Graft-Versus-Host-Disease after Living-Donor Liver Transplantation
Received: 03-Apr-2018 / Accepted Date: 17-Apr-2018 / Published Date: 24-Apr-2018 DOI: 10.4172/2161-069X.1000562
Abstract
Graft-versus-host-disease is a rare complication seen after solid organ transplantation with a high mortality rate. Graft-versus-host-disease presents with fever, rash, diarrhea and cytopenia that causes challenges in differential diagnose of cytomegalovirus infection and drug reaction. We present a case who transplanted from living-donor due to hepatocellular carcinoma and chronic hepatitis C infection. He was hospitalized with a pre-diagnosis of graftversus- host-disease, cytomegalovirus infection and drug reaction.
Keywords: Graft-versus-host disease; Living-donor liver transplantation; CMV infection
Introduction
Graft-versus-host-disease (GVHD) is an immune reaction that occurs due to recognition of the host’s cell surface antigens and stimulation of cell mediated cytotoxicity by donor T lymphocytes [1]. It is a rare complication of solid organ transplantation, though it occurs frequently after hematopoietic stem cell transplantation. GVHD after cadaveric liver transplantation is described firstly by Burdick at al. [2]. Risk factors for GVHD after cadaveric liver transplantation is described as recipient over 50 years old, young donor, age difference more than 20 years between donor and recipient, HLA class one mismatch and glucose intolerance. Some researches demonstrated that one-way HLA-matching is a risk factor for recipients of living-donor liver transplantation [3,4]. The incidence of GVHD after orthotopic liver transplantation is reported to be 0.1% by the United Network for Organ Sharing (UNOS) [5]. The mortality rate is more than 75% in published reports [6]. Most of the data comes from cadaveric liver transplantation whereas there exist only few case reports in the literature for living-donor liver transplantation related GVHD, and actual incidence is not known. Acute GVHD presents with fever, rash, diarrhea, and pancytopenia typically occur between 1st-8th week after transplantation [7]. Differential diagnosis of acute GVHD includes cytomegalovirus (CMV) infection and immunosuppressive drug reaction; those cannot be easily distinguished [6]. We want to present a case who was transplanted from living-donor and hospitalized in our clinic with pre-diagnosis of GVHD, CMV infection and drug reaction.
Case Report
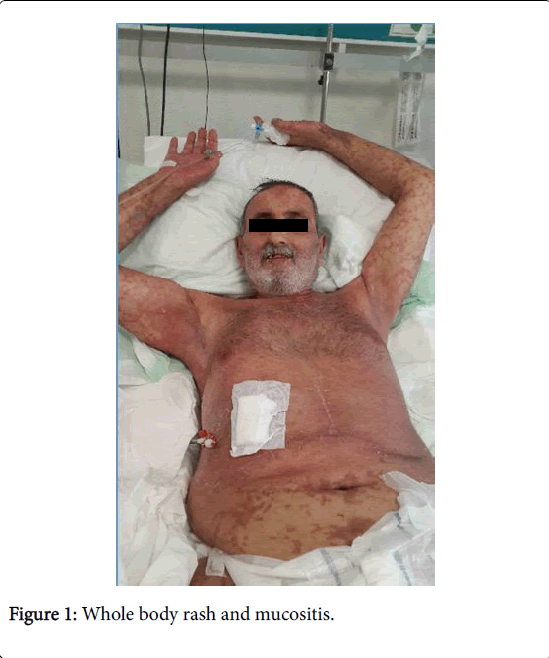
Written informed consent was taken from the patient. He was 56 years old male patient, diagnosed as hepatocellular carcinoma (HCC) and chronic hepatitis C infection. He was also type 2 diabetic, treated with oral anti-diabetics. His blood type was Rh negative-0 sub-group, and was transplanted from a living donor; his son. He was taken immunosuppressive therapy; tacrolimus, everolimus, methylprednisolone and prophylactic antibiotics, sulphomethaxasoltrimethoprim and acyclovir. He admitted to our hospital for fever on 26th day of transplantation. On the 2nd day of his admission, he developed erythematous rash starting from chest and neck, then spreading to the whole body (Figure 1). He had bloody-mucous diarrhea up to 8-10 times a day. The vital signs were normal, except tachycardia (127 beats/min). Transaminases were normal, alkaline phosphatase was 144 IU/L, gamma-glutamyl transferase was 270 IU/L, total bilirubin was 6.49 mg/dL, direct bilirubin was 5.80 mg/dL, creatinine was 1.53 mg/dL, urea was 102 mg/dL, white blood count (WBC) was 9550/μL, hb was 16.2 g/dL and peripheral platelet count was 123.000/mL, respectively.
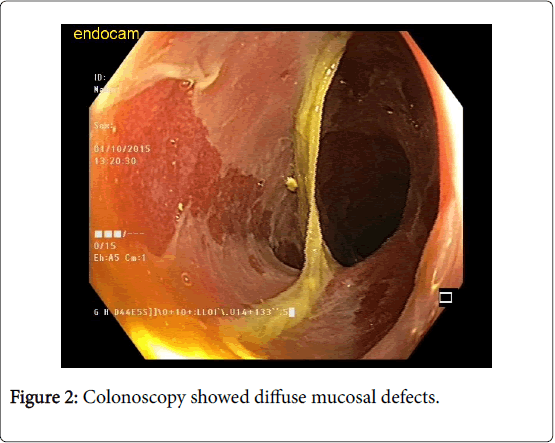
Immunosuppressive therapy and antibiotics were stopped. Colonoscopy showed diffuse mucosal defects (Figure 2). Skin biopsy and colonoscopic biopsies were taken immediately, and high dose intravenous corticosteroids were started. Histopathologic examination of skin biopsy showed compact parakeratosis on the surface, acanthosis on the epidermis; diffuse single cell necrosis and lymphocyte exocytosis. On the superficial dermis, rare perivascular melanophages and mixed inflammatory lymphocyte infiltration including eosinophils were seen. Significant apoptotic changes and necrobiotic debris were seen on foveolar and glandular epithelium in colon specimens.
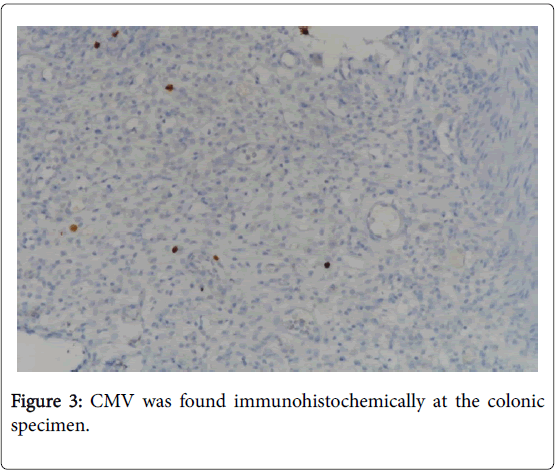
Apoptotic bodies and regenerative changes were found in the microscopic examination on the cryptic epithelium of cecum, transverse colon, and rectal samples. Mucosal denudation was present in the rectal biopsy. Pathological specimens were evaluated for GVHD. CMV was found immunohistochemically at the colonic specimen (Figure 3), and PCR result was positive for CMV-DNA on colonic tissue. On 33rd day of transplantation, he was diagnosed as septic shock; broad spectrum antibiotherapy was introduced empirically. There was no growth in the blood and stool cultures. He was deceased on the 34th day of transplantation due to septic shock and multiorgan failure which was 1 week after the onset of GVHD.
Discussion
GVHD is a rare complication with high mortality rates seen after orthotropic liver transplantation. Although most of the reported cases were seen after cadaveric transplantation, there were a few case reports about GVHD after living-donor liver transplantation. The incidence of GVHD after living-donor liver transplantation is unknown. In our center, we performed more than 1000 liver transplantations (more than 60% living-donor), and present case was the first one who experienced GVHD after living-donor liver transplantation. Mechanism of GVHD, seen after orthotropic liver transplantation has not been fully understood, but is thought to have emerged in a similar way to the immunological response of transfusion-associated GVHD. Our case was diagnosed as grade 3 GVHD according to International Bone Marrow Registiration (IBMTR) severity index (Tables 1 and 2).
| Stage | Skin Involvement (Maculopapular rash) | Liver involvement (Bilirubin level) | Gastrointestinal involvement (Stool volume) |
|---|---|---|---|
| 1 | 0 | Below 2.00 | 500 ml and less |
| 2 | 25% or less | 2.0-2.9 | More than 500 |
| 3 | 25-50% | 3.0-5.9 | More than 1000 |
| 4 | 50% or more | 6.0-14.9 | More than 1500 |
| 5 | Bullous changes | 15.0 or higher | More than 2000 |
Table 1: International Bone Marrow Registiration (IBMTR) grades acute GVHD as follows [8].
| Grade | Skin involvement | Liver involvement | GI involvement | Functional Loss |
|---|---|---|---|---|
| 0 | 0 | 0 | 0 | None |
| 1 | 01-02 | 0 | 0 | None |
| 2 | 3 | 1 | 1 | Mild |
| 3 | 4 | 02-03 | 02-04 | Moderate |
| 4 | 5 | 4 | 5 | Severe |
Table 2: International Bone Marrow Registiration (IBMTR) grades acute GVHD as follows [8].
Risk factors for GVHD seen after orthotropic liver transplantation were described by Akbulut S et al. which were the presence of close HLA matching between the recipient and donor, blood transfusion prior to transplantation, immunesuppressive treatment before transplantation, glucose intolerance, rejection before GVHD, autoimmune hepatitis, alcoholic liver disease, HCC, re-transplantation, a large age discrepancy between donor (younger) and recipient (older), recipient age >65 years, and multiorgan transplantation [9]. Murali et al. described risk factors for GVHD that were being recipient over 50 years old, young donor, age difference more than 20 years between donor and recipient, HLA class one mismatch and glucose intolerance [10]. Present case had most of the risk factors for GVHD. As in our case, there is higher GVHD risk in patients who had HCC-related transplant [10]. Risk factors for living-donor liver transplantation are not exactly known, however, transplantation from a donor with 1-way HLA match carries an extremely high risk of developing GVHD in living-donor liver transplantation [11]. GVHD is seen between 3 to 5 weeks after the transplantation. There occur erythematous and maculopapular rash that starts from any part of the body including palms, soles, volar faces of extremities and trunk. Rash may not be itchy, but it is recognized by the patient. Keratinocyte apoptosis and lymphocyte exocytosis in epidermis and vacuolar alteration in dermoepidermal junction are the characteristic features of histopathological skin examination [10]; these findings were seen in our patient's skin biopsy. GVHD affects all three hematopoietic lines. Alloreactive T lymphocytes engraft in the bone marrow of recipient and attack to hematopoietic cells by immune-mediated mechanisms. Cytopenia is usually found in the first few months after orthotropic liver transplantation, due to infections (Herpes virus, CMV, Epstein-Barr virus and parvovirus B19) and drugs (Mycophenolate mofetil, valganciclovir, trimethoprim-sulphomethaxosol). Cytopenia is known to be a poor prognostic factor in GVHD and leukopenia-related sepsis is reported as one of the reasons for mortality [10].
Gastrointestinal manifestations are seen frequently in GVHD. Diarrhea is the most common symptom seen after solid organ transplantation. 10-13% of patients have diarrhea in the first four months of transplantation. Clostridium difficile related infections, CMV colitis and drugs, mostly mycophenolate mofetil and mTor inhibitors, give rise to diarrhea. Superficial ulceration of the gastrointestinal mucosa, exudation and erythema are found in endoscopy, but sensitivity and specificity of the signs are very low; so histopathological correlation is needed. Rectosigmoid biopsy is the most sensitive method for differential diagnosis. Increased cryptic epithelial apoptosis, cryptic loss and neutrophil infiltration are described under light microscope [10], as same as our findings of histopathologic examination of colonic biopsy samples. Fever and rash occur early period of GVHD, which is similar to immunosuppressive drug reaction and CMV infection seen in immunocompromised patients. This similarity may cause misdiagnose. Skin biopsy which is not pathognomonic shows dyskeratosis, lymphocyte exocytosis, perivascular lymphocyte infiltration in dermis, apoptosis in epidermal rete pegs, satellite lymphocytes near to dyskeratotic epidermis [12,13]. Epithelial apoptosis is also found in CMV infection, but diagnosed by viral inclusions in immunohistochemistry [10]. CMV-DNA positivity in PCR supports the diagnosis. CMV infection and gastrointestinal involvement of GVHD is frequently seen together; and we diagnosed our case as coexistence of GVHD and superimposed CMV infection. This co-existency causes some difficulties in diagnosis and also in treatment [14]. Co-existence of GVHD and CMV colitis is known to have a negative influence on prognosis and survival [15]. On the other hand, immunological changes caused by GVHD adversely affect antiviral treatment for CMV. HLA incompatible donor transplantation is a risk factor for CMV infection by itself.
Conclusion
In conclusion, in patients receiving immunosuppressive treatment after solid organ transplantation, fever and rash development should be possibly linked with GVHD, but CMV infection, drug reactions should be remembered in the differential diagnosis. CMV coexistence with GVHD should also be kept in mind.
References
- Thin L, Macquillan G, Adams L, Garas G, Seow C, et al (2009) Acute graft-versus-host disease after liver transplant: Novel use of etanercept and the role of tumor necrosis factor alpha inhibitors. Liver Transpl 15: 421-426.
- Burdick JF, Vogelsang GB, Smith WJ, Farmer ER, Bias WB, et al (1988) Severe graft-versus-host disease in a liver-transplant recipient. N Eng J Med 318: 689-691.
- Whitington PF, Rubin CM, Alonso EM, McKeithan TW, Anastasi J, et al (1996)Â Complete lymphoid chimerism and chronic graft-versus-host disease in an infant recipient of a hepatic allograft from an HLA-homozygous parental living donor. Transplantation 62: 1516-1519.
- Kiuchi T, Harada H, Matsukawa H, Kasahara M, Inomata Y, et al (1998) One-way donor-recipient HLA-matching as a risk factor for graft-versus-host disease in living-related liver transplantation. Transpl Int 11: S383-384.
- Taylor AL, Gibbs P, Bradley JA (2004) Acute graft versus host disease following liver transplantation: the enemy within. Am J Transplant 4: 466-474.
- Smith DM, Agura E, Netto G, Collins R, Levy M, et al (2003) Liver transplant-associated graft-versus-host disease. Transplantation 75: 118-126.
- Post GR, Black JS, Cortes GY, Pollack RB, Wolff DJ, et al (2011) The utility of fluorescence in situ hybridization (FISH) analysis in diagnosing graft versus host disease following orthotopic liver transplant. Ann Clin Lab Sci 41: 188-192.
- Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, et al (1995) 1994 consensus conference on acute GVHD grading. Bone Marrow Transplant 15: 825-828.
- Akbulut S, Yilmaz M, Yilmaz S (2012) Graft-versus-host disease after liver transplantation: a comprehensive literature review. World J Gastroenterol 18: 5240-5248.
- Murali AR, Chandra S, Stewart Z, Blazar BR, Farooq U, et al (2016) Graft versus host disease after liver transplantation in adults: A case series, review of literature, and an approach to management. Transplantation 100: 2661-2670.
- Soejima Y, Shimada M, Suehiro T, Hiroshige S, Gondo H, et al (2004) Graft-versus-host disease following living donor liver transplantation. Liver Transpl 10: 460-464.
- Ferrara JL, Deeg HJ (1991) Graft-versus-host disease. N Engl J Med 324: 667-674.
- Dignan FL, Clark A, Amrolia P, Cornish J, Jackson G, et al (2012) Diagnosis and management of acute graft-versus-host disease. Br J Haematol 158: 30-45.
- Ljungman P, Hakki M, Boeckh M (2011) Cytomegalovirus in hematopoietic stem cell transplant recipients. Hematol Oncol Clin North Am 25: 151-169.
- Cho BS, Yahng SA, Kim JH, Yoon JH, Shin SH, et al (2013) Impact of cytomegalovirus gastrointestinal disease on the clinical outcomes in patients with gastrointestinal graft-versus-host disease in the era of preemptive therapy. Ann Hematol 92: 497-504.
Citation: Oytun MG, Danis N, Tekin F, Sezak M, Ozutemiz O, et al. (2018) Graft-Versus-Host-Disease after Living-Donor Liver Transplantation. J Gastrointest Dig Syst 8: 562. DOI: 10.4172/2161-069X.1000562
Copyright: © 2018 Tekin F. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 6530
- [From(publication date): 0-2018 - Nov 20, 2025]
- Breakdown by view type
- HTML page views: 5505
- PDF downloads: 1025