Gene Expression Analysis of Fibroblasts from Patients with Bipolar Disorder
Received: 25-Sep-2015 / Accepted Date: 16-Nov-2015 / Published Date: 23-Nov-2015 DOI: 10.4172/2472-095X.1000103
Abstract
Bipolar disorder is a severe, lifelong psychiatric disease. The main underlying pathophysiology of the disease is still incomprehensible. Various studies have suggested that many genes of small impact in combination with environmental factors contribute to the expression of the disease. In this study comparative transcriptomic profiling to characterize skin fibroblasts’ gene expression of bipolar disorder patients compared to healthy controls has been performed. Skin fibroblast cells from bipolar disorder patients (n=10) and marched healthy controls (n=5) have been cultured. RNA was extracted and then hybridized onto Illumina Human HT-12 v4 Expression BeadChips. Differentially expressed genes between bipolar disorder samples and healthy controls were identified by performing unequal t-test on log 2 transformed expression values. The resulting gene list was obtained by setting the p-value threshold to 0.05 and by removing genes that presented a fold change ≥ |0.5| (in log 2 scale). We concluded to 457 differentially expressed genes. Among them 127 showed an upregulation and 330 were downregulated. Τhe expression alterations of selected genes were validated by quantitative real-time polymerase chain reaction. In order to derive better insight into the biological mechanisms related to the differentially expressed genes, the lists of significant genes were subjected to pathway analysis and target prioritization indicating various processes such as calcium ion homeostasis, positive regulation of apoptotic process and cellular response to retinoic acid.
Keywords: Skin fibroblasts, Bipolar disorder, Transcriptome, Psychiatric diseases, Pathway analysis, Microarrays
15740Abbreviations
ADH: Alcohol Dehydrogenase; ADHD: Attention Deficit Hyperactivity Disorder; BD: Bipolar Disorder; Ct: Threshold Cycle; DE: Differentially Expressed; FC: Fold Change; FDR: False Discovery Rate; GAPDH: Glyceraldehyde-3-Phosphate Dehydrogenase; GEO: Gene Expression Omnibus; GO: Gene Ontology; KEGG: Kyoto Encyclopedia of Genes and Genomes; MAPK: Mitogen Activated Protein Kinases; PCR: Polymerase Chain Reaction; qPCR: Real-time Quantitative Polymerase Chain Reaction; SZ: Schizophrenia; SNPs: Single Nucleotide Polymorphisms; StRAnGER: Statistical Ranking Annotated Genomic Experimental Results
Introduction
Bipolar disorder (BD) also known as manic depressive illness is a prevalent and severe psychiatric disorder characterized by mania cycled with depression. Patients with BD have to deal with big suicide risk, loss of productivity, high clinical and social costs, decreased life quality of themselves and their families, lifetime duration of the disease and problems in their social relations [1,2]. BD has three subtypes: bipolar I (one or more manic or mixed episodes usually followed by major depressive episodes), bipolar II (one or more major depressive episodes followed by one or more hypomanic episodes) and cyclothymic disorder (many hypomanic episodes and depressive symptoms for more than two years) [3]. The disease is treatable mainly with lithium in combination with antidepressants, other mood stabilizers and with psychotherapy [4]. The pharmacological treatment mainly targets imbalanced biogenic monoamine neurotransmitter systems [5]. Still, the disease remains incurable with possible relapse episodes even after maintenance treatment [6].
The etiology of BD remains obscure. It seems to be a complex disease with many different underlying molecular mechanisms. There are many studies indicating a combination of environmental and genetic factors that lead to the onset of the disease. The genetic background is indicated from the high heritability of the disease based on twin, family and adoption studies [7]. Despite the strong evidence of the genetic background, the genetic causes remain unknown due to the involved complex, interacting genetic factors in combination with environmental factors. None of the susceptible genes identified so far can be characterized as the major contributor to the disease mechanism [4].
Microarray studies
The microarray technology allows studies of genetic contributions to complicated disorders. This technology provides quantification of gene expression in a genome-wide level, in different tissues [8,9]. Many studies have compared RNA levels of healthy control and BD samples mainly from brain and blood [10,11]. The aim of such studies is to identify upregulated and downregulated genes as well as the pathways that they are involved. Blood samples have the advantage that they can be easily collected. Τhe brain is the most relevant tissue for BD studies and differences in gene expression between the two tissue types are expected. Postmortem brain samples though, concern dead patients leading to decreased ability of sample collection. Additionally, taking in mind that the RNA is relatively instable, postmortem factors such as hypoxia and brain tissue pH may affect the gene expression levels. Consequently, the measured results from such studies of postmortem brain samples could lead to disturbed disease expression profile. This might also be one of the reasons for the heterogeneity that these studies present [12].
Fibroblasts as a cell model
Experiments using skin fibroblasts have been performed in studies of psychiatric diseases in search of their causes and diagnoses. Such studies are particularly important where no or only poor animal models exist for psychiatric diseases. The rationale of these experiments is based on the idea that changes in the brain, or generally alterations concerning a psychiatric disease can be modeled or reflected by changes in the physiology and biochemistry of the peripheral tissues. This idea is enhanced by the fact that fibroblast cells retain the regulatory genomic features of the individuals from where they were obtained even after several passages of culture. In this way it is possible to study how inheritable DNA and RNA mutations, or generally how modifications in genes affect normal biological functions in psychiatric diseases [13]. Fibroblast cells are considered to be free of medication, hormonal and dietary effects after four to six passages in culture, but still epigenetic effects of neuroleptics on transcripts in fibroblasts cannot be totally eliminated for the first days in their cultures [14].
Skin fibroblast cells are easily obtained, cultured and maintained and in comparison to other cell types they can be grown in larger amounts and remain stable for many generations [13]. Fibroblast cells are a particularly good model for studies of cell membrane amino acid transport. The endothelial cells of blood brain barrier express similar amino acid transport systems with fibroblast cells. There are enzymes and receptors expressed in fibroblast cells that are also expressed in neurons [15]. In addition it has been reported that fibroblasts have the capacity to be transformed into neurons, an appropriate cell type for molecular studies related to central nervous system disorders [16,17].
Fibroblast studies have provided knowledge of human genetic neurological diseases such as Lesch–Nyhan syndrome, lysosomal storage disorders and Tay-Sachs disease (Niemann-Pick disease) [18]. Moreover, fibroblast cell model has been also used in the study of molecular mechanisms implicated in BD [13]. In any case, confirmatory studies concerning brain cells may be needed when using the fibroblast cell model, as it does not fully represent the diseased tissue usually located in the brain. The relation of homeostasis and molecular alterations in fibroblast cells with the pathophysiology of the disease might not always be clear. However, fibroblast studies may point ways for further studies based on new hypothesis, drug testing and diseased individuals’ sub-classification [14].
Amino acid transport and fibroblasts
The neurotransmitters dopamine and serotonin are synthesized in the central nervous system, and their availability depends partly on their precursor’s availability. The precursors of dopamine and serotonin are tyrosine and tryptophan respectively [19]. In order to study their transport into the brain through the blood brain barrier, fibroblast cells have been used as an experimental model [15]. Studies on fibroblast cells of BD compared to healthy control fibroblast cells have shown a decreased tyrosine transport across fibroblast cell membranes. BD has been linked to altered neurotransmission systems (altered dopaminergic, serotonergic and noradrenergic functions) and to aberrations in regions of brain related to emotional processes. The altered transport of tyrosine might be an indication of decreased dopamine access into the brain as tyrosine is the precursor of dopamine. That could further affect the dopaminergic and noradrenergic neurotransmission, and subsequently other neurotransmission systems, such as the serotonergic system [20].
Aberrant amino acid transport and membrane dysfunctions
Dysfunctional amino acid transport is a repeating finding in mental diseases, such as schizophrenia (SZ), BD and autism [15,19]. These findings in combination with other studies indicate dysfunctional signal transduction as well as altered membrane metabolism and composition, considering that amino acid transporters are embedded in cell membranes [21,22]. BD has been previously related to membrane dysregulation through the membrane theory. The basic hypothesis of this theory is that abnormalities are generated due to increased removal of essential fatty acids in combination with reduced fatty acid incorporation into membrane phospholipids [23]. Additional studies on molecular mechanisms, gene mutations, gene expression alterations, cytokine effects, membrane functionality and structure, as well as transport studies are necessary in order to translate the research findings that are available so far [24].
In order to further elucidate the relation of the abovementioned molecular mechanisms with gene expression, we used high-throughput transcriptomic profiling of skin fibroblast cells from BD patients. Analyses on transcriptomic and functional level were performed in order to elucidate BD related abnormalities through BD related transcriptomic signatures in fibroblasts.
Materials and Methods
Fibroblast cultures and RNA extraction
Fibroblast cells were cultured from skin biopsies obtained from 10 patients (six men and four women aged 29–77 years) diagnosed with bipolar type-1 disorder [20], according to DSM-IV criteria [25]. Fibroblasts from 5 volunteers, without a diagnosis of a psychiatric disease were used as controls. The Ethical Committee at Karolinska University Hospital approved the study. Τhe required informed consents from patients and controls were obtained before performing the study. The fibroblast cells were cultured in 75 cm2 plastic tissue culture flasks, containing minimal essential medium supplemented with 10% fetal bovine serum, l-glutamine (2 mM/l), penicillin (100 mg/ ml), streptomycin (100 mg/ml) and Amnio-MaxTM. The cells were maintained in a humidified atmosphere of 5% CO2 in air at 37°C for approximately 4 weeks, until sufficient amount of cells had achieved. The cells were harvested by using EDTA and trypsin solutions and were washed and stored in -80°C until they were used for RNA extraction. Cell lines between 8th and 13th passages (number of splitting) were used for the experiments. All growth media, antibiotics and fetal bovine serum were obtained from Gibco Invitrogen cell culture (Sweden). Tissue culture flasks were from Costar Europe Ltd., Costar NY.
Total RNA was extracted from all samples by using NucleoSpin® RNA ll kit (Macherey & Nagel Ltd., Oensingen, Switzerland) according to the instructions provided by the manufacturer. RNA samples extracted from the five healthy control subjects were pooled into two samples for the microarray analysis, in order to limit the individual case-to-case variation of gene expression that is unrelated to BD. RNA quality and quantity for all samples were assessed using a high-resolution electrophoresis system Bioanalyzer 2100 (Agilent, Santa Clara, California).
Microarray hybridization and data analysis
Gene expression analysis was performed by Cambridge Genomic Services (Department of Pathology, Cambridge University, Tennis Court Road, Cambridge CB2 1QP) using Illumina HumanHT-12 v 4 Expression BeadChip (>47 000 probes). The data discussed in this publication have been deposited in National Center for Biotechnology Information Gene Expression Omnibus (GEO) [26] and are accessible through GEO ID: GSE69486 (http://www.ncbi.nlm.nih.gov/geo/query/ acc.cgi?acc=GSE69486).
The raw signal intensity data were imported into the Flexarray software tool [27] for microarray data analysis. Briefly, background corrected values were log2 transformed and normalized with the quantile method [28]. A probe was considered present if the detection p-value set by Illumina was below 0.01. Probes scoring above 0.01 were considered as absent. Probes absent in more than 40% of the replicates in both conditions (BD and healthy control samples) were excluded from further analysis. Data filtering was applied after normalization procedure.
Statistical analysis
For the identification of differentially expressed (DE) genes unequal t-test on log2 transformed expression values was performed. Fold change (FC) values were calculated for each gene as the difference of the mean intensity of the control samples from patient samples. For any comparison, genes with a p-value ≤ 0.05 and a FC (in log2 scale) ≥ |0.5| were considered to be differentially expressed. Multiple testing correction was applied using false discovery rate (FDR) adjustment of Benjamini and Hochberg [29], with the adjusted p-value cutoff ≤ 0.05.
Reverse transcription, real-time quantitative polymerase chain reaction (qPCR) validation and correlation with microarray data
500 ng of RNA were reverse-transcribed using Superscript III reverse transcriptase (Invitrogen, Carsland, CA, USA) and random hexamer primers according to the manufacturer’s instructions. qPCR was performed with SYBR Green I (Biorad, Hercules, CA, USA) on an iQ5 real-time polymerase chain reaction (PCR) detection system (Biorad, Hercules, CA, USA). All assays were performed in triplicate in a 25 μL reaction by a standard thermal protocol: 95°C for 3 min, followed by 40 cycles of 95°C for 30s and 60°C for 60 s. Specificity of the amplified PCR product was assessed through a melting curve analysis. The primers used in the qPCR are listed in (Table 1). Primers were designed with Primer-BLAST and one primer per pair was designed to span an exon-exon junction [30]. All of the PCR primers were synthesized by VBC-Biotech (Vienna, Austria). Threshold cycle (Ct) values obtained from the exponential phase of the qPCR amplification plot for each target transcript were normalized to Ct values from assays of transcripts encoding the glyceraldehyde-3-phosphate dehydrogenase (GAPDH). GAPDH was chosen as the housekeeping gene for the normalization of target genes as it was consistently expressed in similar levels in all microarray samples. Gene expression was presented using a modification of the 2−ΔΔCt method, first described by Livak et al. [31]. The mean expression of the two sample groups was compared using one-tailed t-test. From these values, fold-differences in the levels of transcripts between BD and healthy control cell cultures were calculated by dividing the average 2-ΔCt of the two conditions as it is described by Schmittgen et al. [32].
| Gene | Forward primer | Reverse primer |
|---|---|---|
| SLC39A8 | 5′ TCCCCACGAGTTAGGAGACT 3′ | 5′ GTTGCCCACCAAAATGCCAA 3′ |
| LPCAT2 | 5′ TCCAGGTGGCATTTAAGCTGT 3′ | 5′ AAATTGAGTCCCCTTGGGCT 3′ |
| CYP26B1 | 5′CTCATGGCATCCTGCACAGT3′ | 5′GGGATCTGGAAACCATCAAGC3′ |
| ENPP1 | 5′TTTCAAGTTATATGGCGCTACTTTC3′ | 5′TCTTGGTTACGGATGACTCTTCTT3′ |
| GAPDH | 5'GTGTCCGTCGTGGATCTGA3' | 5'GCTTCACCACCTTCTTGATGTCAT3' |
| PPARG | 5′GTCTCATAATGCCATCAGGTTTGG 3′ | 5′ TTTGCTTTGGTCAGCGGGAA 3′ |
Table 1: The nucleotide sequences of the qPCR primers used to assay gene expression are shown.
Pathway analysis and target prioritization of DE genes
Statistical enrichment analysis was performed for the list of DE genes using the Statistical Ranking Annotated Genomic Experimental Results (StRAnGER) web application [33]. StRAnGER is a bioinformatic tool that based on the gene ontology (GO) term annotations [34] and the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways [35], performs statistical overrepresentation tests, further corrected by resampling methods. More specifically, the annotation based analyses provide a list of terms and pathways, derived from hypergeometric tests with p-values smaller than 0.05. These terms have also been reordered according to bootstrapping for correction of statistical distributionrelated areas. For further prioritization of genes of interest, functional implication of genes in cellular processes is studied. Through this process candidate hub genes are indicated. For the identification of hub genes we used the bioinformatic tool GOrevenge, based on the theoretical topology of the GO terms-gene interaction network. After we have imported the list of interest we adopted the following settings: Aspect: Biological Process, Distance: Resnik, Algorithm: BubbleGene and Relaxation: 0. By selecting those settings, interactions contributing to the bias related to the existence of functionally unnecessary terms are excluded. Consequently the centrality or in other words relation of the genes to specific biological phenotypes is estimated [36].
Results
Gene expression changes
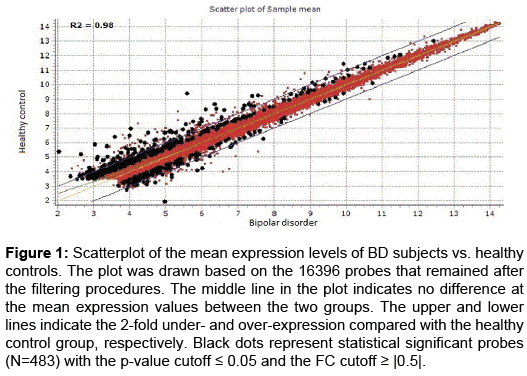
The bioinformatic analysis revealed 16396 present probes out of 47316 probes after filtering procedures. Among them 1275 probes considering only the p-value cutoff were differentially expressed between BD samples and healthy control samples. After the filtering step, the scatterplot (Figure 1) was drawn based on the 16396 remaining probes. The majority of probes were expressed in a similar way between the healthy control samples and BD samples. Statistical significant probes (N=483) with the p-value cutoff (≤ 0.05) and the FC cutoff (≥ |0.5|) are also depicted in (Figure 1).
Figure 1: Scatterplot of the mean expression levels of BD subjects vs. healthy controls. The plot was drawn based on the 16396 probes that remained after the filtering procedures. The middle line in the plot indicates no difference at the mean expression values between the two groups. The upper and lower lines indicate the 2-fold under- and over-expression compared with the healthy control group, respectively. Black dots represent statistical significant probes (N=483) with the p-value cutoff ≤ 0.05 and the FC cutoff ≥ |0.5|.
The DE genes were defined on the basis of FC cutoff of the mean expression values of the two groups and the p-value cutoff (0.05). The number of DE genes according to the corresponding FC cutoff is presented in (Table 2). After adjusting for multiple testing and applying the adjusted p-value ≤ 0.05, the number of genes dramatically decreased to 15. The list of genes after the FDR adjustment is presented in (Table 3). These results yielded conservative results preventing from further gene expression analysis. Thus standard statistical analysis was used for further exploration of the biological signal of the data. More specifically, genes with a p-value ≤ 0.05 and a FC (in log2 scale) ≥ |0.5| were used for the pathway analysis and the identification of hub genes. The DE genes according to these criteria are listed in (Supplementary Table 1).
| Upregulated genes | Downregulated genes | DE genes | FC cutoff (log2) |
|---|---|---|---|
| 546 | 683 | 1229 | No cutoff |
| 127 | 330 | 457 | 0.5 |
| 16 | 76 | 92 | 1 |
Table 2: Number of DE genes of BD patients as compared to healthy control samples with p-value cutoff ≤0.05 at different FC cutoff levels.
| Gene Symbol | Adjusted p-value | Fold change (log2) | DEFINITION |
|---|---|---|---|
| ACYP2 | 0.0491466 | -0.1139538 | acylphosphatase 2, muscle type |
| ADH1B | 0.0491466 | -2.881953 | alcohol dehydrogenase IB (class I), beta polypeptide |
| CASP2 | 0.0491466 | -0.41584188 | caspase 2, apoptosis-related cysteine peptidase, transcript variant 3 |
| CD164 | 0.02969887 | -0.7348687 | CD164 molecule, sialomucin |
| EPHB6 | 0.0491466 | -2.543039 | EPH receptor B6 |
| SEPT7P3 | 0.02516149 | -1.516207 | septin 7 pseudogene 3 |
| LRPAP1 | 0.0491466 | 0.5580598 | low density lipoprotein receptor-related protein associated protein 1 |
| LRRC56 | 0.0491466 | 1.335376 | leucine rich repeat containing 56 |
| LSR | 0.0491466 | -0.9326392 | lipolysis stimulated lipoprotein receptor, transcript variant 3 |
| LTF | 0.0491466 | 0.6395543 | lactotransferrin |
| NOMO1 | 0.02516149 | 0.568135033 | NODAL modulator 1 |
| RPP30 | 0.02516149 | -1.13074 | ribonuclease P/MRP 30kDa subunit |
| RPS28 | 0.0491466 | -0.3264609 | ribosomal protein S28 |
| SLC7A1 | 0.0491466 | 0.4278626 | solute carrier family 7 (cationic amino acid transporter, y+ system), member 1 |
| YME1L1 | 0.0491466 | 0.27648385 | YME1-like 1 (S. cerevisiae), nuclear gene encoding mitochondrial protein, transcript variant 1 |
Table 3: DE genes based on the adjusted p-value cutoff (0.05) that occurred after the FDR Benjamin-Hochberg adjustment.
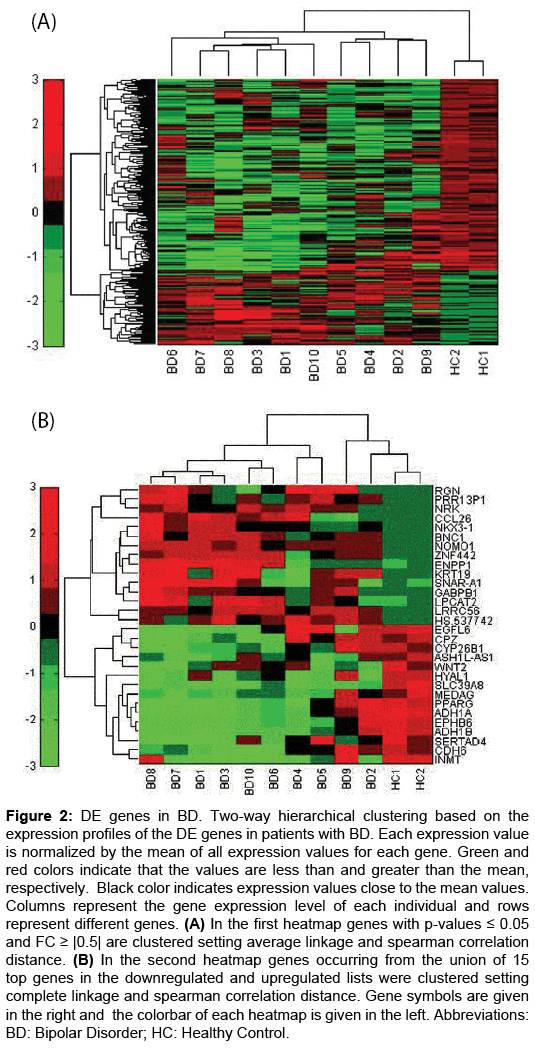
The hierarchical clustering of the gene expression could separate BD subjects and healthy control samples as shown in (Figure 2A and 2B), where (Figure 2A) depicts the expression profile of all DE genes, while (Figure 2B) depicts the expression profile of the top 15 downregulated genes and the top 15 upregulated genes. Genes of interest that performed particular expression alterations are described in the following sections.
Figure 2: DE genes in BD. Two-way hierarchical clustering based on the expression profiles of the DE genes in patients with BD. Each expression value is normalized by the mean of all expression values for each gene. Green and red colors indicate that the values are less than and greater than the mean, respectively. Black color indicates expression values close to the mean values. Columns represent the gene expression level of each individual and rows represent different genes. (A) In the first heatmap genes with p-values ≤ 0.05 and FC ≥ |0.5| are clustered setting average linkage and spearman correlation distance. (B) In the second heatmap genes occurring from the union of 15 top genes in the downregulated and upregulated lists were clustered setting complete linkage and spearman correlation distance. Gene symbols are given in the right and the colorbar of each heatmap is given in the left. Abbreviations: BD: Bipolar Disorder; HC: Healthy Control.
qPCR results
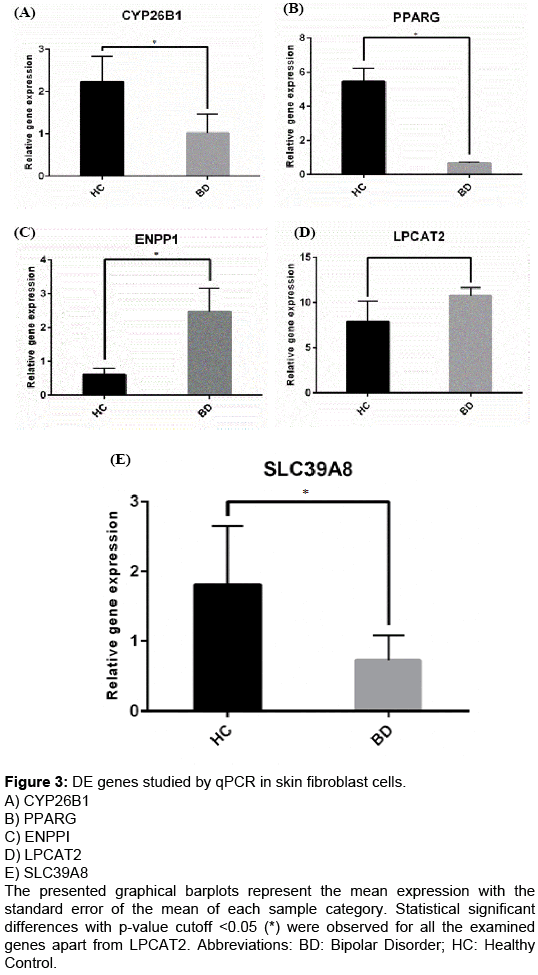
In order to validate the microarray experiment, we compared the expression levels of five DE representative genes, obtained from the qPCR and the microarray experiment. The selection of genes that were chosen for the qPCR analysis was based on their high differential expression as well as on the fact that some of them were shown to be master regulators after the GOrevenge analysis (PPARG, ENPP1). All genes were confirmed to show the same directional changes in skin fibroblasts of BD samples as compared to healthy controls (Table 4; Figure3A-3E).
| FOLD CHANGEa | P-VALUE | |||
|---|---|---|---|---|
| Gene | microarray | qPCR | microarray | qPCR |
| SLC39A8 | -10.0932 | -2.4635 | 0.042 | 0.0378 |
| PPARG | -7.722 | -7.0811 | 0.0015 | 0.0005 |
| CYP26B1 | -3.5438 | -2.1790 | 0.0067 | 0.0489 |
| ENPP1 | 3.4367 | 4.1257 | 0.0152 | 0.0392 |
| LPCAT2 | 2.7212 | 1.3703 | 0.016 | 0.3060 |
Table 4: Comparison of the qPCR and microarray results for the expression alterations of genes SLC39A8, PPARG, CYP26B1, ENPP1, and LPCAT2 in skin fibroblast cells of BD patients compared to healthy controls.
Figure 3: DE genes studied by qPCR in skin fibroblast cells.
A) CYP26B1
B) PPARG
C) ENPPI
D) LPCAT2
E) SLC39A8
The presented graphical barplots represent the mean expression with the standard error of the mean of each sample category. Statistical significant differences with p-value cutoff <0.05 (*) were observed for all the examined genes apart from LPCAT2. Abbreviations: BD: Bipolar Disorder; HC: Healthy Control.
Pathway analysis
The DE genes of (Supplementary Table 1) were submitted to StrAnGER web application for the elucidation of the overrepresented GO terms and KEGG pathways. Full results are presented in (Supplementary Tables 2-13).
Identification of candidate hub genes
GOrevenge online tool was used in order to identify genes that play a central role according to the GO tree. The list of all genes that have shown to present differential expression, as well as the lists of the upregulated and the downregulated genes were submitted to GOrevenge and the analysis was performed based on GO term annotation of Homo sapiens. Top candidate linker genes, meaning genes participating in many different cellular processes, were derived after the gene prioritization (Supplementary Tables 14-16).
Discussion
Expression alterations of selected hub genes
The genes that were shown to be important in our study based on our various bioinformatic analytical tools were examined in relation to their implication in BD and in related mental diseases. More specifically by applying the statistical criteria and according to GOrevenge results (Supplementary Table 14), PPARG and ENPP1 were among the candidate hub genes and their gene expression alterations between BD and healthy control samples have been also confirmed from the qPCR analysis. The gene PPARG encodes a peroxisome proliferator-activated receptor that belongs to a family of nuclear receptors, while ENPP1 gene encodes an ecto-nucleotide pyrophosphatase/phosphodiesterase, which is a type II transmembrane glycoprotein [provided by RefSeq].sw
Findings from the GOrevenge analysis showed that the upregulated MAP2K6 and the downregulated MAPK9 (Supplementary Table 14) genes were also candidate hub genes. Mitogen activated protein kinases (MAPK) are generally involved in neuronal differentiation, survival and plasticity and various other cellular processes. MAP2K6 belongs to the signal transduction pathways related to cell cycle arrest, transcription initiation and apoptosis. MAPK9 kinase targets transcription factors, and as a result facilitates early gene expression in response to cell stimuli [provided by RefSeq]. The importance of these genes is enhanced by the action of lithium and valproate on MAPK/ extracellular signal-regulated kinases signaling cascades, as it has been reported [37].
GOrevenge analysis (Supplementary Table 14) also showed that WNT2 was among the downregulated top ranked genes. WNT2 belongs to a family of genes that encode secreted signaling proteins [provided by RefSeq]. According to the literature the Wnt signaling pathway may be affected in BD. More specifically, it has been found that in monozygotic twins discordant for BD there is a differential expression in genes involved in Wnt signaling pathway. It should be pointed out that the already mentioned PPARG gene belongs in the Wnt signaling pathway. Association studies of 317 families with 554 BD offspring and their parents identify a relation of BD with the peroxisome proliferator-activated receptor (PPAR) family. In general there are a lot of indications suggesting Wnt polymorphisms are associated with BD probably through inflammatory pathways. Further studies are suggested for the better understanding of the associations between Wnt gene polymorphisms and BD and their relations with the serotonin signaling [38]. It is of great interest that WNT2 gene is also involved in the etiology of autism [39].
Comparison with datasets of patients with BD and other psychiatric diseases
In this study we compared our significant genes (Supplementary Table 1) with the genes identified as significant in a gene expression study of postmortem brains of BD patients [11] and 38 genes were found to be common (see Supplementary Table 17). The DE gene list of this study was also compared with the corresponding list from a mega-analysis of genome-wide gene expression studies of post-mortem brain BD samples [12]. The following genes were found to be common: EPHB6, CLEC3B, NID2, RHOBTB3, KLF4, PDE4DIP, and LPCAT2. We also performed another comparison with a list of genes located into regions repeatedly linked with BD [40]. According to this comparison the following genes were found to be common: CD164, SLC25A10, CIDEA, MBP, RIPK4, APOL6, FOXRED2 and SMTN. In a genome-wide association study of BD in Canadian and UK populations [41] among the identified single nucleotide polymorphisms (SNPs), genes TBC1D22B, MAP2K6 and ZNF536 from our list were included. In a list of DE genes from biopsied olfactory neuroepithelium of BD patient [42] genes HYAL1, UGP2 and BAZ1A from our list were identified.
It is very interesting to comment some of the top downregulated genes, including ADH1A and ADH1B (Supplementary Table 1). Both genes encode alcohol dehydrogenase (ADH) enzyme family members [provided by RefSeq]. The genes of ADH isoforms are expressing enzymes for the catalysis of retinol and ethanol oxidation as well as enzymes implicated in the metabolism of neurotransmitters related to BD and other mental diseases (e.g. norepinephrine, dopamine and serotonin). Zuo et al. studied the associations between neuropsychiatric diseases and ADH variants demonstrating common ADH variants associated with SZ and autism. More specifically rare ADH variants are associated with alcohol dependence and common ADH variants with SZ and autism, possibly affecting the metabolic pathways of monoamines in all psychiatric diseases. ADH1B encodes the enzyme ββADH, which inhibits the metabolite 5-hydroxyindole acetaldehyde to 5-hydroxytryptophol and enhances the formation of the metabolite 5-hydroxyindoleacetic acid, which is an important metabolite of serotonin [43]. Since 5-hydroxyindoleacetic acid is a reflection of serotonin, these findings indicate altered serotonin availability. Serotonin neurotransmission is strongly correlated to BD and there are studies indicating high levels of this metabolite in postmortem brains (frontal and parietal cortex) of BD patients [44]. Other studies on manic and depressed patients have identified decreased levels in 5-hydroxyindoleacetic acid in the cerebrospinal fluid. Of note, the selective serotonin reuptake inhibitors are used for the treatment of mania and depression, enhancing the importance of the serotonin metabolism in this psychiatric disorder [20]. ADH1A variation is also associated to personality traits and substance dependence. The encoded enzyme aaADH enzyme probably oxidizes retinol. Retinol is playing an important role for the preservation of the dopaminergic neurons of the brain and of dopamine, serotonin and norepinephrine [45].
Another important downregulated gene which was identified in this study is SLC39A8 (Table 4). The altered expression of this gene between BD samples and healthy controls was also confirmed by the qPCR analysis. This gene is a solute carrier, with similar structural characteristics of zinc transporters. It is located in the plasma membrane and mitochondria, and participates in the import of zinc into the cells during inflammation. In a study of Carrera et al., where the authors look for nonsynonymous SNPs that show associations with SZ of European population, the downregulated gene SLC39A8 is detected [46].
Statistically significant pathways
Among the GO categories and KEGG pathways presented in (Supplementary Tables 2-13), emphasis was given in specific categories, namely cellular response to retinoic acid and retinoic metabolism, circadian rhythm regulation, calcium ion homeostasis, small GTPase mediated signal transduction and cell cycle and apoptosis related terms.
Cellular response to retinoic acid and retinoic metabolism
The GO term analysis resulted in cellular response to retinoic acid including the downregulated genes PPARG, AQP1, TNC, CYP26B1 as well as WNT2 and the KEGG pathway analysis resulted in retinoic acid metabolism including the downregulated genes ADH1A, ADH1B, CYP26B1 and the upregulated gene RETSAT. In accordance to these results a study on brains from patients with SZ, BD, and major depression demonstrates upregulation of the retinoic acid-inducible gene 1, indicating possible changes in signaling pathways such as retinoic acid pathway [47]. It has been also suggested that there is a relationship between affective disorders and retinoids. The indications for such a support are clinical and preclinical results for either decreased or increased levels of retinoic acid related to depression symptoms. Mouse models exposed to 13-cis-retinoic acid resulted in depression-like behavior and a decrease in learning and memory. Retinoic acid has also the ability to cross into the brain and moderate directly the neurotransmitter system of hippocampus and striatum, both associated to depression mechanisms [48].
Regulation of circadian rhythm
The functional analysis detected among the downregulated GO terms the regulation of circadian rhythm represented by the genes PRKDC, MAPK9, PPARG and EZH2. Circadian abnormalities have been related to BD [49]. It is typical for BD patients to present alterations in their sleep and wake cycle during the state of mania and depression. The circadian abnormalities are also indicated through measurements of body temperature and hormonal secretion profiles. Fibroblasts have been used as a model for studies on circadian oscillators of BD patients. These studies are based on the fact that similar features of the clock mechanisms lie behind the circadian related gene expression of fibroblasts and of the central clock located in the suprachiasmatic nuclei of the anterior hypothalamus. Alterations in the expression of clockcontrolled genes may cause other downstream effects, contributing to the pathogenesis of BD [38].
Small GTPase mediated signal transduction
Among the GO terms of the analysis, the regulation of small GTPase mediated signal transduction was identified according to the downregulated genes ARHGAP40, ARHGAP12, ARAP3, RHOT1, HMHA1, ARAP1 and ARHGAP29. Previous microarray experiments on postmortem brain samples have also shown that genes involved in G-protein signaling pathways perform an altered expression in patients with BD. The transcriptional changes in the second-messenger signaling pathways of BD patients may have a regulatory effect on downstream molecules of signal transduction, on cell membrane receptors and generally on cell response to extracellular stimuli. Other studies in BD show alterations in the subunits of G-proteins and in signal transduction. Additionally, lithium and other antidepressant drugs act on second-messenger signaling system [50].
Cell cycle and apoptotic alterations
The GO term analysis also identified differences in cell cycle related GOs, such as downregulation of mitotic cell cycle (PIM1, BIRC5, TTC28) and downregulation of epithelial cell proliferation (LAMC1, OSR2, GAS1, HYAL1). Additionally GO terms related to apoptosis were identified, such as downregulation of positive regulation of apoptotic process (CAMK1D, MAPK9, CASP2, FAM162A, FLCN, DDX20, BNIP3L, LPAR1, SLIT2, PRKDC, PPARG, AIFM2) and positive regulation of apoptotic signaling pathway (PIM1, BIRC5, TT28). Expression profile of fibroblasts from schizophrenic patients also concludes to altered expression of mRNA transcripts and proteins related to cell cycle [51]. Gene expression profiling of biopsied olfactory neuroepithelium also identifies genes related to apoptosis and cell cycle. Increase of cell death reported in this group enhanced the hypothesis of an altered balance between neurogenesis and cell death in BD [42].
Calcium ion homeostasis
Concerning the GO term calcium ion homeostasis the following genes were downregulated: MUC1, WNT2, CYP26B1, AQP1 and PPARG. Calcium ions are very important for the regulation of the synthesis and release of neurotransmitters as well as for the neuronal excitability. Deregulated Ca2+ signaling is a reproducible observation in BD studies. Thus, many studies of peripheral cells from BD samples have investigated intracellular Ca2+. These studies report altered Ca2+ release and concentration in platelets, lymphocytes and neutrophils of BD [49]. Genome-wide association studies in BD conclude to a small number of replicated loci, including CACNA1C an alpha-1 subunit of a voltage-dependent calcium channel. Calcium channels facilitate the influx of calcium ions into the cell upon membrane polarization. Another replicated polymorphism is identified in ANK3, which is responsible for the regulation of assembly of voltage-gated sodium channels at the node of Ranvier. These polymorphisms indicate altered membrane organization and possible channelopathies [52]. In a recent study the transcriptome of pluripotent stem cells, which were derived from skin fibroblasts of BD patients and were differentiated into neurons is examined, and among the differentiated genes there are genes involved in calcium signaling [52]. These findings are in relevance to the GO terms identified as highly ranked in this study, such as calcium ion homeostasis, receptor complex and receptor internalization.
Amino acid transport and BD
Consisting findings of aberrant amino acid transport across the cell membrane of fibroblast cells from patients with SZ, BD, autism and Attention Deficit Hyperactivity Disorder (ADHD) indicate alterations in the expression levels of the genes encoding the amino acid transporters [15,20,53,54].
This study has shown the downregulation of the gene SLC6A6 (Supplementary Table 1), which encodes a membrane protein, member of sodium and chloride-ion dependent transporters responsible for the transport of taurine and beta-alanine [provided by RefSeq]. Aberrant alanine transport is also reported in fibroblast cells from children with ADHD and autism [15,54]. It has to be mentioned that alanine transport is related to tyrosine transport in a competitive way [55]. It is also noticed that gene SLC7A1 (Table 3), which encodes a solute carrier family 7 (cationic amino acid transporter, y+ system), member 1 [provided by RefSeq] presents a trend towards being upregulated. Although tyrosine is transported through the isoforms of system L encoded by other genes, aberration in the cationic amino acid transporter, y+ system may also affect tyrosine transport [56].
Apart from the genes encoding specifically the amino acid transporters, the altered expression of other genes could also contribute to the dysregulation of the observed amino acid transport systems. Such downregulated genes are involved in the molecular function of ATPase activity, such as DDX20, MSH5, RFC3, MYO1E, FIGNL1, SPAST, DDX51, VWA8, MCM4, RHOBTB3, ATP2B1 and TOP1MT. There are indications for deregulated cation distribution in patients with major affective disorders for the last 30 years. The first findings reported abnormalities in sodium balance. Later findings mention more specific evidence of alterations in the activity of Na+, K+ adenosine triphosphatase of BD patients. This enzyme is responsible for the active transport of sodium and potassium through the cell membrane. Additionally, active amino acid transport systems may be subsequently influenced concluding to deregulation of the active transport of amino acids, implicated in BD [57].
The gene PLA2R1, which encodes a phospholipase A2 receptor has been found to be downregulated in this study and since this receptor participates in clearance of phospholipase A2 this gene might result in the increase of action of this phospholipase, a hypothesis which is in accordance with previous findings in BD and in other psychiatric disorders [58,59]. Nevertheless phospholipase A2 activity as well as phospholipase C and inositol monophosphatase are inhibited by lithium and this leads to alterations in phospholipid and fatty acid signal transduction mechanisms in BD patients [24].
ADH1A (presenting the greater downregulation among the DE genes) and ADH1B genes are implicated in tyrosine metabolism as well as in fatty acid metabolism (Supplementary Table 13). KEGG pathway analysis with StRAnGER actually ranked these pathways among the statistical significant pathways resulted from the downregulated genes. Tryptophan metabolism was also identified among the significant pathways due to the downregulated gene AFMID and INMT. Alterations in the metabolism of these amino acids suggest further alterations in the dopamine and serotonin availability and functionality [55].
Conclusion
This is the first microarray study of skin fibroblast cells from BD patients that has been performed in order to elucidate the underlying biological mechanisms. The RNA samples from the five healthy control subjects have been sub-pooled into two samples to limit the individual caseto- case variation of gene expression that is unrelated to BD. In this way Illumina HumanHT-12 v4 Expression BeadChip with a 12-sample format has been used, enabling relative large gene expression studies to be completed relatively quickly with a modest decrease in the total number of subjects. Several studies have shown that this sampling approach is statistically valid and can offer at the same time equal power and even increase the effectiveness of microarray experiments [60].
The medication status of the BD patients was recorded at the stage of sample collection. Due to the heterogeneity of the utilized drugs and the lack of knowledge of the way they affect the neurobiological mechanisms, it would be difficult to make conclusions concerning their effects. Still, it is improbable that the medication profile at the period of biopsy affects the results, as the fibroblast cells were cultured in vitro for some generations. Preferably, the outcomes from the fibroblasts of BD patients may reflect inherited characteristics that have the ability to pass on through many cell passages [14].
In conclusion this study revealed 457 genes that were differentially expressed. Among them, the five top upregulated genes were ENPP1, SNAR-A1, LPCAT2, KRT19 as well as RGN, and the five top downregulated genes were ADH1A, SLC39A8, PPARG, ADH1B and INMT. The genes of ADH isoforms are involved in the metabolism of neurotransmitters such as dopamine [43]. The downregulation of genes ADH1A and ADH1B may be related to findings concerning decreased levels of the catecholamine metabolite homovanillic acid in cerebrospinal fluid of patients in the state of depression. Thus, dopamine withdrawal in depression states and dopamine excess in the manic states continuously reported in BD may be related to DE genes involved in the catecholaminergic metabolism. The downregulated genes ADH1B and INMT are involved in the serotonin metabolism. Alterations in serotonin metabolic pathways are indicated by other studies reporting lower levels of the serotonin metabolite 5-hydroxyindoleacetic acid in the cerebrospinal fluid, which is correlated to manic symptoms. Additionally, in depressed and euthymic patients, a decrease in serotonergic activity has been observed [61].
Additionally, we identified statistically significant GO terms that are related to pathways such as calcium ion homeostasis, regulation of mitotic cell cycle and epithelial cell proliferation. Furthermore we identified DE genes related to small GTPase mediated signal transduction, cellular response to retinoic acid and retinoic metabolism and circadian rhythm regulation. Aberrant Ca2+ signaling is a repeated observation in BD studies [52], and this study indicates that these Ca2+ abnormalities may be related to gene expression alterations in BD patients. Cell cycle alterations are also reported after gene expression profiling of skin fibroblasts from schizophrenic patients and gene expression profiling of olfactory neuroepithelium from BD patients [42,51]. Deregulations in small GTPase signal transduction indicated from this study are in accordance with previous findings concerning the effect of lithium and other antidepressants on second-messenger signaling systems [50]. Retinoic acid can enter the brain and affect the neurotransmitter systems in striatum and hippocampus, which are related to the depression mechanisms [48]. Alterations in the expression of circadian rhythm related genes may lead to the observed abnormalities of sleep wake cycles of manic states in BD and may also cause further downstream effects involved in the BD pathogenesis [38].
It should also be mentioned that concerning the amino acid transport mechanisms which have been shown to be implicated in the pathophysiology of BD, various genes and pathways have been found to be involved, such as the taurine and alanine transporter SLC6A6, the cationic amino acid transporter SLC7A1, the phospholipase A2 receptor PLA2R1, tyrosine and tryptophan metabolic pathways, ATPase activity and fatty acid metabolism. These findings may explain the alterations observed in tyrosine transport mechanisms in bipolar type-1 disorder, implying an abnormal access of tyrosine in the brain that could lead to dysfunctional neurotransmitter systems [62].
Author Recommended Internet Resources
The National Center for Biotechnology Information (NCBI) Gene Expression Omnibus website: www.ncbi.nlm.nih.gov/geo
StRAnGER web application: grissomweb.vima.ekt.gr/stranger2
References
- Townsend J,Altshuler LL (2012) Emotion processing and regulation in bipolar disorder: a review. Bipolar Disord 14: 326-339.
- Hilty DM,Leamon MH, Lim RF, Kelly RH, Hales RE (2006) A review of bipolar disorder in adults. Psychiatry (Edgmont) 3: 43-55.
- American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders (5th edition.). VA: American Psychiatric Publishing, Arlington.
- El-Mallakh RS,Elmaadawi AZ, Loganathan M, Lohano K, Gao Y (2010) Bipolar disorder: an update. Postgrad Med 122: 24-31.
- Drago A,Crisafulli C, Calabro M, Serretti A (2015) Enrichment pathway analysis. The inflammatory genetic background in Bipolar Disorder. J Affect Disord 179: 88-94.
- Kato T (2007) Molecular genetics of bipolar disorder and depression. Psychiatry ClinNeurosci 61: 3-19.
- Brennand KJ Simone A, Tran N, Gage FH (2012) Modeling psychiatric disorders at the cellular and network levels. Mol Psychiatry 17: 1239-1253.
- Kumarasinghe N,Tooney PA, Schall U (2012) Finding the needle in the haystack: a review of microarray gene expression research into schizophrenia. Aust N Z J Psychiatry 46: 598-610.
- Singh A, Kumar N (2013) A review on DNA microarray technology. IJCRR. 5: 1-5.
- Beech RD, Lowthert L, Leffert JJ, Mason PN, Taylor MM, et al. (2010) Increased peripheral blood expression of electron transport chain genes in bipolar depression. Bipolar Disord. 12: 813-824.
- Chen H, Wang N, Zhao X, Ross CA, O'Shea KS, et al. (2013) Gene expression alterations in bipolar disorder postmortem brains. Bipolar Disord 15: 177-187.
- Seifuddin F,Pirooznia M, Judy JT, Goes FS, Potash JB, et al. (2013) Systematic review of genome-wide gene expression studies of bipolar disorder. BMC Psychiatry 13: 213.
- Mahadik SP, Mukherjee S, Horrobin DF, Jenkins K, Correnti EE, et al. (1996) Plasma membrane phospholipid fatty acid composition of cultured skin fibroblasts from schizophrenic patients: comparison with bipolar patients and normal subjects. Psychiatry Res. 63: 133-42.
- Johansson AS, Owe-Larsson B, Asp L, Kocki T, Adler M, et al. (2013) Activation of kynurenine pathway in ex vivo fibroblasts from patients with bipolar disorder or schizophrenia: cytokine challenge increases production of 3-hydroxykynurenine. J Psychiatr Res 47: 1815-1823.
- Fernell E,Karagiannakis A, Edman G, Bjerkenstedt L, Wiesel FA, et al. (2007) Aberrant amino acid transport in fibroblasts from children with autism. NeurosciLett 418: 82-86.
- Han DW, Tapia N, Hermann A, Hemmer K, Hoing S, et al. (2012) Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell 10: 465-472.
- Cattane N,Minelli A, Milanesi E, Maj C, Bignotti S, et al. (2015) Altered gene expression in schizophrenia: findings from transcriptional signatures in fibroblasts and blood. PLoS One 10: e0116686.
- Connolly GP (1998) Fibroblast models of neurological disorders: fluorescence measurement studies. Trends PharmacolSci 19: 171-177.
- Flyckt L,Edman G, Venizelos N, Borg K (2011) Aberrant tyrosine transport across the fibroblast membrane in patients with schizophrenia--indications of maternal inheritance. J Psychiatr Res 45: 519-525.
- Persson ML, Johansson J, Vumma R, Raita J, Bjerkenstedt L, et al. (2009) Aberrant amino acid transport in fibroblasts from patients with bipolar disorder. NeurosciLett 457: 49-52.
- Ramchand CN, Peet M, Clark AE, Gliddon AE, Hemmings GP (1996) Decreased tyrosine transport in fibroblasts from schizophrenics: implications for membrane pathology. Prostaglandins LeukotEssent Fatty Acids. 55: 59-64.
- Bezchlibnyk Y, Young LT (2002) The neurobiology of bipolar disorder: focus on signal transduction pathways and the regulation of gene expression. Can J Psychiatry 47: 135-148.
- Bennett CN,Horrobin DF (2000) Gene targets related to phospholipid and fatty acid metabolism in schizophrenia and other psychiatric disorders: an update. Prostaglandins LeukotEssent Fatty Acids 63: 47-59.
- Horrobin DF, Bennett CN (1999) Depression and bipolar disorder: relationships to impaired fatty acid and phospholipid metabolism and to diabetes, cardiovascular disease, immunological abnormalities, cancer, ageing and osteoporosis. Possible candidate genes. Prostaglandins LeukotEssent Fatty Acids. 60: 217-234.
- American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders (DSM-IV). American Psychiatric Press,Washington, DC.
- Edgar R,Domrachev M, Lash AE (2002) Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 30: 207-210.
- Blazejczyk M (2007) FlexArray: Statistical Data Analysis Software for Gene Expression Microarrays, Made with Life Scientists in Mind. Canadian Bioinformatics Help Desk (CBHD) Newsletter 69.
- Bolstad BM, Irizarry RA, Astrand M, Speed TP (2003) A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics. 19: 185-193.
- Hochberg Y,Benjamini Y (1990) More powerful procedures for multiple significance testing. Stat Med 9: 811-818.
- Ye J,Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, et al. (2012) Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics 13: 134.
- Livak KJ,Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta DeltaC(T)) Method. Methods 25: 402-408.
- Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3: 1101-1108.
- Chatziioannou AA, Moulos P (2011) Exploiting statistical methodologies and controlled vocabularies for prioritized functional analysis of genomic experiments: the StRAnGER web application. Frontiers in Neuroscience. 5.
- Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, et al. (2000) Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet 25: 25-29.
- Kanehisa M, Goto S (2000) KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Research 28: 27-30.
- Moutselos K, Maglogiannis I, Chatziioannou A (2011) GOrevenge: a novel generic reverse engineering method for the identification of critical molecular players, through the use of ontologies. IEEE Trans Biomed Eng. 58: 3522-3527.
- Howrigan DP, Laird NM, Smoller JW, Devlin B, McQueen MB (2011) Using linkage information to weight a genome-wide association of bipolar disorder. Am J Med Genet B Neuropsychiatr Genet 156B: 462-471.
- Watkins CC,SawaAPomper MG (2014) Glia and immune cell signaling in bipolar disorder: insights from neuropharmacology and molecular imaging to clinical application. Transl Psychiatry 4: e350.
- Marui T,Funatogawa I, Koishi S, Yamamoto K, Matsumoto H, et al. (2010) Association between autism and variants in the wingless-type MMTV integration site family member 2 (WNT2) gene. Int J Neuropsychopharmacol 13: 443-449.
- Serretti A,Mandelli L (2008) The genetics of bipolar disorder: genome 'hot regions,' genes, new potential candidates and future directions. Mol Psychiatry 13: 742-771.
- Xu W, Cohen-Woods S, Chen Q, Noor A, Knight J, et al. (2014) Genome-wide association study of bipolar disorder in Canadian and UK populations corroborates disease loci including SYNE1 and CSMD1. BMC Med Genet 15: 2.
- McCurdy RD, Feron F, Perry C, Chant DC, McLean D, et al. (2006) Cell cycle alterations in biopsied olfactory neuroepithelium in schizophrenia and bipolar I disorder using cell culture and gene expression analyses. Schizophr Res. 82: 163-173.
- Zuo L, Wang K, Zhang XY, Pan X, Wang G, et al. (2013) Association between common alcohol dehydrogenase gene (ADH) variants and schizophrenia and autism. Hum Genet 132: 735-743.
- Young LT,Warsh JJ, Kish SJ, Shannak K, Hornykeiwicz O (1994) Reduced brain 5-HT and elevated NE turnover and metabolites in bipolar affective disorder. Biol Psychiatry 35: 121-127.
- Zuo L, Gelernter J, Kranzler HR, Stein MB, Zhang H, et al. (2010) ADH1A variation predisposes to personality traits and substance dependence. Am J Med Genet B Neuropsychiatr Genet 153B: 376-386.
- Carrera N,Arrojo M, Sanjuan J, Ramos-Rios R, Paz E, et al. (2012) Association study of nonsynonymous single nucleotide polymorphisms in schizophrenia. Biol Psychiatry 71: 169-177.
- Haybaeck J, Postruznik M, Miller CL, Dulay JR, Llenos IC, et al. (2015) Increased expression of retinoic acid-induced gene 1 in the dorsolateral prefrontal cortex in schizophrenia, bipolar disorder, and major depression. Neuropsychiatr Dis Treat. 11: 279-289.
- Bremner JD, Shearer KD, McCaffery PJ (2012) Retinoic acid and affective disorders: the evidence for an association. J Clin Psychiatry 73: 37-50.
- Manji HK, Quiroz JA, Payne JL, Singh J, Lopes BP, et al. (2003) The underlying neurobiology of bipolar disorder. World Psychiatry 2: 136-146.
- Ryan MM, Lockstone HE, Huffaker SJ, Wayland MT, Webster MJ, et al. (2006) Gene expression analysis of bipolar disorder reveals downregulation of the ubiquitin cycle and alterations in synaptic genes. Mol Psychiatry 11: 965-978.
- Wang L,Lockstone HE, Guest PC, Levin Y, Palotas A, et al. (2010) Expression profiling of fibroblasts identifies cell cycle abnormalities in schizophrenia. J Proteome Res 9: 521-527.
- Chen HM, DeLong CJ, Bame M, Rajapakse I, Herron TJ, et al. (2014) Transcripts involved in calcium signaling and telencephalic neuronal fate are altered in induced pluripotent stem cells from bipolar disorder patients. Transl Psychiatry 4: e375.
- Flyckt L, Venizelos N, Edman G, Bjerkenstedt L, Hagenfeldt L, et al. (2001) Aberrant tyrosine transport across the cell membrane in patients with schizophrenia. Arch Gen Psychiatry 58: 953-958.
- Johansson J, Landgren M, Fernell E, Vumma R, Ahlin A, et al. (2011) Altered tryptophan and alanine transport in fibroblasts from boys with attention-deficit/hyperactivity disorder (ADHD): an in vitro study. Behav Brain Funct. 7: 40.
- Vumma R, Wiesel FA, Flyckt L, Bjerkenstedt L, Venizelos N (2008) Functional characterization of tyrosine transport in fibroblast cells from healthy controls. NeurosciLett 434: 56-60.
- Hyde R, Taylor PM, Hundal HS (2003) Amino acid transporters: roles in amino acid sensing and signalling in animal cells. Biochem J 373: 1-18.
- Logotheti M, Papadodima O, Venizelos N, Chatziioannou A, Kolisis F (2013) A Comparative Genomic Study in Schizophrenic and in Bipolar Disorder Patients, Based on Microarray Expression Profiling Meta-Analysis. Scientific World Journal.
- Ross BM, Hughes B, Kish SJ, Warsh JJ (2006) Serum calcium-independent phospholipase A2 activity in bipolar affective disorder. Bipolar Disord 8: 265-270.
- Ross BM, Hudson C, Erlich J, Warsh JJ, Kish SJ (1997) Increased phospholipid breakdown in schizophrenia. Evidence for the involvement of a calcium-independent phospholipase A2. Arch Gen Psychiatry. 54: 487-494.
- Peng X,Wood CL, Blalock EM, Chen KC, Landfield PW, et al. (2003) Statistical implications of pooling RNA samples for microarray experiments. BMC Bioinformatics 4: 26.
- Semeniken K, Dudás B (2012) Bipolar Disorder: Diagnosis, Neuroanatomical and Biochemical Background. InTech.
- Lieberman HR, Thompson LA, Caruso CM, Niro PJ, Mahoney CR, et al. (2015) The catecholamine neurotransmitter precursor tyrosine increases anger during exposure to severe psychological stress. Psychopharmacology (Berl) 232: 943-951.
Citation: Logotheti M, Papadodima O, Chatziioannou A, Venizelos N, Kolisis F (2015) Gene Expression Analysis of Fibroblasts from Patients with Bipolar Disorder. J Neuropsychopharmacol Mental Health 1: 103. DOI: 10.4172/2472-095X.1000103
Copyright: © 2015 Logotheti M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 14313
- [From(publication date): 1-2016 - Aug 29, 2025]
- Breakdown by view type
- HTML page views: 13177
- PDF downloads: 1136