Multifactorial Cognitive Training can Slow Down the Cognitive Decline in Early Alzheimer Patients
Received: 15-Jun-2019 / Accepted Date: 05-Jul-2019 / Published Date: 12-Jul-2019 DOI: 10.4172/2161-0460.1000470
Abstract
Introduction: Alzheimer’s disease (AD) is the most frequent dementia in the over 70-year-old people. Among the strategies examined to slow down the progression of AD, cognitive stimulation is underexplored. Yet, numerous studies showed that at early stage of AD, the brain is still able to show plasticity. Therefore, an adapted cognitive training can be considered as an efficient tool to fight the cognitive side of the pathology.
Objective: In the present study we investigate the potential of a memory and attention cognitive training to face the cognitive deficit observed in mild to moderate AD patients (n=12; MMSE: 22.1 ± 0.9).
Methods: We proposed a training program (15 sessions from 90 to 120 min every two weeks) using a large set of memory and attention tasks as well as various semantic tasks. Particular attention was given to the personalization of the difficulty level and content of the tasks as well as to the minimization of errors and negative experiences. Another important aspect of the training was the involvement of the caregiver in the training process during and between the sessions.
Results: Our results show a positive impact of our training program on one memory process, i.e., the recall, and one executive ability i.e., verbal fluency. The impact on recall highlight that a memory training using recognition as retrieval mode can help improving recall in AD patients in the early stage as we already showed it in aMCI patients. The impact on verbal fluency shows that training the manipulation of semantic material through many different tasks may improve access to the lexicon.
Conclusion: The present results bring new data showing that at early stage of the disease, an adapted cognitive training program has the potential to slow down the cognitive decline of the disease.
Keywords: Cognitive stimulation; Multifactorial cognitive training; Recognition memory; Computerized task; Training customization; Caregiver involvement; Memory; Attention
Introduction
According to the World Health Organization, dementia cases will represent 65.7 million people by 2030. Among those affected by dementia over 70 years of age, more than 60% will have Alzheimer’s disease (AD) [1,2]. Although the time course of neurodegeneration cannot be stopped, large efforts are provided to slow the cognitive decline of AD. Among the strategies examined, e.g. mainly pharmacological approaches but also noninvasive brain stimulation such as transcranial magnetic stimulation or physical activities, cognitive stimulation is underexplored. Numerous brain activity studies have shown that at early stage of AD, the brain is still able to show plasticity. Therefore, an adapted cognitive training can potentially be effective, and considered as an efficient tool to fight the cognitive side of the pathology. Among the studies investigating brain activity in AD patients, many of them reported a modification of brain activity in patients with early AD; e.g. increased activity in specific brain areas, when the patients are engaged in a cognitive task [3]. These new patterns of brain activation are interpreted as compensatory phenomenon. Some of these studies have gone further and have shown evidence of better cognitive performances related to the modification of brain activity. Indeed, in a study aiming at providing evidence for an effective compensatory role of the brain functional connectivity modification observed in patients with early AD, Grady et al. showed a correlation between the engagement of a network involving bilateral dorsolateral prefrontal and occipital cortices and better performances in semantic and episodic recognition tasks [4]. In another study by Stern et al., it has been shown that patients with early AD recruited a network of brain area during the difficult version of a verbal recognition task, whereas this network was not recruited in control subjects [5]. Results also showed that in the AD group, being able to perform a difficult version of the task, was associated with an increased activity in the left posterior temporal cortex, the calcarine cortex, the posterior cingulate, and the vermis.
To go even further, in a recent study, Barban et al. have investigated the impact of a cognitive training focusing on memory and executive functions, on the resting state functional connectivity in mild AD patients [6]. Results showed that the cognitive training have induced an improvement of attention abilities in parallel to functional connectivity modifications at rest. Especially, an increased connectivity was observed within the posterior Default Mode Network (DMN), a specific network observed during the resting state, when the active control period did not. Interestingly, and as highlighted by the authors, within the DMN, the regions showing a modified connectivity were memory related ones, i.e., the precuneus, the anterior cingulum and the medial temporal lobe. This latest study clearly shows that a cognitive intervention can be proposed to AD patients because their brain is still able to display plasticity mechanisms in line with improved cognitive performances.
As already mentioned earlier, studies investigating the impact of a cognitive support is underrepresented in the literature of AD therapy when compared to the field of amnesic Mild Cognitive Impairment (aMCI) therapy for example. Nevertheless, a recent review identified a total of 81 control trials fitting with the search criteria “studies that reported on intervention studies regarding cognition-oriented care approaches for dementia in older people diagnosed as having Alzheimer’s disease or at risk for Alzheimer’s disease” between 2009 and 2017 [7]. Among these studies, 25 concerned cognitive skills training interventions in dementia patients including AD patients and out of these, only few studied the impact of a “pure” cognitive intervention in a population of mild to moderate AD patients. One can cite the study of Fernandez-Calvo et al. investigating the impact of a multi-intervention program (48 sessions of 90 min, 3 sessions/week) on improving cognitive, functional, affective, and behavioral symptoms in people with mild AD (Clinical Dementia Rating 1 and Minimal Mental State Examination score>18) [8]. The sessions targeted performance of cognitive tasks, practice of daily problem-solving strategies, use of restorative strategies to address goals such as learning or re-learning of information, utilization of compensatory strategies, training in daily activities, and recreational activities. In addition, the program included activities to carry out at home, and psychoeducational sessions with both patients and informal caregivers to promote control and selfefficacy. AD patients with awareness of their deficits showed positive effects on all outcome measures in comparison with the control group, while AD patients with unawareness showed improvements in noncognitive symptoms only. In another study from Giovagnoli et al., the authors investigated the cognitive impact of a cognitive training (2 sessions/week for 12 weeks) in mild to moderate AD patients when compared to active music therapy, e.g., free use of melodic instruments, or neuroeducation, e.g., inform about brain anatomy and functions or how to cope with dementia [9]. During the cognitive training, visual (e.g., newspapers) and visuo-spatial (video clip without audio) material have been used, and at the end of each session, the patient was asked tailored questions about the information manipulated in order to create new association between internal and external stimuli. The cognitive training stimulated initiative, comprehension, attention, set shifting, information processing, executive functions, and memory. The authors have reported an improvement of initiative and no change of episodic memory in the cognitive training group when initiative was unchanged after both active music therapy and neuroeducation, and episodic memory declined after neuroeducation. Additional results from Huntley et al. investigating the impact of an adaptive structured digit span task (18 sessions, 2 sessions/week for 8 weeks) on working memory and larger memory abilities, reported improvement in verbal working memory, logical memory, and general cognitive performance assessed with the MMSE and ADAS-Cog scores, but no executive impact [10,11]. Again, the positive impact from these cognitive training programs, even though they are very different in contain, are arguments that show the greater interest that should be given to the study of the impact of a cognitive intervention in patients with early AD, when it is used as a tool to slow the progress of the disease.
Based on this literature, in the present study, we chose to investigate the impact of a multifactorial cognitive training proposed to mild to moderate AD patients. We were interested in proposing this cognitive training program to AD patients because it has already proven successful in a population of healthy elderly subjects but also in a population of aMCI patients [12]. Given the evident potential of this training program, we thus decided to adapt it to the mild to moderate AD population and to evaluate its impact on cognition.
Material And Methods
Participants and screening for eligibility
All participants have provided written, informed consent according to procedures approved by the local Ethics Committee, which followed the recommendations of the Declaration of Helsinki. In this study, we included 25 patients with AD. All patients were recruited from the Behavioral Neurology Department of Sainte-Marguerite Hospital, (Marseille, France). All patients underwent an MRI study and routine biological screening. None of the patients had visual complaints in daily living or showed complete Balint’s syndrome, or apperceptive or associative visual agnosia. No patients showed problems in understanding the instructions for assessments and training. None of the patients took any psychotropic medication during the time of the study. Probable AD was diagnosed according to criteria developed by the National Institute of Neurological and Communicative Disorders and Stroke (NINCDS) and the Alzheimer’s Disease and Related Disorders Association (ADRDA) [12,13]. Patients were evaluated with the Mini-Mental State Examination (MMSE) and the Clinical Dementia Rating (CDR) scale [14,15]. Exclusion criteria for the study were severe auditory, visual, verbal, or motor deficits, and recent participation in a cognitive training or neuropsychological research program.
AD patients were randomly assigned to one of the two experimental groups: one group participated to the memory and attention training (Training group, n=12), and one group did not participate in our training but did their usual care, most often memory stimulation sessions at a speech therapist or memory center (Control group, n=13). Table 1 shows the mean demographic and clinical characteristics of participants.
| Characteristics | Trained group (n=12) | Control group (n=13) |
|---|---|---|
| Age (years) | 81 ± 1.68 | 77.2 ± 2.28 |
| Women/Men (n) | 7/5 | 9/4 |
| Education (years) | 3.7 ± 0.4 | 4.1 ± 0.32 |
| MMSE (/30) | 22.1 ± 0.87 | 23 ± 0.95 |
| CDR (5) | 1.2 ± 0.11 | 1.0 ± 0 |
Data are expressed as mean ± SEM
Table 1: Baseline demographic data of participants.
Pre- and post-training assessments
All patients underwent standard neuropsychological assessment. The neuropsychological battery assessed memory, attention, executive skills and depression. It included the following tests: the Cognitive subscale of the Alzheimer’s Disease Assessment Scale (ADAS-Cog), the Trail-Making Test (TMT), the Frontal Assessment Battery (FAB), the phonemic verbal fluency of Cardebat, the semantic verbal fluency of the Isaac set test, the twelve words recall test from Signoret’s Memory Efficiency Battery, and the forward and the backward digit span test. Behavioral assessment included the 30-item version of the Geriatric Depression Scale (GDS) [16-21].
Training tasks
The training program used in the present study is partly a simplified version of Herrera et al. and Chambon et al. [11]. Computerized memory or attention tasks were programmed in Java (Release 1.4) and conducted on a Microsoft Windows-based computer with a 12- in. tactile screen and a resolution of 1.024 × 768 pixels. Each picture was 256 × 256 pixels in size. Responses to training tasks were given using a tactile screen, a standard keyboard (using only 2 keys), or a computer mouse. Pencil-paper cognitively stimulating activities were developed on a computer and presented to the patient with plastic sheets. These activities consisted in 7 tasks which stimulate semantic field, vocabulary, associations, understanding, etc.
Memory training tasks: For the memory training, we used tasks detailed in Herrera et al. [1]. The first one is the visual Recognition Memory task (v-RM) consisted in recognizing pictures leaned among equal number of distractors. Participants started the task with a number of pictures to memorize that was adjusted according to their performance evaluated during the pre-training session (session 1) where the patient was tested for his/her visual memory capacity by the span control task (mean visual span equal 9 ± 0.55 and mean auditory span equal 4.9 ± 0.5). The v-RM task contains two variants (V1 and V2 corresponding to two levels of difficulty). We also used the visuospatial Recognition Memory task (vs-RM) consisted in recognizing among two layouts, the target layout previously shown. The vs-RM included two variants (V1 and V2 corresponding to two levels of difficulty). We determined the percentage of correct responses.
Attention training tasks: For the memory training, we used tasks detailed in Herrera et al. and Chambon et al. [1,11]. The visual Focused Attention task (v-FA) consisted in detecting a target picture among distractors. This task included three variants (V1, V2 and V3 corresponding to three levels of difficulty). We determined the percentage of correct responses and the response time (s). The visuospatial Focused Attention Task (vs-FA) consisted in detecting a target picture among distractors (50% distractor, 50% target) on the right or the left side of the screen. We determined the percentage of correct responses and the response time (s).
Pencil-paper cognitively stimulating activities: The learning and memorizing stories task was proposed to the patient at each session. As soon as the story ended, questions were asked to the patient and he/she must answer an MCQ including possible answers. When the patient could not answer, the story was repeated sentence by sentence, working on understanding and memorization strategies (associations, mental imagery, etc.). We continued until the patient encoded the information. As the sessions progressed and the subject progressed in the memorization of the story, we introduced a mode of restitution in recall and the mode of restitution in recognition was no longer systematic but came in support of a failed recall. Thus, patients were alternatively assessed for recognition and recall. The matching task consisted in performing categorical or functional matching. Four pictures were presented to the patient. Among them, one target picture must be match with one of the other pictures based on their categorical or functional belonging. The looking from the target task: Words (between 10 and 20) were randomly presented on a sheet and the patient must find among them the target word as fast as possible. Another task consisted in choosing the numbers whose addition corresponded to the target total. Five to eight numbers were presented on a sheet and the patient had to determine what summed numbers are to obtain the target number. The Which picture depicts the sentence? task. In this task, three pictures were presented, and the patient had to choose the picture among the three which depicted the most a presented sentence. Another variant consisted to complete part of the sentence with the corresponding picture. The Find the same words task: Two columns of a series of 5 to 10 words or non-words are presented. Each column contains the same words but in a different random, and the patient must to link the same words. The Find pictures starting by a given letter task: Pictures are presented (between 6 and 12) and the patient had to choose the pictures starting by a given letter (vowels or consonants). For all the tasks, except for the learning and memorizing of stories task, the patient performed 10 trials and we measured the response time for each trial, the number of correct responses, errors, and how long the task lasts. The patient does not access the next trial until he/she produces a correct response. The level of difficulty is expressed by an increased similarity between target and distractors.
Procedure
Assessments and training took place at the Sainte-Marguerite Hospital (Marseille, France). Three neuropsychologists were involved in the study: One performed the screening for eligibility, one administered and scored assessments during the pre- and post-testing phases (this person was kept blind to the group membership of participants), and one supervised the training sessions.
Training group
Before training, each participant was familiarized with the computer material, and completed a session to establish the baselines to be used at the beginning of the training. Each participant took part in 15 individual training sessions (from 90 to 120 min every two weeks), a detailed presentation of the organization of the training is presented in Table 2. Training sessions were supervised by a neuropsychologist in the presence of the caregiver (most often spouse or child). Each session began with a welcome discussion with the patient and the caregiver where significant events from the patient’s or caregiver’s perspective were discussed. Then, the patient was trained and time was taken to discuss memory strategies that could be applied in daily demanding memory situations (e.g., store the objects in the same place, take notes, etc.). Positive feedback was constantly given to the patient. Whatever the task used for training, an unlimited amount of time was used to make the patient feel comfortable.
| Session | Computerized tasks | Paper pencil tasks | ||
|---|---|---|---|---|
| Memory | Attention | Story | Other tasks | |
| v-RM V1 | v-FA V1 | Yes | 1a & 3 | |
| vs-RM V1 | v-FA V1 | Yes | 1b & 3 | |
| v-RM V1 | v-FA V1 | Yes | 1a & 4 | |
| vs-RM V1 | vs-FA | Yes | 2 & 3 | |
| v-RM V1 | -- | Yes | 1b & 6 | |
| vs-RM V1 | -- | Yes | 1a & 3 | |
| -- | -- | Yes | 3 & 2 | |
| v-RM V2 | v-FA V2 | Yes | 4 & 6 | |
| -- | v-FA V2 | Yes | 5 & 3 | |
| vs-RM V2 | -- | Yes | 1b & 2 | |
| vs-RM V2 | -- | Yes | 4 & 3 | |
| -- | v-FA V2 | Yes | 6 & 2 | |
| -- | vs-FA | Yes | 3 & 4 | |
| -- | v-FA V3 | Yes | 6 & 3 | |
Note: v-RM=visual Recognition Memory task; vs-RM=visuospatial Recognition Memory task; v-FA=visual Focused Attention task; vs-FA=visuospatial Focused Attention Task; V=Variant; 1a=Categorical matching task; 1b=Functional matching task; 2=Looking from the target task; 3=Choosing the numbers whose addition corresponded to the target total; 4=Which picture depicts the sentence? 5=Find the same words; 6=Find pictures starting by a given letter. Training consisted of 15 sessions. The duration of a session was from 90 to 120 min but it could be up to 2 hours when more time was needed for explanations with both the patient and the caregiver. For the story, some patients were able to learn up to 5 stories while others could only learn two.
Table 2: Organization of the training.
Before starting using computerized memory or attention tasks, the neuropsychologist informed the patient about the Trained processes. The caregiver was also involved in this debrief because he/she was responsible for allowing the patient to work daily at home on the content of the current session. Examples of the task were shown and then the patient performed some trials in which no mistake was possible. Finally, the patient practices the task only when the neuropsychologist thought that he/she has understood what he/she needed to do. The difficulty consisted in the fact that the patient must do the task by making a minimum number of errors to have a positive reinforcement. Training of these tasks was individualized for each patient by the neuropsychologist who had the opportunity to increase or decrease the difficulty by modulating parameters such as the number of pictures or words to remember the choice of the target, or the addition of a distractor. As patients improved across sessions, these parameters were manipulated so that the tasks would continue to challenge their abilities but without putting them in distress because they failed. The training with the pencil-paper cognitively stimulating activities is based on the same philosophy.
Control group
Control participants were tested according to the same neuropsychological schedule as the training group to check for possible retest effects.
Statistical analysis
Data are expressed as the mean ± S.E.M. Statistical analyses were carried out using Statistical (Version 7.1, 2005) software. Unpaired t-test was used to compare baselines of subjects’ characteristics, including age and screening inclusion measures. The results from each evaluation and training task were analyzed with repeated measures ANOVA. Subsequent post hoc Duncan-corrected for multiple comparisons t-tests were used. The pertinent, paired t-test was used to highlight the impact of a parameter modification during the training (p significant at 0.05).
Results
Demographic and clinical characteristics
Unpaired t-test performed on each of the mean demographic and clinical characteristics of participants showed no difference between the Control and the Trained group.
Cognitive assessment
Results of the pre- and post-training cognitive assessments are presented in Table 3. A repeated measures ANOVA performed on the (B-A) score from the TMT test, the FAB score, the Isaacs set test total score, the reverse span, and the ADAS-cog score showed no group or time effect.
| Variables | Control | Trained | ||
|---|---|---|---|---|
| Positive impact | Pre | Post | Pre | Post |
| MMSE (/30) | 23.2 ± 0.8 | 21.9 ± 0.8* | 22.1 ± 0.9 | 23.2 ± 0.9 * |
| Cardebat letter P | 17.0 ± 2.3 | 13.1 ± 1.9* | 14.8 ± 1.8 | 17.4 ± 2.3 * |
| Cardebat letter R | 11.2 ± 2.1 | 10.1 ± 1.5 * | 13.1 ± 2.4 | 14.2 ± 1.9* |
| Direct span | 4.9 ± 0.2 | 4.4 ± 0.3* | 4.7 ± 0.3 | 5.4 ± 0.1 * |
| 12 words | 4.3 ± 0.5 | 3.6 ± 0.5$ | 4.5 ± 0.4 | 4.5 ± 0.4 * |
| No impact | Pre | Post | Pre | Post |
| Isaacs set test | 25.5 ± 2.4 | 23.0 ± 1.8 | 23.3 ± 1.6 | 27.8 ± 0.6 |
| Reverse span | 2.9 ± 0.2 | 3.2 ± 0.2 | 3.1 ± 0.2 | 3.4 ± 0.4 |
| TMT (B-A) | 171.6 ± 30.1 | 166.5 ± 26.2 | 153.0 ± 22.3 | 162.0 ± 28.7 |
| FAB (/30) | 12.8 ± 1.2 | 13.3 ± 0.7 | 12.8 ± 0.8 | 14.3 ± 0.8 |
| ADAS Cog (/70) | 13.9 ± 1.6 | 17.0 ± 2.5 | 13.0 ± 1.4 | 10.6 ± 0.6 |
Data are expressed as mean ± SEM. *Post-hoc t-test; $paired t-test in control
Table 3: Results of the cognitive evaluations performed before and after training.
A repeated measures ANOVA performed on the phonemic verbal fluency, letter P, showed a Group x Time interaction (F(1,11)=9.37, p<0.011). Subsequent post-hoc tests indicated that the performance of the Control group decreased between the two assessments whereas performances of the Trained group remained stable.
A repeated measures ANOVA performed on the phonemic verbal fluency, letter R, showed a Group x Time interaction (F(1,11)=5.32, p<0.041). Subsequent post-hoc tests indicated that the performance of the Control group remained stable between the two assessments whereas performances of the Trained group improved.
A repeated measures ANOVA performed on the mean of the two best free recalls of the twelve words of the Signoret’s Memory Efficiency Battery showed a Time effect (F(1,19)=5.53, p<0.03). Additional paired t-test performed between pre- and post-training assessments showed a decrease of performances in the Control group (t=4.27, p=0.003) but no difference in the Trained group.
A repeated measures ANOVA performed on the MMSE score revealed a Time effect (F(1,11)=6.31, p<0.029) and a Time x Group interaction (F(1,11)=22.36, p=0.0006). Subsequent post-hoc tests indicated that the performance of the Control group decreased between the two assessments whereas performances of the Trained group remained stable.
Training performances when using computerized training tasks
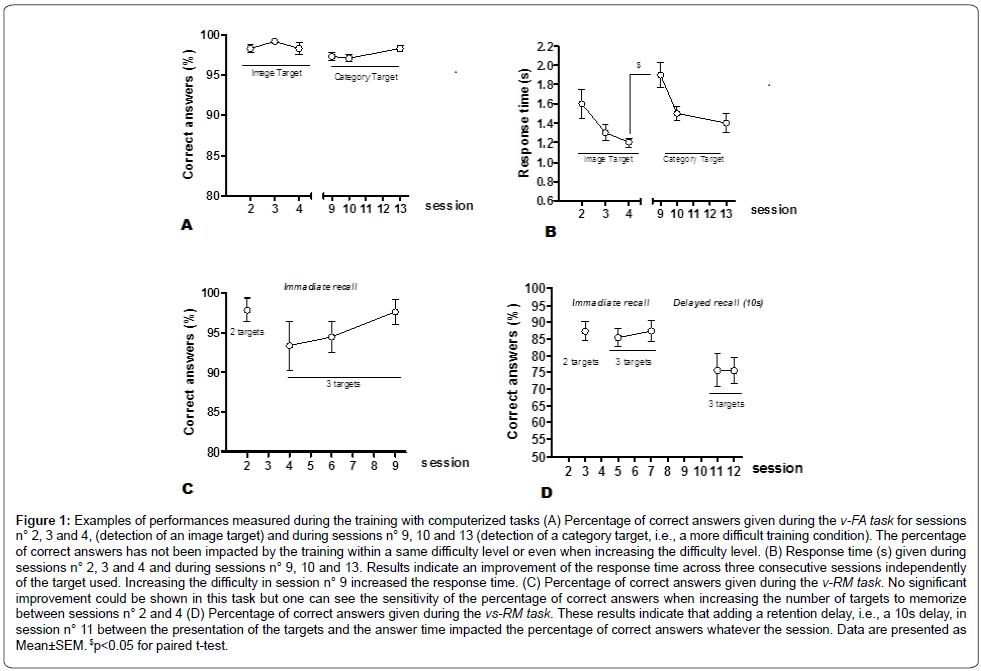
A repeated measures ANOVA performed on the percentage of correct answers in the v-FA training task (Figure 1A) showed a Target effect (F(1,11)=7.00, p=0.023) but no Session effect and no Interaction between the two factors. These results indicate that using a category target instead of a single image impacted the percentage of correct answers whatever the session.
Figure 1: Examples of performances measured during the training with computerized tasks (A) Percentage of correct answers given during the v-FA task for sessions n° 2, 3 and 4, (detection of an image target) and during sessions n° 9, 10 and 13 (detection of a category target, i.e., a more difficult training condition). The percentage of correct answers has not been impacted by the training within a same difficulty level or even when increasing the difficulty level. (B) Response time (s) given during sessions n° 2, 3 and 4 and during sessions n° 9, 10 and 13. Results indicate an improvement of the response time across three consecutive sessions independently of the target used. Increasing the difficulty in session n° 9 increased the response time. (C) Percentage of correct answers given during the v-RM task. No significant improvement could be shown in this task but one can see the sensitivity of the percentage of correct answers when increasing the number of targets to memorize between sessions n° 2 and 4 (D) Percentage of correct answers given during the vs-RM task. These results indicate that adding a retention delay, i.e., a 10s delay, in session n° 11 between the presentation of the targets and the answer time impacted the percentage of correct answers whatever the session. Data are presented as Mean±SEM. $p<0.05 for paired t-test.
A repeated measures ANOVA performed on the response time in the v-FA training task (Figure 1B) showed a Target effect (F(1,11)=6.66, p=0.025), a Session effect (F(2,22)=33.46, p<0.0001) but no Interaction. These results indicate that using a category target instead of a single image impacted the response time whatever the session, but also that the sequence of training sessions allowed to decrease this response time whatever the target used. A paired t-test performed between the response time of session n°4 and the response time of session n°9 indicated that using a category target, instead of a single image, significantly increased the response time.
A paired t-test performed on the percentage of correct answers in the v-RM training task (Figure 1C) showed no effect of the increased number of targets to memorize between sessions n°2 and 4. Repeated measures ANOVA performed on the percentage of correct answers showed no significant Session effect. But one can note that when increasing the number of target images from 2 to 3 the percentage of correct answers stays over 90% and even shows a tendency to improve between sessions n°4 and 9.
A paired t-test performed on the percentage of correct answers from sessions n°3 and 5 of the vs-RM training task (Figure 1D) showed no effect of the increased number of targets used from 2 to 3, i.e., performances stay over 85%. A repeated measures ANOVA performed on the percentage of correct answers showed a Delay effect (F(1,7)=5.55, p=0.05) but no Session effect and no Interaction. These results indicate that when adding a retention delay, i.e., a 10s delay, between the presentation of the targets and the answer, it impacts the percentage of correct answers whatever the session.
Training performances when using paper and pencil training tasks
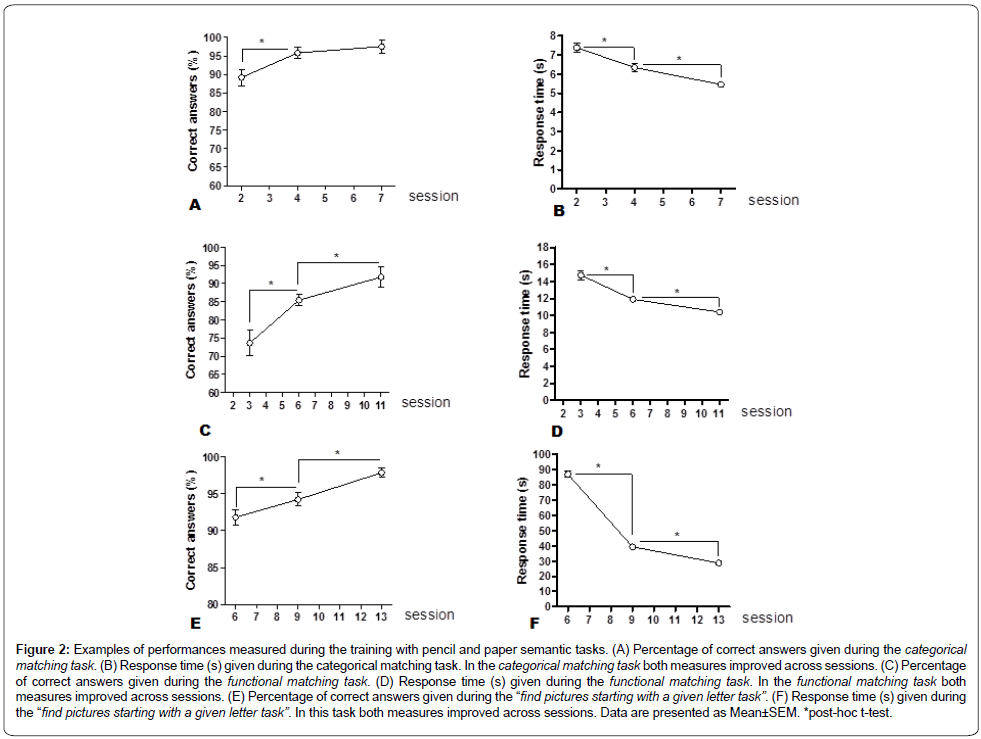
A repeated measures ANOVA performed on the percentage of correct answers in the categorical matching training task (Figure 2A) showed a Session effect (F(2,22)=7.7, p=0.003). Subsequent posthoc test showed an improved percentage of correct answers between sessions n°2 and 4.
Figure 2: Examples of performances measured during the training with pencil and paper semantic tasks. (A) Percentage of correct answers given during the categorical matching task. (B) Response time (s) given during the categorical matching task. In the categorical matching task both measures improved across sessions. (C) Percentage of correct answers given during the functional matching task. (D) Response time (s) given during the functional matching task. In the functional matching task both measures improved across sessions. (E) Percentage of correct answers given during the “find pictures starting with a given letter task”. (F) Response time (s) given during the “find pictures starting with a given letter task”. In this task both measures improved across sessions. Data are presented as Mean±SEM. *post-hoc t-test.
A repeated measures ANOVA performed on the response time in the categorical matching training task (Figure 2B) showed a Session effect (F(2,22)=28.65, p<0.0001). Subsequent post-hoc tests showed an improved response time between sessions n°2 and 4 as well as between sessions n°4 and 7.
A repeated measures ANOVA performed on the percentage of correct answers in the functional matching training task (Figure 2C) showed a Session effect (F(2,22)=28.29, p<0.0001). Subsequent posthoc tests showed an improved percentage of correct answers between sessions n°3 and 6 as well as between sessions n°6 and 11.
A repeated measures ANOVA performed on the response time in the functional matching training task (Figure 2D) showed a Session effect (F(2,22)=26.98, p<0.0001). Subsequent post-hoc tests showed an improved response time between sessions n°3 and 6 as well as between sessions n°6 and 11.
A repeated measures ANOVA performed on the percentage of correct answers in the find the 3 pictures starting by a given letter training task (Figure 2E) showed a Session effect (F(2,22)=15.22, p<0.0001). Subsequent post-hoc tests showed an improved percentage of correct answers between sessions n°6 and 9 as well as between sessions n°9 and 13.
A repeated measures ANOVA performed on the response time in the find the 3 pictures starting by a given letter training task (Figure 2F) showed a Session effect (F(2,22)=354.49, p<0.0001). Subsequent posthoc tests showed an improved response time between sessions n°6 and 9 as well as between sessions n°9 and 13.
Discussion
Our results indicate that the training program we proposed to AD patients had a positive impact on the MMSE score, the phonemic verbal fluency for letter P and R, the direct span and the 12-words test corresponding to a maintenance of performance when in the Control group performances declined between the two assessments sessions which were distant of 9 months. In other evaluation tasks, i.e., the TMT, the Isaacs set test, the reverse span, the FAB and the ADAS-Cog we could not show any significant impact of the training.
MMSE and ADAS-cog are comprehensive tests that assess a person’s cognitive functions and memory abilities. The results we obtained at the MMSE show a positive impact of our training in AD patients. Indeed, we could observe maintenance of the MMSE score in the Trained patients when this score decreased in Control ones. Performances in the ADAS-cog where very close to show a positive impact of the training (Time x Group interaction: F(1,11)=4.64, p=0.054) with a tendency to decrease performances in Control patients and increase performances in Trained patients. Interestingly, other studies have also shown benefits after a cognitive training at the same early stage of the disease in one or both of these two tests, e.g. Buschert et al. reported improvement in the ADAS-Cog but not in the MMSE whereas Huntley et al. showed improvement in both tests [10,22].
When looking into the details at the sub-scores of the MMSE and ADAS-cog we could observe that the global score of the Control patients was mainly impacted because of deficient recall performances in both tests and that this deficit has been reduced in Trained patients (data not shown). Such specific impact of our training on recall agrees with the preservation of performance observed for the 12-words recall test. Taken together these results highlight a clear impact of our training program on memory and in particular on recall. These results are in line with those we reported in a previous study investigating the benefits of a memory and attention training in a population of amnesic Mild Cognitive Impairment (aMCI) patients [1]. Indeed, in this study we identified that training aMCI patients to access the stored information using the recognition process had a positive impact on the untrained recall process thanks to recollection which is involved in both recall and recognition. The similarity between these results and the present results indicate that in early stage of AD such transfer of benefits is still possible. This reinforces the notion of the existence of brain plasticity in patients with early AD suggested by the work of some authors [4-6].
It is interesting to link the improvement in recall performance observed after training and the improvement in performance observed in computerized training tasks that use recognition as a means of retrieving stored information but also the ones that train attention. We believe that the training of both attention and recognition memory has facilitated the processing of information through a better focus, which in turn has facilitated memory encoding. In addition, the work on the coding of stories has encouraged the deployment of many strategies by patients. We also think that the way we used the restitution mode for the remembrance of stories played a crucial role in improving patient recall. Indeed, patients were alternatively assessed for recognition and recall. At the beginning of the training, the recognition mode prevailed. Then, as the sessions progressed, the mode of restitution in recognition was no longer systematic but came in support of a failed recall. As training progressed, the time intervals for recall and/or recognition became more numerous and longer and patients performed well due to a good encoding. Thus, we believe that both the encoding and restitution processes have been impacted by our training due to the combined use of both modes of restitution. On the other hand, we cannot determine in what proportion each process has been impacted.
Executive function is seen as a set of controlled cognitive processes that play the role of the central administrator of many different cognitive mechanisms, including memory and attentional capacities, and which is crucial for goal-directed cognitive activities [23,24]. Here we used the FAB to quantify global frontal abilities of AD patients, including executive function. This test revealed no effect of our training what may be due to a lack of specificity of the score which reflects cognitive functions but also non-executive aspects of frontal control such as imitation behavior or motor control.
Another aspect of executive function which was evaluated was verbal fluency abilities. We investigated both semantic and phonemic verbal fluency because they display a difference of already in nonpathological aging, i.e., semantic verbal fluency appears to be more sensitive than phonemic verbal fluency, and also because they rely on different cognitive processes, i.e., phonemic verbal fluency only relies on clustering when semantic verbal fluency relies on clustering and switching [25-27]. In the context of AD, it is thus not surprising that both types of verbal fluency are not equally impacted by the cognitive training. Indeed, our results show that our cognitive training positively impacted the phonemic verbal fluency in the trained AD patients when performances in the semantic verbal fluency assessed with the Isaacs set test did not show any significant impact, but only a tendency to improvement after the training period (results indicate a Time x Group interaction: F(1,11)=4.31, p=0.06) when performances of Control patients decreased. The positive impact of our training on phonemic verbal fluency and thus, based on literature, on clustering, can be related to some of the training exercises used. Indeed, among the various pencil-paper cognitively stimulating activities of the training, we proposed many tasks which aimed at working with semantic material. Among them, the categorical and functional matching tasks and the “find the three pictures which name is starting by a given letter” may have played the most important role in this positive impact. With these semantical tasks, patients were asked to provide effortful cognitive activity to “actively navigate” within a semantical field and to pull words together based on the given criteria. The repetition of such tasks may have facilitated the clustering capacity reflected by a better performance in phonemic verbal fluency tasks. Among the already cited cognitive training programs which can be near to our, none of them report benefits on verbal fluency but it must be notified that most of them used global test such as the MMSE or the ADAS-cog to assess benefits of their training.
In addition to this significant impact of the training, we would also like to point out the results of the TMT. In the control group, 5 patients were unable to perform the TMT-B in pre-assessment. In postassessment, one patient deteriorated and became unable to perform the TMT-B. This makes a total of 6 control patients who were unable to perform the TMT-B in post-evaluation. On the other hand, of the 5 trained patients who could not perform TMT-B at the pre-assessment, one was able to perform it in post-assessment. No statistically significant effects could be found between the control group and the trained group because the trained patient who successfully completed the TMT-B in post-evaluation took a very long time to complete it, considerably increasing the time of completion of the trained group, making it comparable to the control group. This could be added to our significant results to argue that training may have a larger effect on executive functions such as flexibility.
When comparing our training program with the few cognitive programs proposed to a similar AD population, one can notice that those which could report benefits mostly have provided session at a frequency of 2 or even 3 sessions per week, when we proposed one session every two weeks [8,9,10,22]. It is essential to take into account that we have given a major place to the caregiver, who seems to us to be a central element in the process of cognitive stimulation. Indeed, we think that the involvement and appropriation of cognitive stimulation by the caregiver is an essential element in the success of patient care. In our protocol, it was arranged with the caregiver that the key elements of the current stimulation session would be repeated daily with the patient. This is to strengthen through repetition the skills acquired thanks to the cognitive stimulation and to facilitate their transfer in everyday life situations. So, the results we have achieved are also because we have put the caregiver at the heart of our training program by involving him/ her in all the sessions, which allowed him/her to know the training exercises and thus to train the patient at home. Interestingly, we had a very good feedback from caregivers who found an improvement in patients’ moods and self-esteem as well as an improvement in many everyday life situations. The evaluation of the transfer of benefits to daily life was assessed qualitatively by the caregiver’s feedback. It will be necessary in the future to quantify it in order to better objectify it and to be able to intervene on it.
Conclusion
To conclude, our results bring evidences to promote the development of cognitive stimulation programs adapted to patients at the early phase of AD i.e., with adapted difficulty level and using a retrieval mode which is still functional, they can benefit from cognitive evaluation. We also highlight the importance of involving the people surrounding the patient, and particularly the caregiver, to help in transferring the benefits of stimulation to everyday life situations. However, it should also be noted that the ability of early AD patients to progress is limited. Indeed, when we wanted to introduce retention periods longer than 10 seconds, add a distractor to generate interference or a working memory task, the subjects’ performance collapsed. Considering the results obtained, which show a potential benefit of the stimulation on flexibility, we think it would be appropriated in the future to add to our training, tasks involving some processes that occur in working memory, such as updating, as well as tasks leading to resistance to interference in order to obtain larger benefits.
References
- World Health Organization Report (2012) Dementia: A public health priority. WHO Library Cataloguing-in-Publication Data. Publications of the World Health Organization.
- Pariente J, Cole S, Henson R, Clare L, Kennedy A, et al. (2005) Alzheimer's patients engage an alternative network during a memory task. Ann Neurol 58: 870-879.
- Grady CL, McIntosh AR, Beig S, Keightley ML, Burian H, et al. (2003) Evidence from functional neuroimaging of a compensatory prefrontal network in Alzheimer's disease. J Neurosci 23: 986-993.
- Stern Y, Moeller JR, Anderson KE, Luber B, Zubin NR, et al. (2000) Different brain networks mediate task performance in normal aging and AD: Defining compensation. Neurology 55: 1291-1297.
- Barban F, Mancini M, Cercignani M, Adriano F, Perri R, et al. (2017) A Pilot Study on Brain Plasticity of Functional Connectivity Modulated by Cognitive Training in Mild Alzheimer's Disease and Mild Cognitive Impairment. Brain Sci 7: 50.
- Carrion C, Folkvord F, Anastasiadou D, Aymerich M (2018) Cognitive Therapy for Dementia Patients: A Systematic Review. Dement Geriatr Cogn Disord 46: 1-26.
- Fernández-Calvo B, Contador I, Ramos F, Olazarán J, Mograbi DC, et al. (2015) Effect of unawareness on rehabilitation outcome in a randomized controlled trial of multicomponent intervention for patients with mild Alzheimer’s disease. Neuropsychol Rehabil 25: 448-477.
- Giovagnoli AR, Manfredi V, Parente A, Schifano L, Oliveri S, et al. (2017) Cognitive training in Alzheimer’s disease: A controlled randomized study. Neurol Sci 38: 1485-1493.
- Huntley JD, Hampshire A, Bor D, Owen A, Howard RJ (2017) Adaptive working memory strategy training in early Alzheimer’s disease: Randomized controlled trial. Br J Psychiatry 210: 61-66.
- Chambon C, Herrera C, Romaiguere P, Paban V, Alescio-Lautier B (2014) Benefits of computer-based memory and attention training in healthy older adults. Psychol Aging 29: 731-43.
- Herrera C, Chambon C, Michel BF, Paban V, Alescio-Lautier B (2012) Positive effects of computer-based cognitive training in adults with mild cognitive impairment. Neuropsychologia 50: 1871?1881. 44.
- McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM (1984) Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology. 34: 939-944.
- McKhann GM, Albert MS, Sperling RA (2012) Changing Diagnostic Concepts of Alzheimer's Disease. Alzheimer's Disease - Modernizing Concept, Biological Diagnosis and Therapy. Karger 28: 115?121.
- Folstein MF, Folstein SE, McHugh PR (1975). “Mini-mental state†: A practical method for grading the cognitive state of patients for the clinician. J Psy Res 12: 189-198.
- Hughes CP, Berg L, Danziger WL, Coben LA, & Martin RL (1982). A new clinical scale for the staging of dementia. Br J Psychiatry 140: 566-572.
- Mohs RC, Rosen WG, Davis KL (1983) The Alzheimer's disease assessment scale: An instrument for assessing treatment efficacy. Psychopharmacol Bull 19: 448-450.
- Reitan RM (1958).Validity of the trail making test as an indication of organic brain damage. Percept Mot Skills. 8: 271-278.
- Dubois B, Slachevsky A, Litvan I, Pillon B. The FAB: A frontal assessment battery at bedside. Neurology 25: 71-77.
- http://www.scirp.org/(S(i43dyn45teexjx455qlt3d2q))/reference/ReferencesPapers.aspx?ReferenceID=927851
- Isaacs B, Kennie AT (1973) The Set test as an aid to the detection of dementia in old people. Br J Psychiatry 123: 467-470.
- Yesavage JA, Brink TL, Rose TL, Lum O, Huang V, et al. (1982) Development and validation of a geriatric depression screening scale: A preliminary report. J Psy Res 17: 37-49.
- Buschert VC, Giegling I, Teipel SJ, Jolk S, Hampel H, et al. (2012) Long-term observation of a multi- component cognitive intervention in mild cognitive impairment. J Clin Psychiatry 73: e1492-e1498.
- Norman DA, Shallice T (1986) Attention to action: Willed and automatic control of behavior. In Consciousness and self-regulation: Advances in research. New York: Plenum Press, USA.
- Stuss DT (1992) Biological and psychological development of executive functions. Brain Cogn 20: 8-23.
- Mejia S, Pineda D, Alvarez LM, Ardila A (1998) Individual differences in memory and executive function abilities during normal aging. Int J Neurosci 95: 271-284.
- Bryan J, Luszcz MA (2000) Measurement of Executive Function: Considerations for Detecting Adult Age Differences. J Clin Exp Neuropsychol 22: 40-55.
- Troyer AK, Moscovitch M, Winocur G (1997) Clustering and switching as two components of verbal fluency: Evidence from younger and older healthy adults. Neuropsychology 11: 138-146.
Citation: Alescio-Lautier B, Sambucchi N, Michel BF, Chambon C (2019) Multifactorial Cognitive Training can Slow Down the Cognitive Decline in Early Alzheimer Patients J Alzheimers Dis Parkinsonism 9: 470. DOI: 10.4172/2161-0460.1000470
Copyright: © 2019 Alescio-Lautier B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 4836
- [From(publication date): 0-2019 - Dec 18, 2025]
- Breakdown by view type
- HTML page views: 3888
- PDF downloads: 948