ||||||||
| Stage II | 13 (26%) | 11 (84.6%) | 2 (15.4%) | NR | 100% | 100% | 80.8% | |||||||||||
| Stage III | 12 (24%) | 7 (58.3%) | 5 (41.7%) | NR | 100% | 66.7% | 58.3% | |||||||||||
| Stage IV | 18 (36%) | 10 (55.6%) | 8 (44.4%) | NR | 88.9% | 55.6% | 55.6% | |||||||||||
| Prognostic group | ||||||||||||||||||
| Unfavorable | 30 (60%) | 17 (56.7%) | 13 (43.3%) | 0.035§ | NR | 93.3% | 59.4% | 55.7% | 0.226 | 0.009† | ||||||||
| Favorable | 20 (40%) | 17 (85%) | 3 (15%) | NR | 100% | 100% | 81.2% | (0.085-0.604) | ||||||||||
| Risk group | ||||||||||||||||||
| Low favorable | 16 (32%) | 15 (93.8%) | 1 (6.3%) | <0.001‡ | NR | 100% | 100% | 90.9% | ----- | <0.001† | ||||||||
| Low unfavorable | 3 (6%) | 1 (33.3%) | 2 (66.7%) | 50 month | 100% | 100% | 0% | |||||||||||
| Intermediate | 18 (38%) | 18 (100%) | 0 (0%) | NR | 100% | 100% | 100% | |||||||||||
| High | 13 (26%) | 0 (0%) | 13 (100%) | 25 month | 84.6% | 7.6% | 0% | |||||||||||
| Sirt1 | ||||||||||||||||||
| Low | 21 (42%) | 20 (95.2%) | 1 (4.8%) | <0.001§ | NR | 100% | 100% | 92.9% | 17.085 | <0.001† | ||||||||
| High | 29 (58%) | 14 (48.3%) | 15 (51.7%) | 48 month | 93.1% | 58.2% | ----- | (6.399-45.617) | ||||||||||
| CD163 | ||||||||||||||||||
| Low | 27 (54%) | 24 (88.9%) | 3 (11.1%) | 0.001§ | NR | 100% | 100% | 85.8% | 9.232 | <0.001† | ||||||||
| High | 23 (46%) | 10 (43.5%) | 13 (56.5%) | 36 month | 91.3% | 46.6% | ----- | (3.227-26.407) | ||||||||||
| Tryptase | ||||||||||||||||||
| Low | 28 (56%) | 25 (89.3%) | 3 (10.7%) | <0.001§ | NR | 100% | 100% | 86.4% | 10.508 | <0.001† | ||||||||
| High | 22 (44%) | 9 (40.9%) | 13 (59.1%) | 36 month | 90.9% | 44.1% | ----- | (3.576-30.873) | ||||||||||
| Categorical variables were expressed as number(percentage); continuous variables were expressed as mean ±SD& median (range). Ÿ Mann Whitney U test; §Chi-square test; ‡Chi-square test for trend; † Log rank test; NR denote not reached yet; HR Hazards Ratio; 95%CI: 95% confidence interval; p<0.05 is significant. |
||||||||||||||||||
Table 4: Correlation between clinicopathological criteria, Sirt1, CD163, Tryptase expression and survival of our patients.
Discussion
In our study we found that Sirt1 was overexpressed in the nuclei of both Reed-Sternberg (RS) cells and the background of tumor infiltrating lymphocytes in 58% cases of cHL and in the remaining 42% of cases there were no lymphocytes expressing Sirt1 at all. Sirt1 expression was associated with poor clinic-pathological parameters and poor prognosis, and our results were the same like Quesada et al., and Frazzi et al. [27,28]. Our results were explained that one of the Sirt1 de-acetylation targets is FoxP3 that is a transcription factor which is essential for T regulatory lymphocytes (Tregs) differentiation and function. So, Sirt1 de-acetylation of FoxP3 could result in FoxP3 destabilization and increased its degradation that decreased Tregs activity, those tregs stimulate the immunity against cancer and had onco-suppressive role, so, inhibition of Sirt1 increases the suppressive action of Tregs cells [29,30]. Moreover, Sirt1 inhibition in cHL allows RS cells to promote CD4+ naïve T cells differentiation toward Tregs as RS could promote differentiation of CD4+ naïve T cells toward both Tregs and cytotoxic T cells that are both increase their onco-suppressive role [31]. All these results are in line with ours that Sirt-1 over expression in cHL is a marker of poor prognosis and its inhibition was a novel therapeutic target to improve patient outcome and also histone deacetylase inhibitors (such as vorinostat [SAHA]) could increase the efficacy of the present therapeutic agents by down regulating Sirt1 and decreasing Sirt1 deacetylase activity [32,33], which could decrease the inhibitory effects on Tregs and restore the immune balance in cHL microenvironment. We found that cHL patients with Sirt1 over expression had higher incidence of recurrence after successful therapy which suggest the possibility that immune dys-regulation played an essential role in the cHL pathogenesis and could decrease Tregs resulting in imbalance between them and cytotoxic T-cells. This was in agreeing with immune dysregulation theory that played a role in cHL pathogenesis. Proofs of theory of the role of inhibition of Sirt1 pathway and Tregs stimulation to restore immune balance in cHL are recently proved [34,35]. In our study we demonstrate that, in patients with cHL, the recurrence of the disease after successful therapy can be correlated with Sirt-l expression so it carries prognostic significances, so Histone deacetylase inhibitors can increase the efficacy of pre-existing drugs by inhibiting Sirt1 gene mRNA/Sirt1 protein function, which subsequently improve prognosis and decrese remission in cHL [29,30,32-34]. Targeting Sirt1 in the RS cells also may facilitate their ability to promote naïve T cell differentiation toward Treg cells over CTL [31]. Further examination of this pathway and its relationship to T cell microenvironment and immune dysregulation in cHL is warranted, as it carries therapeutic implications for improved patient out-comes. Another explanation of how Sirt-1 overexpression increasing cHL progression is that it could deacetylate p53 causing its inactivation so decreasing growth arrest or apoptosis of malignant cells and hence increasing oncogenic functions [36,37]. Similar to our results in cHL Sirt1 over expression is associated with poor prognosis in malignancies of many organs as stomach and liver [38,39]. Recently, many studies had focused on the role of the microenvironment of cHL in prediction of treatment outcome. In our study, we evaluated the presence of 2 types of benign reactive inflammatory cells in the microenvironment of cHL that are the macrophages and mast cells detecting their presence and number by CD163 & Tryptase expression respectively then we correlating their number with clinicopathological and prognostic status. Patients with high counts of mast cells evidenced by tryptase over expression and macrophages evidenced by CD163 overexpression were found to have more advanced stages (stage III–IV, vs. I–II), poor clinicopathological criteria, high incidence of disease relapse, poor response to therapy and poor survival. Survival analyses were performed reveal significant differences between the groups. Our results are like Englund et al., who proved similar results to ours but in pediatric HL [40], also the study by Kamper et al. that had supported the value of evaluating tumor-infiltrating macrophages in pretreatment samples for prediction of patient outcome in cHL [41]. Our study confirms results of previous studies about the prognostic role of tumor-infiltrating macrophages in cHL and we used the macrophage marker CD163 that was found to be more accurate than CD68. Klein et al., Koh et al., Tudor et al., found similar results to us in cHL [42-44]. On the contrary from us Kayal et al., results showed no significant difference between macrophages infiltration, clinicopathological parameters or cHL prognosis [45].
Similar to our results Barros et al. proved that progression-free survival rate (PFS) was lower in cases with high numbers of CD163+ macrophages [46]. But, Gupta et al. presented that there was no association between survival rates in macrophages evaluated with either CD163 or CD68 [47]. These conflicting data could be due to different number of patients used, variable follow up period, different clones of antibodies used or different method of evaluation.
There are many questions regarding the exact mechanisms of how RS cells can attract the macrophages, how macrophages affect RS cell growth and how RS cells could accquire immune isolation by such interaction. Recently, there are many discovered molecules that had been incriminated in macrophage chemotaxis and signaling in cHL pathobiology, e.g. CSF1R, MIF, fractalkine and CD74 [48], some of these molecules could be promising targets for discovering novel drug therapies that target the macrophages directly or by causing disturbances in the signaling pathways between RS cells and the surrounding tumor microenvironment.
We found that high number of mast cell infiltration in cHL as evidenced by tryptase overexpression was correlated to advanced stage and the presence of B-symptoms that was similar to Molin et al. who found that high mast cell infiltration had found to be related to advanced disease and poor relapse-free survival in adults [49]. Andersen et al. detect the relation between high mast cell count and poor outcome of patients similar to our results but that was only restricted to the mixed cellularity subtype [20]. In our results higher incidence of disease remission after successive therapy was higher in patients with higher mast cells count that was explained by RS cells stimulation by mast cells through CD30-CD30L interaction [49].
Conclusion
We evaluated the clinicopathological and prognostic roles of the microenvironment in cHL and found a significant relations between Sirt1 expression which was related to Tregs differentiation and oncosuppressive role, macrophages as evidenced by CD163 expression, mast cells as evidenced by tryptase expression, poor clinicopathological parameters, advanced stage and poor patient prognosis, which may help to detect novel therapeutic targets to improve cHL outcome mainly in advanced and relapsed patients. Because of the conflicting results between our study and different previous studies we recommend that a large scale study on large number of patients with different subtypes to prove our results and detect the possible use of targeted therapies against the inflammatory infiltrate in the cellular microenvironment of HL.
References
- Eichenauer DA, Engert A, André M, Federico M, Illidge T, et al. (2014) Hodgkin's lymphoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-updagger. Ann Oncol 3: 70-75.
- Evens AM, Hutchings M, Diehl V (2007) Treatment of Hodgkin lymphoma: the past, present, and future. Nat Clin Pract Oncol 5: 543-556
- Quddus F, Armitage JO (2009) Salvage therapy for Hodgkin’s lymphoma. Oncologist 15: 161-163.
- Mirsadraee S, Farzadnia M, Heidary F, Rahimi M, Jabbari NH, et al. (2011) A comparative immumohistochemical diagnosis of Hodgkin’s Lympho- ma with conventional method. SID 54: 214-221.
- Enblad G, Molin D, Glimelius I, Fischer M, Nilsson G (2007) The potential role of innate immunity in the pathogenesis of Hodgkin’s lymphoma. Hematol Oncol Clin North Am 21: 805-823.
- Glimelius I, Edström A, Amini RM, Fischer M, Nilsson G, et al. (2006) IL-9 expression contributes to the cellular composition in Hodgkin lymphoma. Eur J Haematol 76: 278-283.
- Carafa V, Nebbioso A, Altucci L (2012) Sirtuins and disease: the road ahead. Front Pharmacol 3: 4.
- Nihal M, Ahmad N, Wood GS (2014) SIRT1 is up-regulated in cutaneous T-cell lymphoma, and its inhibition induces growth arrest and apop-tosis. Cell Cycle 13: 632-640.
- Quesada AE, Nguyen ND, Rios A, Brown RE (2014) Morphoproteomics identifies constitutive acti¬vation of the mTORC2/Akt and NF-kappaB pathways and expressions of IGF-1R, Sirt1, COX-2, and FASN in peripheral T-cell lympho¬mas: pathogenetic implications and therapeu¬tic options. Int J Clin Exp Pathol 7: 8732-8739.
- Schaer DJ, Schaer CA, Buehler PW, Boykins RA, Schoedon G, et al. (2006) CD163 is the macrophage scavenger receptor for native and chemically modified hemoglobins in the absence of haptoglobin. Blood 107: 373-380.
- Law SK, Micklem KJ, Shaw JM, Zhang XP, Dong Y, et al. (1993) A new macrophage differentiation antigen which is a member of the scavenger receptor superfamily. Eur J Immunol 23: 2320-2325.
- Högger P, Dreier J, Droste A, Buck F, Sorg C, et al. (1998) Identification of the integral membrane protein RM3/1 on human monocytes as a glucocorticoid-inducible member of the scavenger receptor cysteine-rich family (CD163). J Immunol 161: 1883-1890.
- Lau SK, Chu PG, Weiss LM (2004) CD163: a specific marker of macrophages in paraffin-embedded tissues. Am J Clin Path 122: 794-801.
- Tanaka T, McRae BJ, Cho K, Cook R, Fraki JE, et al. (1983) Mammalian tissue trypsin-like enzymes. Comparative reactivities of human skin tryptase, human lung tryptase, and bovine trypsin with peptide 4-nitroanilide and thioester substrates. J Biol Chem 258: 13552–13557.
- Cromlish JA, Seidah NG, Marcinkiewicz M, Hamelin J, Johnson DA, et al. (1987) Human pituitary tryptase: molecular forms, NH2-terminal sequence, immunocytochemical localization, and specificity with prohormone and fluorogenic substrates. J Biol Chem 262: 1363-1373.
- BS Wilkins, SL Buchan, JWebster, Jones JB (2001) Tryptase-positive mast cells accompany lymphocytic as well as lymphoplasmacytic lymphoma infiltrates in bone marrow trephine biopsies. Histopathology 39: 150–155.
- Rygoł B, Kyrcz-Krzemień S, Pająk J (2007) Polskie Archiwum Medycyny Wewnętrznej 117: 1-2.
- Jang KY, Hwang SH, Kwon KS, Kim KR, Choi HN, et al. (2008) SIRT1 expression is associated with poor prognosis of diffuse large B-cell lymphoma. Am J Surg Pathol 32: 1523–1531.
- Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141: 39-51.
- Keresztes K, Szollosi Z, Simon Z, Tarkanyi I, Nemes Z, et al. (2007) Retrospective analysis of the prognostic role of tissue eosinophil and mast cells in Hodgkin’s lymphoma. Pathol Oncol Res 13: 237-242.
- Andersen MD, Kamper P, Nielsen PS, Bendix K, Riber-Hansen R, et al. (2016) Tumour-associated mast cells in classical Hodgkin’s lymphoma: correlation with histological subtype, other tumour-infiltrating inflammatory cell subsets and outcome. Eur J Haematol 96: 252-259.
- Campo E, Steven H Swerdlow, Nancy L, Pileri S, Stein H, et al. (2011) The 2008 WHO classification of lymphoid neoplasms and beyond: evolving concepts and practical applications. Blood 117: 5019-5032.
- Lister TA, Crowther D, Sutcliffe SB, Glatstein E, Canellos GP, et al. (1989) Report of a committee convened to discuss the evaluation and staging of patients with Hodgkin’s disease: Cotswolds meeting. J Clin Oncol 7: 1630-1636.
- Hsu SM, Raine L, Fanger H (1981) Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J HistochemCytochem 29: 577-580.
- Cao YW, Li WQ, Wan GX, Li XY, Du XM, et al. (2014) Correlation and prognostic value of SIRT1 and Notch1 signaling in breast cancer. J Exp Clin Cancer Res 33: 97.
- Klein JL, Nguyen TT, Bien-Willner GA, Chen L, Foyil KV, et al. (2014) CD163 immunohistochemistry is superior to CD68 in predicting outcome in classical Hodgkin lymphoma. Am J Clin Pathol 141: 381-387.
- Quesada A, Assylbekova B, Jabcuga C, Zhang R, Covinsky M, et al. (2015) Expression of Sirt1 and FoxP3 in classical Hodgkin lymphoma and tumor infiltrating lymphocytes: Implications for immune dysregulation, prognosis and potential therapeutic targeting. Int J Clin Exp Pathol 8: 13241-13248.
- Frazzi R, Valli R, Tamagnini I, Casali B, Latruffe N, et al. (2013) Resveratrol-mediated apoptosis of hodgkin lymphoma cells involves SIRT1 inhibition and FOXO3a hyperacetylation. Int J Cancer 132: 1013-1021.
- Beier UH, Wang L, Han R, Akimova T, Liu Y, et al. (2012) Histone deacetylases 6 and 9 and sirtuin-1 control Foxp3+ regulatory T cell function through shared and isoform-specific mechanisms. Sci Signal 5: ra45.
- Kwon HS, Lim HW, Wu J, Schnölzer M, Verdin E, et al. (2012) Three novel acetylation sites in the Foxp3 transcription factor regulate the sup¬pressive activity of regulatory T cells. J Immunol 188: 2712-2721.
- Tanijiri T, Shimizu T, Uehira K, Yokoi T, Amuro H, et al. (2007) Hodgkin’s reed-sternberg cell line (KM-H2) promotes a bidirectional differentia¬tion of CD4+CD25+Foxp3+ T cells and CD4+ cytotoxic T lymphocytes from CD4+ naive T cells. J Leukoc Biol 82: 576-584.
- Eades G, Yao Y, Yang M, Zhang Y, Chumsri S, et al. (2011) miR-200a regulates SIRT1 expression and epithelial to mesenchymal transition (EMT)-like transformation in mammary epithe¬lial cells. J Biol Chem 286: 25992-26002.
- Zhu Z, Jiang W, McGinley JN, Thompson HJ (2013) Defining the role of histone deacetylases in the inhibition of mammary carcinogenesis by dietary energy restriction (DER): effects of suberoylanilide hydroxamic acid (SAHA) and DER in a rat model. Cancer Prev Res (Phila) 6: 290-298.
- Subbiah V, Brown RE, McGuire MF, Buryanek J, Janku F, et al. (2014) A novel immuno¬modulatory molecularly targeted strategy for refractory Hodgkin’s lymphoma. Oncotarget 5: 95-102.
- Janku F, Oki Y, Falchook GS (2014) Activity of the mTOR inhibitor sirolimus and HDAC inhibitor vorinostat in heavily pretreated refractory Hodgkin lymphoma patients. J Clin Oncol 32: 5.
- Vaziri H, Dessain SK, Ng Eaton E, Imai SI, Frye RA, et al. (2001) hSIR2 (SIRT1) functions as an NAD-dependent p53 deacetylase. Cell 107: 149–159.
- Li S, Banck M, Mujtaba S, Zhou MM, Sugrue MM, et al. (2010) P53- induced growth arrest is regulated by the mitochondrial SirT3 deacetylase. PLoS One 5: e10486.
- Cha EJ, Noh SJ, Kwon KS, Kim CY, Park BH, et al. (2009) Expression of DBC1 and SIRT1 is associated with poor prognosis of gastric carcinoma. Ciln Cancer Res 15: 4453-4459.
- Chen J, Zhang B, Wong N, Lo AW, To KF, et al. (2011) Sirtuin 1 is upregulated in a subset of hepatocellular carcinomas where it is essential for telomere maintenance and tumor cell growth. Cancer Res 71: 4138-4149.
- Englund A, Molin D, Enblad G, Karlén J, Glimelius I, et al. (2016) The role of tumour-infiltrating eosinophils, mast cells and macrophages in Classical and Nodular Lymphocyte Predominant Hodgkin Lymphoma in children. Eur J Haematol 97: 430-438.
- Kuppers R (2009) The biology of Hodgkin's lymphoma. Nat Rev Cancer 9: 15-27.
- Klein J, Nguyen T, Bien-Willner G, Chen L, Foyil KV, et al. (2014) CD163 Immunohistochemistry Is Superior to CD68 in Predicting Outcome in Classical Hodgkin Lymphoma. Am J Clin Pathol 141: 381-387.
- Koh YW, Park CS, Yoon DH, Suh C, Huh J, et al. (2014) CD163 expression was associated with angiogenesis and shortened survival in patients with uniformly treated classical Hodgkin lymphoma. PLoS One 9: e87066.
- Tudor CS, Bruns H, Daniel C, Distel LV, Hartmann A, et al. (2014) Macrophages and dendritic cells as actors in the immune reaction of classical hodgkin lymphoma. PLoS ONE 9: e114345.
- Kayal S, Mathur S, Karak AK, Kumar L, Sharma A, et al. (2014) CD68 tumor-associated macrophage marker is not prognostic of clinical outcome in classical Hodgkin lymphoma. Leuk Lymphoma 55: 1031-1037.
- Barros MH, Hassan R, Niedobitek G (2012) Tumor-associated macrophages in pediatric classical Hodgkin lymphoma: association with Epstein-Barr virus, lymphocyte subsets, and prognostic impact. Clin Cancer Res 18: 3762-3771.
- Gupta S, Yeh S, Chami R, Punnett A, Chung C (2013) The prognostic impact of tumour-associated macrophages and Reed-Sternberg cells in paediatric Hodgkin lymphoma. Eur J Cancer 49: 3255-3261.
- Lamprecht B, Walter K, Kreher S, Kumar R, Hummel M, et al. (2010) Derepression of an endogenous long terminal repeat activates the CSF1R proto-oncogene in human lymphoma. Nat Med 16: 571-579.
- Molin D, Edstrom A, Glimelius I, Glimelius B, Nilsson G, et al. (2002) Mast cell infiltration correlates with poor prognosis in Hodgkin’s lymphoma. Br J Haematol 119: 122-124.
Citation: Harb OA, Mohamed MS, Ahmed RZ, Alattar AZ, Gertallah LM (2017) Expression of Silent Information Regulator-1 (Sirt1), CD163 and Tryptase; Implications for Immune Dys-regulation, Prognosis and Therapeutic Targeting in Classical Hodgkin Lymphoma. J Clin Exp Pathol 7:313. DOI: 10.4172/2161-0681.1000313
Copyright: © 2017 Harb OA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 4890
- [From(publication date): 0-2017 - Aug 24, 2025]
- Breakdown by view type
- HTML page views: 3953
- PDF downloads: 937