Invasive Ductal Breast Carcinoma in an 18-Year-Old Woman: A Casual Misfortune?
Received: 17-May-2019 / Accepted Date: 30-May-2019 / Published Date: 07-Jun-2019 DOI: 10.4172/2161-0681.1000368
Abstract
Malignant Breast Cancer is a life-threatening condition, with wide-world growing impact. The incidence of neoplastic malignant breast disease is showing a shifting paradigm, occurring overtime in younger ages. The authors report a case of an eighteen year-old Caucasian woman complaining with an induration in her left breast for seven months, diagnosed as invasive ductal carcinoma spread to axillary sentinel lymph node. There was no family history of cancer and the patient had no personal history of risk behaviour. She underwent a breast-conservative surgery and axillary lymph node dissection. After surgery she started chemotherapy, followed by radiation and hormonal therapy. She is observed regularly by Oncology, with no signs of secondary involvement.
Keywords: Breast cancer; Young women; Incidence; High-risk
Introduction
Technological improvements and the increasing degree of suspicion of breast cancer cases have resulted in higher detection of cases at an early stage, not only in women who usually start undergoing the preventive mammography scan after the 40 years of age [1] but also at younger ages, in which a neoplastic diagnostic is far from being the physician’s first suspicion. Apart from the subtypes of breast cancer and its intimate relationship with the feminine hormone levels and the expression of known genetic elements [2,3] there seems to be specific variances as far as the age of diagnosis is concerned, being the menopause an important mark. Young, pre-menopausal women area growing subgroup in the universe of patients diagnosed with breast cancer, not only due to the emergent awareness of physicians and patients themselves for cancer, but also because the diagnostic armoury allows for early diagnosis and intervention.
The purpose of this paper is to report a case of a young patient diagnosed with malignant breast cancer, the surgical and medical treatment undertaken, as well as the early outcome. Searching through Medline, we found 4 similar cases described, what reinforces the importance of reporting these cases to the medical community.
Case Report
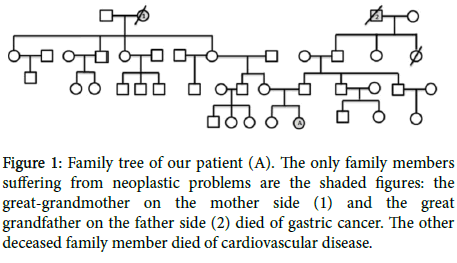
In April 2016, an 18-year-old Caucasian healthy woman was sent to a Senology consultation with complaints of a lump/induration in the left breast for the last 7 months, with no other symptoms associated, namely tiredness or weight loss. She had no personal or family history of cancer except for the great-grandmother on her mother side and the great grand-father on her father side that died of disseminated gastric cancer (Figure 1) Our patient had no children of her own, menarche at 13 years of age with regular menses, with no history of birth control pills, no smoking or drinking habits, as well as no risk behaviours worthy of mention.
Figure 1: Family tree of our patient (A). The only family members suffering from neoplastic problems are the shaded figures: the great-grandmother on the mother side (1) and the great grandfather on the father side (2) died of gastric cancer. The other deceased family member died of cardiovascular disease.
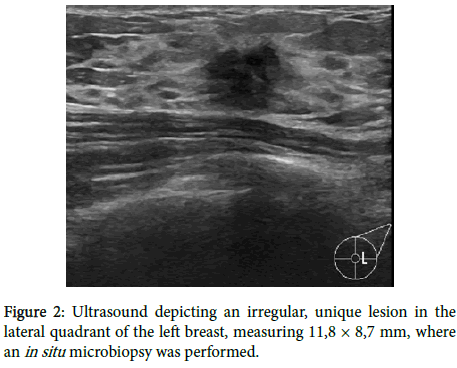
The lump was palpable in the upper outer quadrant of the left breast, circular in shape with irregular borders, painless and mobile, not adherent to deeper tissues, preserving breast symmetry, with no discomfort or discharge. There were no palpable axillary lymph nodes as well. The right breast examination was normal. Considering these findings, a left breast echography was promptly scheduled. The echography revealed an irregular, unique lesion in the lateral quadrant of the left breast, measuring 11,8 per 8,7 mm (Figure 2).
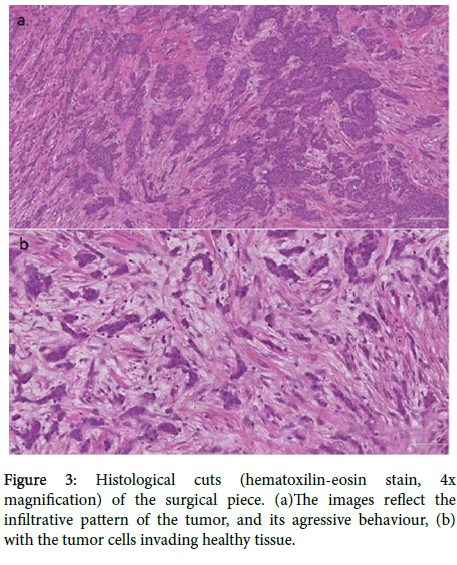
An in situ microbiopsy was performed and sent for histopathological analysis, revealing a grade 2 invasive ductal carcinoma, positive for estrogen (60%) and progesterone receptors (80%), CERB-b2 negative and a low Ki67 (<5%).The histological pattern is shown below (Figure 3) the evident infiltrative ductal pattern (3a), the highly aggressive behaviour of tumoral cell spreading (3b).
The patient was sent for early senology consultation to be informed of the histological diagnosis, as well as to discuss the therapeutic plan. She underwent a Thoraco-Abdominopelvic CT scan, which revealed no signs of secondary organ involvement, as well as a Bone Scintigraphy, which retrieved no evidence of lytic, secondary bone affection. The sentinel lymph node testing was positive, discussed in the multidisciplinary reunion group and a treatment plan elaborated according to the staging (pT1c N1a (sg+) Mx).
Confronted with the diagnostic of a regional, malignant ductal breast carcinoma, the patient was scheduled for a breast-conservative surgery, as well as sentinel lymph node removal and axillary lymph node dissection. The patient was admitted in the Surgery Department and the surgery was performed 45 days after the diagnosis, with no operatory or post-operatory complications. There was no local vascular invasion macroscopically observed and ten axillary lymph nodes were removed. She was dismissed 2 days after the surgical intervention.
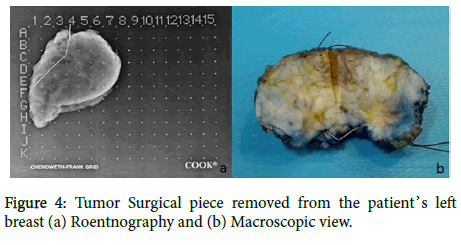
The surgical piece (Figures 4a and 4b) as well as the sentinel and the axillary lymph nodes removed were sent for histopathological analysis. The main tumor piece confirmed the diagnosis performed previously in the ultrasound-guided microbiopsy. There was no vascular invasion, free surgical margins, the sentinel lymph node was positive for metastatic involvement and all the ten samples of axillary lymph nodes didn’t show evidence of metastatic involvement.
A week later the patient returned to a Senology appointment for observation and further consultation by the medical oncologist of our center, where she was explained the treatment plan, according to the genetic testing, negative for the BRAC 1 and 2 mutation gene.
She started chemotheraphy with FEC (5-fluororacil, 770 mg IV; epirubicine 150 gm IV; cyclophosphamide 770 mg IV) and Docetaxel (taxotere 110 mg IV) 30 days after the surgery, with a febrile neutropenic episode (1,9 × 10^9/uL leucocytes, 200 neutrophils (absolute count)) 14 days after the first chemotherapy cycle, delaying the second cycle in 15 days and ending up admitted in the Internal Medicine Department for 5 days for IV antibiotic (piperacillintazobactam 4000 mg + 500 mg) and c-GSF treatment (Lenogastrim 263.0 M.U.I, SC). She recovered successfully, with normalization of leucocyte and neutrophil counts and discharged 5 days later. No pathogens were isolated in the blood and urine cultures harvested in the neutropenic episode.
She completed a total of 3 cycles of FEC, 3 Docetaxel cycles without relevant complications or side-effects. Then underwent concurrent fractioned radiotherapy during a month, a total dosis of 64 Gy, followed by hormonal therapy with Goserrelin (depot formulation 3.6 mg SC) and Tamoxifen 20 mg daily (self-administration).
The patient is currently completing a year of hormonal treatment, and observed regularly in the Medical Oncology consultation.
Discussion
Breast Cancer in young women, progressively diagnosed in early ages, is growing in a worrying fashion. Back in the 90’s, it was a disease mainly restricted to women above 40 year old of age. The advances in medical technology in the last decades, as well as the widespread use of precise diagnostic tools allowed a more prompt staging and hence, the possibility of undergoing conservative surgeries, sparing women from unnecessary mutilation. Only a small percentage (7%) of the cases of breast cancer occurred under the 40 years-old of age [4], presumably because the signs are usually ignored and assumed as benign masses or cysts, what had an important role in delaying the diagnostic.
Nowadays, the concept of breast cancer in ‘young women’ includes those diagnosed under the age of 35 [4] some authors consider they represent a distinct entity based on risk factors, clinical outcomes and tumor biology, which are somehow different from those found in women older than 40 years-old. In the pediatric population, the cases of primary breast cancer are even rarer, resumed to thirty nine cases published so far [5], where the incidence of breast cancer in women younger than 20 years is assumed to be of 1 in 1.000.000 [6,7] what clearly states its rarity. Also, these tumors tend to be more aggressive, with higher risk of local recurrence even after breast conservative surgery, which is associated inevitably with a worse prognosis.
In the case described, our patient noted the induration in her left breast for 7 months, only searching for medical observation after insistence from her family members. As expected, it was thought to be a benign mass, common in young women in her age range. Besides, the absence of risk factors and the poor history of cancer cases in the family raised no suspicion of malignancy.
The diagnostic of malignant ductal carcinoma was an unexpected finding, being so far the youngest patient in our center with the diagnosis of ductal malignant breast cancer. Considering our patient had recently turned eighteen years old, it is reasonable to admit that the breast malignant cancer had appeared when she was still an adolescent when the lump was first noted.
There are few similar cases described in the literature: the first one was described in Japan, in 1969 [8] there was also a case of an 18-yearold [9] with nipple discharge at presentation, resulting in an invasive lobular carcinoma that progressed despite treatment, with an OS of 4 years. There was also a case [10] of an 18-year-old African woman with a metastatic, unresectable metaplastic spindle cell breast carcinoma, with poor prognosis and an OS of 9 months.
All these cases were histologically different from the one of our patient and the outcome was very poor. Our patient still lives two years after the diagnosis, free of disease.
This case emphasizes the importance of the physician’s awareness when faced with similar cases in the daily practice. In our opinion, breast induration complaints in asymptomatic young patients must not be taken lightly, even with no risk factors and an innocuous family history. Besides, considering the harmless of an ultrasound exam, it is reasonable to ask for a breast ultrasound in young women with suspicious or doubtful mammary examination.
Always bearing in mind the rarity of these cases, our aim is merely to be preventive and cautious, hence diminishing the possibility of a neoplastic case go by unnoticed. All young women must be offered, as soon as possible, all therapeutic and viable options, considering as well the effect of long-term toxicity of anti-neoplastic therapies, the possibility of fertility impairment and premature menopause.
The therapeutic decisions made for our patient were carefully considered and the possible scenarios and complications were taken into account. In the Oncology consultation follow-up, two years after the cancer diagnosis, the patient remains asymptomatic, with no signs of intolerance to the hormonal therapy. We think our patient had potential to be candidate to extensive genetic testing, with a broader gene panel, as some worldwide centers already perform.
BRAC 1 and 2 gene mutation is the standard genetic exam available for patients suitable for testing, performed as well in the family members if considered eligible but unfortunately, only some centers have the means to perform extended genetic analysis involving specific exome panels. We would also like to emphasize the significance and importance of considering extended genetic testing besides the regular BRAC 1/2 mutation analysis in pre-menopausal patients with rare oncological breast diagnosis and a BRAC1/2 negative profile. There are mutations in genes described in the literature (CHEK2, PALB2, BRIP1, RAD50, CDH1) what seem to contribute considerably not only to the development of breast cancer in young ages, but also increase the susceptibility to develop gynecological cancer [11]. To date, these have been used for investigational purposes only, but have potential to become powerful tools if available as clinical screening tests, only in selected cases [12]. In nowadays medical practice, there are several genetic tests available: Mammaprint is the most innovative, evaluating the need of chemotherapy and the probability of early tumor recurrence. To date, these are only available in reference oncological centers. The ultimate goal would include a more personalized and efficient treatment plan, sparing young women from unnecessary treatment toxicity and adverse effects in early ages.
Conclusion
Screening is another dubious task in young women. According to the United States Preventive Services Task Force (USPSTF), screening in women prior to 50 year-old of age is subjective [13] and most definitely an individualized decision. The development of breast cancer in young ages is related to well-defined factors (early menarche, late menopause, cancer family history, use of oral contraceptive pills, exposure to radiation and increased body mass index), ethnicity included since Asian women tend to present it early in comparison to western women, where the self-breast examination gained more emphasis [14]. Nowadays, the mammographic density, obtained by mammography, still is a predictor of breast cancer risk in populations with increased risk and in the screening context, high density may justify further image investigation [15] which gives autonomy to the physician to extend the diagnostic work-up. The decision to whom extend that investigation remains unclear and, in the absence of welldefined risk factors, is based on the physician’s intuition. Explicit orientations are needed to guide physicians when confronted with these specific, rare cases.
Declarations
Acknowledgments
The authors would like to thank Dr. Maria Joao Hrotko, the imagiologist responsible for the microbiopsy of the lesion, ultrasoundguided, and to all the nursing personnel at the Outpatient Clinic of our Institution (Hospital José Joaquim Fernandes-ULSBA) that contributed to the care of our patient throughout the whole treatment process.
Authors’ contributions
Ana Luisa Oliveira and André Cabral Pacheco contributions in the elaboration of this article were equitative.
Ana Luisa Oliveira was responsible for the writing and bibliographic research in this case.
Andre Cabral Pacheco was responsible for writing support and performed the surgical procedure in the patient, as well as Dr. Jose Oliveira Catarino, that contributed with valuable insight concerning the surgical methodology.
Pedro Costa contributed actively in the revision of this article, as the physician responsible for the oncological treatment and follow-up of this patient.
Carlos Quintana contributed with important insight concerning the histological diagnostic, representative histological imaging and photos of the macroscopic lesion.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
References
- https://www.cdc.gov/cancer/breast/pdf/BreastCancerScreeningGuidelines.pdf.
- Chavez MacGregor M, Van Gils CH, Van der Schouw YT, Monninkhof E, Van Noord PA, et al. (2008) Lifetime cumulative number of menstrual cycles and serum sex hormone levels in postmenopausal women. Breast Cancer Res Treat 108: 101-12.
- Collaborative Group on Hormonal Factors in Breast Cancer (2012) Menarche, menopause and breast cancer risk: Individual participant meta-analysis, including 118, 964 women with breast cancer from 117 epidemiological studies. Lancet Oncol 13: 1141-1151.
- Anders C, Rebecca J, Litton J, Phillips M, Bleyer A (2009) Breast cancer before age 40 years. Semin Oncol 36: 237-249.
- Kennedy R, Boughey J (2013) Management of pediatric and adolescent breast masses. Sem in Plast Surg 27: 19-22.
- Simmons PS, Jayasinghe YL, Wold LE, Meldon LJ (2011) Breast carcinoma in young women. Obstet Gynecol 118: 529-536.
- Shannon C, Smith IE (2003) Breast cancer in adolescents and young women. Eur J Cancer 39: 2632-2642.
- Okita A, Yukiota T, Yamatsuji H, Fuse T, Sowa M. (1969) Case of breast cancer in an 18-year-old woman. Nihon Rinsho 27: 1290-1294.
- Jozwik M, Posmyk R, Semczuk A, Gogiel Shields M, Kus Slowinska M, et al. (2018) Breast cancer in an 18-year-old female: A fatal case report and literature review. Cancer Biol Ther 19: 543-548.
- Gomes A, Santos V, Dias G, Manso RT, Gonçalves L, et al. (2015) An 18-year-old woman with a 34 cm metaplastic breast carcinoma. J Surg Case Rep 2015: 1-7.
- Kuusisto KM, Bebel A, Vihinen M, Schleutker J, Sallinen SL (2011) Screening for BRCA1, BRCA2, CHEK2, PALB2, BRIP1, RAD50 and CDH1 mutations in high-risk finnish BRCA1/2-founder mutation-negative breast and/or ovarian cancer individuals. Breast Cancer Res 13: R20.
- Janatova M, Kleibl Z, Stribna J, Panczak A, Vesela K, et al. (2013) The PALB2 gene is a strong candidate for clinical testing in BRCA1- and BRCA2-negative hereditary breast cancer. Cancer Epidemiol Biomarkers Prev 22: 2323-2332.
- Siu AL (2016) Screening for breast cancer: US preventive services task force recommendation statement. Ann Intern Med 164: 279-296.
- Zahid AM, Noureen K, Munam S, Parsa AL, Mohammad ZF (2015) Risk of breast cancer among young women and importance of early screening in pakistan. Asian Pac J Cancer Prev 16: 7485-7489.
- Stephen WD, Oliver WE, Prue CA, Richard B, Maureen GC, et al. (2018) Mammographic density and breast cancer risk in breast screening assessment cases and women with a family history of breast cancer. Eur J Cancer 48: e56.
Citation: Oliveira AL, Pacheco AC, Catarino JO, Quintana C, Costa P (2019) Invasive Ductal Breast Carcinoma in an 18-Year-Old Woman: A Casual Misfortune? J Clin Exp Pathol 9: 368. DOI: 10.4172/2161-0681.1000368
Copyright: © 2019 Oliveira AL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 3504
- [From(publication date): 0-2019 - Dec 05, 2025]
- Breakdown by view type
- HTML page views: 2662
- PDF downloads: 842