Review Article Open Access
Oral Malignant Melanoma: A Brief Review
Nambiar S1*, Vishwanath MN2, Bhat S2, Farzana F2, Khwaja T2 and Alrani D21Department of Oral Pathology, SDM College of Dental Sciences, Karnataka, India
2Department of Dental Sciences, Rajiv Gandhi University of Health Sciences, Karnataka, India
- Corresponding Author:
- Swetha Nambiar
Department of Oral Pathology, SDM College of Dental Sciences, Sattur, Dharwad, Karnataka, 580009, India
Tel: +917259469783,0824-2481770
E-mail: drshwethas83@gmail.com
Received Date: August 25, 2016; Accepted Date: September 21, 2016; Published Date: September 23, 2016
Citation: Nambiar S, Vishwanath MN, Bhat S, Farzana F, Khwaja T, et al. (2016) Oral Malignant Melanoma: A Brief Review. J Clin Exp Pathol 6:294. doi: 10.4172/2161-0681.1000294
Copyright: © 2016 Nambiar S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Clinical & Experimental Pathology
Abstract
Malignant melanoma is an aggressive neoplasm affecting the oral cavity. It has the most biologically unpredictable behavior. The tumors can be locally invasive or metastasize to other parts of the body. Pre-existing pigmentations can evolve into malignant melanomas in about 30% of the cases. Hence, early detection of pigmented lesions of undetermined origin is important and should be routinely biopsied. Surgical management in still considered the first line of treatment of malignant melanoma. The deceiving asymptomatic presentation at early stages, rarity and poor prognosis emphasizes on need for highly specialized treatment. This review on oral malignant melanoma provides insight to a very aggressive neoplasm that can affect the oral cavity, it’s biological nature and behavior, early detection, diagnosis and insight into new treatment modalities.
Keywords
Keratinocyte; Melanoma; Melanocytes; Breslow’s tumour thickness; Excision
Introduction
Malignant melanoma is a rare aggressive neoplasm with melanocytes being the tissue of origin [1]. Melanomas usually arise in cutaneous areas, around 1-8% of malignant melanomas arise in the oral mucosa (accounting 0.5% of all oral malignant tumors) [2-5]. Most commonly affected areas in the oral cavity are hard palate and gingiva. Males are more commonly affected than females. The age range of reported cases ranges from 20-80 years [1,5]. Africans are most commonly affected due to presence of melanin pigmentation in oral mucosa of these people. In Asia, Japan has higher incidence of oral melanomas accounting 11-12.4% of all melanomas [5].
The prognosis (5 year survival rate) of oral malignant melanoma is poor, 0-20% whereas the overall survival for head and neck melanomas ranges between 20-48% according to literature review [6,7]. In the early stages, oral melanoma may be asymptomatic. Symptoms such as pain, bleeding and ulceration may present much later, hence most cases of oral melanoma (66.6%) get diagnosed only in advanced stages. The lesions greater than 4 cm in diameter, distant metastases have poor prognosis with mean survival rate less than 16.9 months, with only 6.6% of patients surviving more than five years. Mucosal melanomas frequently invade the underlying tissues and metastatize, have much poorer prognosis than cutaneous melanomas. In the head and neck area, conjunctiva, oral cavity and sinonasal tract are most commonly affected [2,8].
Etiology and Risk Factors
The etiological factors of malignant oral melanoma, unlike cutaneous melanomas are largely unknown. Several hypotheses include smoking, irritation caused by dentures and consumption of alcohol. Intraoral malignant melanomas arise from the few melanocytic cells present in the oral cavity that have the ability to undergo malignant transformation. They can arise de novo, from preexisting pigmented areas (5-30% cases) or from junctional nevus.
In more than two-thirds of the cases, p53 protein alterations have been identified. Loss of heterozygosity at 12p13 and loss of p27KIP1 protein have been detected. Cytogenetic analysis of melanocytespecific gene-1 (MSG-1) marker is helpful as it has a role in the pathogenesis [9,10].
Clinical Features
Tanaka et al., identified five clinical types in oral melanoma: pigmented nodular type, non-pigmented nodular type, pigmented macular type, pigmented mixed type, and non-pigmented mixed type. Oral melanoma may reach a vertical growth phase with/without a radial growth phase. Melanomas can appear as black, gray, purple or even reddish lesions. Amelanotic melanomas do not show any such colour changes (10% cases). Some lesions display uniformity in colour, whereas others exhibit variations in intensity of the colour.
It may present as multiple ulcerated or nodular lesions, be asymmetric and may present with irregular outline. Intraoral sites commonly involved by melanoma are hard palate (32%), followed by maxillary gingiva (16%), mandibular gingiva (7%), tongue (7%), buccal mucosa (7%), upper and lower lip (7%).
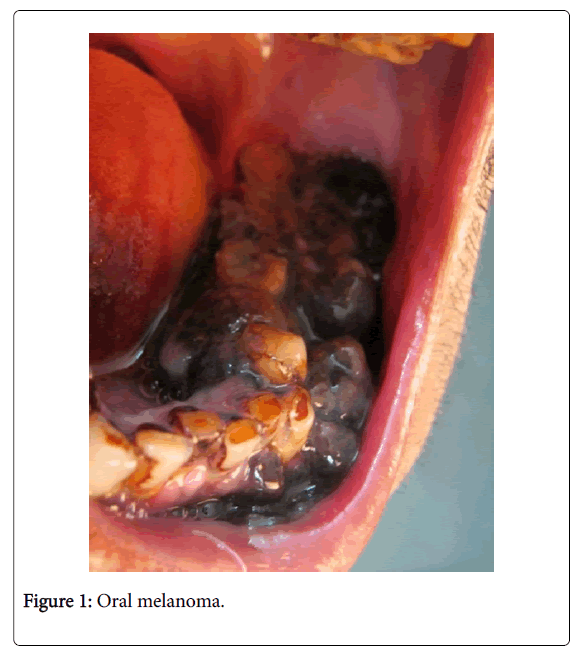
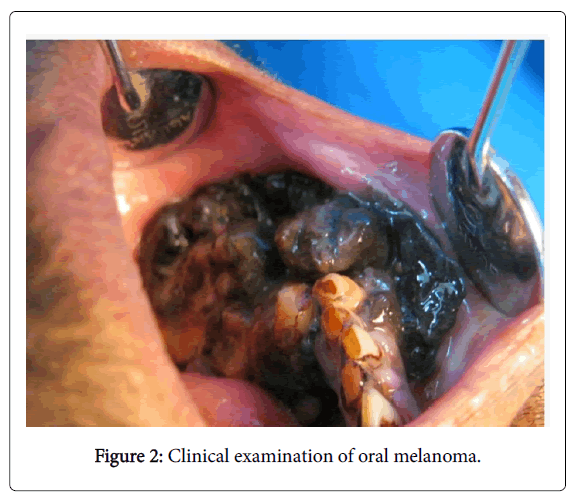
Tongue, parotid and tonsils are most commonly affected in secondary and metastatic melanomas (Figures 1 and 2) [11,12]. Umeda et al. noted that oral melanoma presents with 3 main components: centre with a nodular component, a plaque component that is flat or either slightly elevated and shows deep brownish-black pigmentation and a light brown non elevated macular component [13] (Figure 1).
Mucosal melanoma can be primary or metastatic. It is therefore very important to rule out any other primary malignant melanoma elsewhere in the body. Criteria to diagnose oral melanoma is given by Green et al: 1. Demonstration of clinical and histopathalogical presence of tumour in the oral mucosa. 2. Junctional activity should be seen in the oral mucosa. 3. Establish no other primary site of tumour. The initial radial phase can last for several years before it goes into the next stage of vertical growth where it invades the underlying structures. Induration is generally absent in cases with prolonged radial growth phase or with minimal invasion (Figure 2).
Some researchers felt that oral melanoma with radial growth phase has similarity to acral lentiginous melanoma of the skin. Currently, oral melanoma is classified as a separate entity from it’s cutaneous counterpart [14-19].
Protocol to Diagnose a Suspected Case of Oral Melanoma
Lesion should be photographed to record the size and the colour variations in the lesion. To check for metastasis to lungs, chest radiographs should be taken. CT scan and MRI should be done to evaluate the bone and soft tissue changes. To check for distant metastasis sentinel lymph node biopsy, lymphoscintigraphy, skeletal scintigraphic surveys and PET scan can be performed [17]. Proteomic analyses, melanin-inversion recovery imaging are new alternative that is giving promising results. Radioactively labeled hormone like α- melanocyte-stimulating hormone (MSH) may assist in detection of distant metastases.
FNAC can be carried out if the patient presents with cervical lymphadenitis to check for malignant cells. All oral pigmentations which show asymmetry, border irregularity, color variation, diameter of the lesion >6 m or evolving (ABCDE criteria) should be biopsied, as clinical diagnosis is often difficult, histopathological confirmation is essential [18].
Current studies suggest that an incisional biopsy/FNAC may lead to seeding of tumor cells into adjacent tissue or dissemination of tumor cells through bloodstream or lymphatics, increasing the rate of metastasis. Umeda et al, Rampen et al and Austin et al studied the 5- year survival rate of patients with oral melanoma who underwent surgical procedures like incisional biopsy, tooth extraction was poor (25.9%) compared with those who did not undergo such procedures before definitive surgery (91.7%). Hence, incisional biopsy as a diagnostic tool in oral melanoma has to be re-evaluated [20].
Diagnosis
Clinical staging (Table 1) and Breslow tumor thickness grading system (Table 2) are considered for assessment of the tumour and predicting prognosis [20].
| STAGE I | Level 1: Pure melanoma in situ- no invasion |
| Level 2: Invasion upto lamina propria | |
| Level 3: Invasion into muscle, bone or cartilage | |
| STAGE II | Metastasis to regional lymph nodes (T any N1M0) |
| STAGE III | Metastasis to distant organs (T any N any M0) |
Table 1: Clinical staging system with histologic microstaging for Oral Melanoma.
| Thickness (mm) | Risk of Recurrence |
|---|---|
| <0.76 | Low risk |
| 0.76-1.50 | Low to intermediate risk |
| 1.50-3.99 | Intermediate to high risk |
| >4.00 | High risk |
Table 2: Breslow scale to measure tumour thickness.
Breslow’s tumour thickness grading system measures the thickness of the tumor from the epidermal surface to the invasive front of the tumor. Thin melanomas that are less than 0.76 mm in thickness generally have an excellent prognosis. 1 mm is considered the international standard (cut off point) for thin melanomas beyond which the prognosis is not favourable [20,21] (Table 1).
As the tumor thickness increases, the metastatic potential of primary cutaneous melanoma increases. Most oral melanomas are diagnosed at later stages, hence at the time of initial presentation they are larger than 4 mm. Melanoma that shows metastatic spread to lymph nodes and other parts of the body show worse prognosis [6].
Recent introduction of markers like TA90 immune complex (TA90IC) and MIA protein predict the survival in patients with stage III disease. VEGF, VEGF receptor (VEGFR)1, TGF-β1 and Bcl-2 correlates with advancing stages in melanoma. Fluorescence in situ hybridization (FISH) is also recently used to analyse the genetic markers of oral melanoma [18] (Table 2).
Histopathological Features
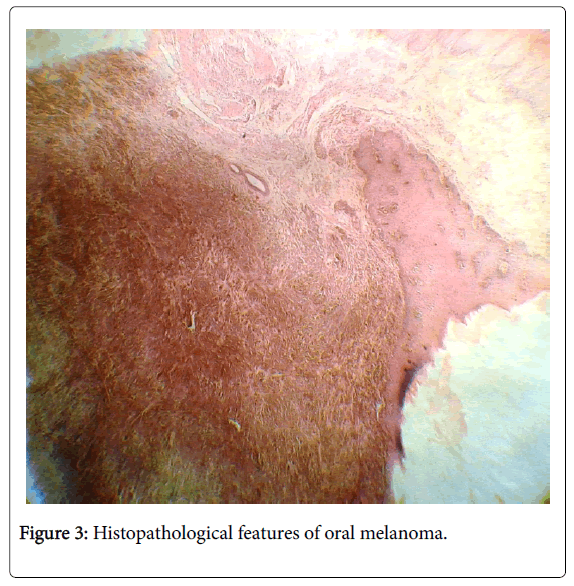
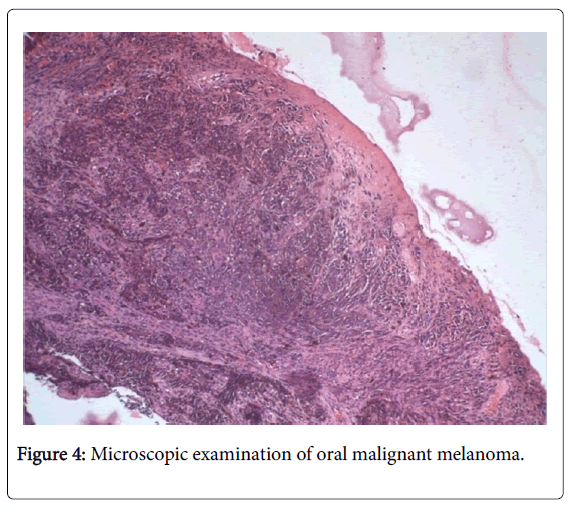
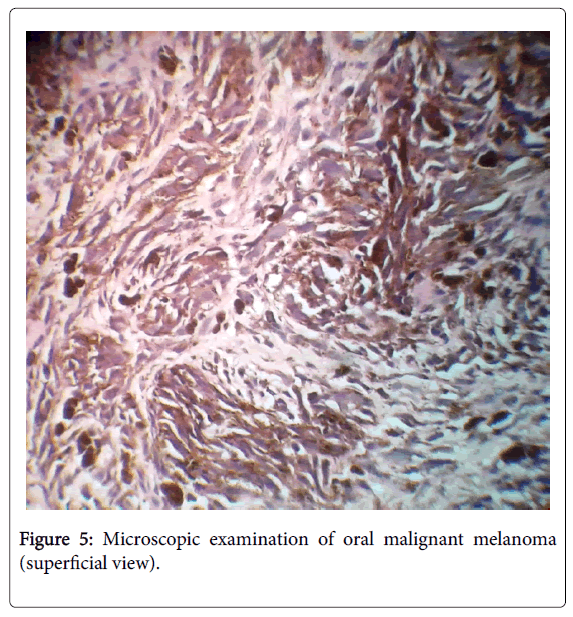
Routine hematoxylin and eosin staining is primarily used to diagnose a case of oral melanoma. The characteristic features of identification are through it’s junctional activity, diffuse arrangement of spherical/irregular shaped cells, cellular atypia with abundant mitotic figures, eosinophilic cytoplasm and nucleolus. Atypical melanocytes, cells that are larger, pleomorphic and hyperchromatic may be seen in the epithelial-connective tissue junction. High density of these atypical melanocytes are indicative of malignant melanoma. (Figures 3-5) In case of amelanotic melanoma where the lesion is completely devoid of any pigmentation, immunohistochemical analysis should be carried out using protein markers like S-100, gp-100, HMB-45, MITF and MART-1 to achieve a definitive diagnosis. Amelonotic melanoma cannot be identified histopathologically, immunohistochemical analysis is essential [8,21] (Figure 3).
Differential Diagnosis
A thorough clinical examination, histopathological evaluation with developing a differential diagnosis is necessary to arrive at a definite diagnosis. All the pigmented lesions that can occur in oral cavity should be considered, special stains and immunohistochemistry techniques should be employed to differentiate from other lesions.
Oral pigmentations can occur either due to physiological or pathological causes. It can be exogenous or endogenous in origin. Thorough clinical assessment of the lesion in terms of color, location, distribution, duration and evolvement should be done. A detailed medical history regarding the use of drugs, family history of malignancies and change in lifestyle should be recorded. Differential diagnosis includes physiologic pigmentation, post inflammatory pigmentation, acquired melanotic nevus, amalgam tattoo, blue nevi, melanosis associated with smoking and medication, melanoplakia, pigmentations in Cushing’s syndrome, melanoacanthoma, Spitz nevi, Addison’s disease, Peutz-Jeghers syndrome, Kaposi’s sarcoma, pigmentation caused due to use of heavy metals [20,21] (Figure 4).
Treatment Modalities
The following protocol should be followed in treating malignant melanoma once t is diagnosed:
1. Primary lesion should be surgically excised with a margin involving atleast 1-2 cm of healthy tissue, tumour extent and thickness should be considered.
2. Sentinal lymph nodes and other lymph nodes involved by metastases should be removed.
3. Radiochemotherapy should be considered post-surgery [1,20].
Surgical removal is the main mode of treatment, but because of anatomic restraints it is often difficult. In patients with early melanoma or in melanoma in situ, radiotherapy has given good results although melanoma is not very radiosensitive. Chemotherapy though under consideration has not given promising results [21].
Immunotherapy has been considered in recent years. Dacarbazine- DTIC and INF-alpha-2b are chemo-immunotherapical treatments which are considered, where different combinations of BCG vaccine and recombinant interleukin-2 (rIL-2) are used. Tumor-infiltrating lymphocytes, Bacillus Calmette- Guerin, irradiated allogeneic tumor cells and monoclonal antibodies may be incorporated into melanoma treatment. Many experimental studies are carried out on mice in vitro to test the efficacy of hollow nanoparticles made of gold on melanoma. It was seen that the gold particles aggregated to peptides and these have an affinity for the melanoma cell receptors and penetrated these cells. After this the mice were injected with infrared light bath, it was seen that the cancer cells that contained the nanoparticles were destroyed. The destruction took place due to three factors: photothermal damage, cell membrane loss and denaturation of proteins. This treatment using gold nanoparticles is known as plasmonic photothermal therapy [18,22-24]. Gene therapy and tumour cell vaccines are considered for further research with modified melanoma cells that contain specific genes that code for cytokines, interferon and growth factors [8,21] circulating melanoma cells are markers of a high relapse risk and shorter disease-free survival whether detected postoperatively or during follow-up [25,26] Tyrosinase mRNA amplification by RT-PCR (reverse transcriptasepolymerase chain reaction) have been used recently for monitoring the efficacy of adjuvant treatment in stage IIB/III oral melanoma patients. Lu-labeled DOTA, MCR-1, CRH-1 are positive regulators of melanogenesis and are potential targets for future research [14,15,22] (Figure 5).
Prognosis
The prognosis of a patient with oral melanoma is very poor with average survival rate of about 18 months from the time of diagnosis. Survival rate is less than 20-22% at the end of five years. Presence of systemic diseases, high clinical stage at presentation, Breslow’s tumor thickness of level 4 and 5, vascular invasion, histopathological variables, cases of amelanotic melanoma, and nodal as well as distant metastases are the factors that play a major role in predicting the prognosis and survival of patients with melanoma [21].
Conclusion
Oral malignant melanoma, if detected and diagnosed early, complete remission is possible. The incidence is very low and usually presents at an advanced stage. This is different in behavior from it’s cutaneous counterpart and no specific mode of treatment is considered ideal based on current available data. Recent advances in molecular research and an increasing understanding of tumour immunology is leading to new innovative immunotherapies that promises to get an effective treatment for oral malignant melanomas in the near future.
References
- Meleti M, Leemans CR, Mooi WJ, Vescovi P, van der Waal I (2007) Oral malignant melanoma: a review of the literature. Oral Oncol 43: 116-121.
- Gupta S, Tandon A, Ram H, Gupta OP (2015) Oral malignant melanoma: Report of three cases with literature review. Natl J MaxillofacSurg 6: 103-109.
- Hicks MJ, Flaitz CM (2000) Oral mucosal melanoma: epidemiology and pathobiology. Oral Oncol 36: 152-169.
- Gu GM, Epstein JB, Morton TH (2003) Intraoral melanoma: long-term follow-up and implication for dental clinicians. A case report and literature review. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 96: 404-413.
- Bhullar RP, Bhullar A, Vanaki SS, Puranik RS, Sudhakara M, et al. (2012) Primary melanoma of oral mucosa: A case report and review of literature. Dent Res J (Isfahan) 9: 353-356.
- Mihajlovic M,Vlajkovic S, Jovanovic P, Stefanovic V (2012) Primary mucosal melanomas: a comprehensive review. Int J ClinExpPathol 5: 739-753.
- Sharma N (2012) Primary oral malignant melanoma: two case reports and review of literature. Case Rep Dent 2012: 975358.
- Moreira RN, Santos CR, Lima NL, Verli FD, Marinho SA (2010) Oral and cutaneous melanoma: similarities and differences. J Clin Med Res 2: 155-158.
- Aguas SC, Quarracino MC, Lence AN, Lanfranchi-Tizeira HE (2009) Primary melanoma of the oral cavity: ten cases and review of 177 cases from literature. Med Oral Patol Oral Cir Bucal 14: E265-271.
- Femiano F, Lanza A, Buonaiuto C, Gombos F, Di Spirito F, et al. (2008) Oral malignant melanoma: a review of the literature. J Oral Pathol Med 37: 383-388.
- Gondak RO, da Silva-Jorge R, Jorge J, Lopes MA, Vargas PA (2012) Oral pigmented lesions: Clinicopathologic features and review of the literature. Med Oral Patol Oral Cir Bucal 17: e919-924.
- Munde A, Juvekar MV, Karle RR, Wankhede P (2014) Malignant melanoma of the oral cavity: Report of two cases. ContempClin Dent 5: 227-230.
- Umeda M, Komatsubara H, Shigeta T, Ojima Y, Minamikawa T, et al. (2008) Treatment and prognosis of malignant melanoma of the oral cavity: preoperative surgical procedure increases risk of distant metastasis. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 106: 51-57.
- Kumar A, Bindal R, Shetty DC, Singh HP(2012) Primary oral malignant melanoma: Clinicopathological series of four cases. Dent Res J 2012; 9: 338-344.
- Hashemi Pour MS (2008) Malignant melanoma of the oral cavity: a review of literature. Indian J Dent Res 19: 47-51.
- Manigandan T, Sagar GV, Amudhan A, Hemalatha VT, Babu NA (2014) Oral malignant melanoma: A case report with review of literature. ContempClin Dent 5: 415-418.
- Rani GS, Kumar TV, Kolasani B, Begum MR, PriyaSrinivasan A (2014) Primary malignant melanoma of maxilla: report of a case with discussion. Case Rep Dent 2014: 624306.
- Zbytek B, Carlson JA, Granese J, Ross J, Mihm MC, et al. (2008) Current concepts of metastasis in melanoma. Expert Rev Dermatol 3: 569-585.
- Kaul S, Kumar P (2013) Significance of early detection of oral malignant melanoma in improving prognosis. South Asian J Cancer 2: 199.
- Mohan M, Sukhadia VY, Pai D, Bhat S (2013) Oral malignant melanoma: systematic review of literature and report of two cases. Oral Surg Oral Med Oral Pathol Oral Radiol 116: e247-254.
- Rajendran R, Sivapathasundharam B, editors. Shafer’s textbook of Oral Pathology. 6th ed. Noida (India): Elsevier; 2009. Pg no: 120-126.
- Meleti M, Leemans CR, Mooi WJ, van der Waal I (2007) Oral malignant melanoma: the amsterdam experience. J Oral MaxillofacSurg 65: 2181-2186.
- Gorsky M, Epstein JB (1998) Melanoma arising from the mucosal surfaces of the head and neck. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 86: 715-719.
- Andreadis D, Epivatianos A, Poulopoulos A, Nomikos A, Papazoglou G, et al. (2006) Detection of C-KIT (CD117) molecule in benign and malignant salivary gland tumours. Oral Oncol 42: 57-65.
- Uchiyama Y, Murakami S, Kawai T, Ishida T, Fuchihata H (1998) Primary malignant melanoma in the oral mucosal membrane with metastasis in the cervical lymph node: MR appearance. AJNR Am J Neuroradiol 19: 954-955.
- Manganaro AM, Hammond HL, Dalton MJ, Williams TP (1995) Oral melanoma: case reports and review of the literature. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 80: 670-676.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 23668
- [From(publication date):
October-2016 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 22392
- PDF downloads : 1276