Research Article Open Access
Oxidative Stress: A Comparison of Different Groups of Patients with Hepatitis-C in Hemodialysis Units
| Da Silva NMO1*, Kalb AC2, Vidales-Braz BM2, Dos Santo DM2, Zanella RC3, Martinez EP2 and Martinez AMB2 | |
| 1Institute Educar Brazil, Brazil | |
| 2Universidad Federal of Rio Grande, Brazil | |
| 3Hospital Pompéia, Brazil | |
| *Corresponding Author : | Da Silva NMO Institute Educar Brasil, Faculdade Dom Bosco, Brazil Tel: +55 53 3026-2099 E-mail: nayleoliveira@gmail.com |
| Received November 02, 2015; Accepted December 03, 2015; Published December 07, 2015 | |
| Citation: Da Silva NMO, Kalb AC, Vidales-Braz BM, Dos Santos DM, Zanella RC Martinez EP,Martinez AMB, et al. (2015) Oxidative Stress: A Comparison of Different Groups of Patients with Hepatitis-C in Hemodialysis Units. Kidney Disord Clin Pract 1: 104. doi:10.4172/kdc.1000104 | |
| Copyright: © 2015 Da Silva NMO, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at
Abstract
There is a close relationship between patients undergoing hemodialysis (HD) treatment who are infected with hepatitis C virus (HCV) and oxidative stress (OS), which accelerates atherosclerotic processes and reduces the quality of life of the patients. Hemodialyzed patients have high morbidity and mortality. This study evaluated OS in patients undergoing HD against HCV infection. Four groups of patients (n=40 per group) were evaluated, including healthy patients (Group 1), patients on HD (Group 2), patients with HCV (Group 3) and patients with HCV on HD (Group 4). The groups were compared for the total antioxidant capacity (TAC) and the lipid peroxidation. The TAC decreased in the group 2 patients. In the analysis of the lipid peroxidation in erythrocytes, a significant increase (p<0.05) of the concentration of malondialdehyde (MDA) was observed in group 2 when compared to the group 3 patients. There was no significant difference (p>0.05) between the patients in groups 1, 3 and 4 and between group 2 when compared to group 4. The lipid peroxidation in the blood plasma demonstrates a significant association (p<0.05) in group 4. These results indicate that the HD process and HCV infection alter the peroxidation process by accelerating the process of atherosclerosis and reducing the quality of life of these patients.
| Keywords |
| Hepatitis C; Erythrocytes; Hemodialysis; Atherosclerosis |
| Introduction |
| The prevalence of hepatitis C (HCV) virus is a public health problem, and there is a high incidence of HCV in hemodialysis (HD) patients, which affects the quality of life, life expectancy and rejection after transplantation [1,2]. Apart from the exposure to viral agents, the patients in replacement therapy units are in a constant oxidative stress process (OS) that is due to the biocompatibility of the dialyzer membrane, an imbalance of the oxidant and antioxidant systems and the effects of the dialysate components [3,4]. |
| Even with the advanced technologies associated with renal replacement therapy, morbi-mortality remains high. A decrease in lipid peroxidation accompanied by abnormal activity of the antioxidant system becomes a contributing factor in many diseases, particularly in atherosclerosis that is commonly associated with the patients in question [5]. It is worth highlighting that HD patients have important biochemical disorders. |
| There is evidence that the re-use of hemodialyzer membranes results in decreased superoxide and a subsequent increase of antioxidant activity. By contrast, it has been reported that the re-use of dialyzers may increase the oxidative response of leukocytes [6]. |
| The HCV viral structures, such as the core protein and the nonstructural protein 3 (NS3) seem to be sufficient to produce mitochondrial alterations and generate OS [7]. Waris et al. emphasizes the role of OS in patients infected with HCV and the possible relationship with the injury process of hepatocytes [8]. Patients with chronic hepatitis C (CHC) have been shown to have a decreased production of reactive oxygen species (ROS) in the liver, which are responsible for the oxidation of intracellular molecules and the activation of transcription factors [8,9]. |
| The OS in patients with HCV in HD units is widely studied with controversial results. Some authors, such as Nascimento et al., reported increased OS in infected patients, whereas others reported decreased OS [10,11]. Based on these results, the present study investigated a possible relationship of OS in patients of HD units and with HCV infection. |
| Materials and Methods |
| One hundred and sixty patients were evaluated and divided into 4 groups, each group with 40 patients. The groups consisted of healthy patients (Group 1), patients on HD (Group 2), patients with HCV (Group 3) and patients with HCV on HD (Group 4). The inclusion criteria were an age>18 years and the participation in a dialysis program for at least three months. The study was approved by the Ethics Committee with the approval number 08981712.1.0000.5324/822124. All of the patients signed an informed consent form. The HD patients were submitted to dialysis three times per week with sessions of 3-4 hours/day, with arteriovenous fistula. No patients with hepatitis C were undergoing antiviral treatment. The sociodemographic and clinical data were obtained through questionnaires and medical records. |
| Measurement of the total antioxidant capacity |
| The total antioxidant capacity (TAC) against peroxyl radicals (ROO•) was determined according to Amado et al.. Aliquots of testis homogenates were placed in a medium containing 30 mM HEPES (pH 7.2), 200 mM KCl, 1 mM MgCl2 and 40 nM of the fluorogenic compound 2’,7’-dichlorofluorescein diacetate (H2DCF-DA; Invitrogen) in the presence or absence of 2,2’-azobis (2-methylpropionamidine) dihydrochloride (ABAP; 4 mM; Aldrich), which generates ROO• by a thermal decomposition at 37°C. The fluorescence was considered as a measure of the ROS production and was read in a spectrofluorimeter equipped with a microplate reader (Victor 2; Perkin-Elmer) at wavelengths of 485 and 530 nm for excitation and emission, respectively. The total fluorescence generation was calculated by integrating the fluorescence units (FU) over the period of the measurement. The results were expressed as the area difference of FU × min in the same sample with and without the ABAP addition and standardized to the ROS area without ABAP (background area). The relative difference between the ROS area with and without ABAP was considered a measure of the antioxidant competence of the testis, where the area difference is inversely proportional to the antioxidant capacity. |
| Measurement of lipid peroxidation |
| A determination of the lipid peroxides was performed through an estimation of the malondialdehyde (MDA) content in the testis homogenates, employing the thiobarbituric acid-reactive substances (TBARS) fluorimetric method. Aliquots of the sample extracts (10 μl) were added to a reaction solution containing 150 μl of 20% acetic acid, 150 μl of thiobarbituric acid (0.8%), 50 μl of Milli-Q water and 20 μl of sodium dodecyl sulfate (SDS, 8.1%). This mixture was heated at 95°C for 30 min for derivatization. Following cooling for 10 min, 100 μl of Milli-Q water and 500 μl of n-butanol were added. After centrifugation (3,000 × g for 10 min at 15°C), 150 μl of the organic phase was placed in a microplate reader, and the fluorescence was recorded with wavelengths of 520 and 580 nm for excitation and emission, respectively. The concentration of TBARS (nM/mg of wet tissue) was calculated, employing a standard curve of tetramethoxypropane (TMP, Acros Organics) as the malondialdehyde equivalent. |
| Statistical analyses |
| Statistical analyses were performed using the STATA software version 12.1, Stata Corp, Texas. The descriptive analysis was performed using percentages for the categorical variables, the mean and standard deviation for the symmetric quantitative variables, and the median and interquartile range for the asymmetric variables. For the bivariate analyzes, χ2 tests (chi-square) and Fisher’s exact test were used to test differences of the categorical variables, and t test or Wilcoxon test were used for the continuous variables. In the multivariate analysis, backwards Poisson regression was used to assess the independent effect of the variables, where the prevalence ratios and their respective robust confidence intervals of 95% were calculated. Wald test was used as a statistical test. The value of p<0.05 was used as a cutoff point for the statistical significance. |
| Results |
| The average age of the groups was 59.4 years (±16.0); 51.7% were male; 67.2% were living without a partner. The clinical and laboratory data are shown in Table 1. No significant differences were observed between the groups in the following variables: sodium, hemoglobin and viral load (p>0.01). Aspartate transaminase (AST), alanine transaminase (ALT) and creatinine were significantly different (p<0.01). The sodium analysis in groups 2 and 4 was performed before and after HD but was not statistically significant. The determination of the viral load (VL) of HCV did not show significant differences; however, it was observed that the VL of group 3 was lower than group 4. |
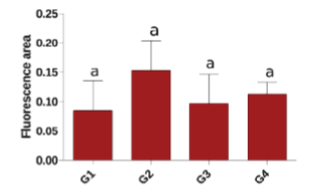
| According to Figure 1, a decrease was observed in the TAC of the group 2 patients when compared with the other groups, although it was not significant (p>0.05). |
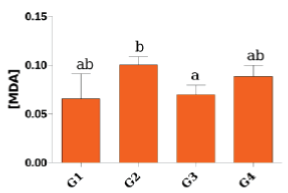
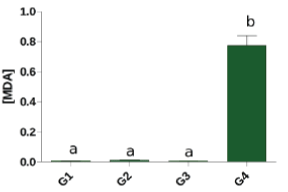
| Figure 2 shows the lipid peroxidation in erythrocytes, where a significant increase (p<0.05) was noted in the MDA concentration of the group 2 patients compared with group 3. There was no significant difference (p>0.05) between the patients in groups 1, 3 and 4 and between group 2 and group 4. In relation to the lipid peroxidation in the blood plasma, Figure 3 demonstrates a significant association (p<0.05) in group 4 compared with the others. |
| Discussion |
| In the present study, hepatic (AST and ALT) and blood parameters were analyzed as the oxidative stress parameters in the plasma and leukocytes of patients on hemodialysis and infected with HCV, patients who only underwent hemodialysis and control patients. We emphasize that the mean AST and ALT values among these groups were statistically significant. The values of the hepatic enzymes in the group 4 patients increased when compared with group 2 but were still within the normal range. Altered levels of transaminases are uncommon in patients with HCV who are undergoing hemodialysis treatment, suggesting that aminotransferases are poor predictors of hepatocellular injury in these patients, as the following are believed to occur in HD patients: an enzyme inhibition of the aminotransferases by uremic toxins, a pyridoxine deficiency (cofactor), a loss of enzymes on dialysis, an interference from other dialyzable substances in the determination of enzymatic activity of transaminases and lactate consumption by the necessary factors for the determination of transaminase activity [12,13]. As expected, the creatinine level was statistically significant among the groups because it was higher than the normal levels in the end-stage of renal disease (ESRD). |
| The VL values showed small differences (p>0.05) when analyzed between groups 3 and 4, suggesting a possible destruction and/or adherence of viral particles in the dialyzer membrane [14-16]. It is known that the VL influences the hepatitis C treatment and is used to evaluate the result compared with the virologic response, indicating lower or higher liver damage. |
| Accelerated aging, cataracts, atherosclerosis, a decreased number of red blood cells, increased hemolysis and platelet dysfunction are associated with patients with chronic renal failure. Such diseases are related to a hyperproduction of free radicals [17]. |
| It is known that the increasing OS in patients with CRF on HD can be an aggravating factor in the development of atherosclerosis, which is responsible for the high rate of morbidity and mortality among these patients [18]. OS is usually associated with inflammation in patients with end-stage renal disease (ESRD) in injured tissues, such as the inflammation related to reacting to invading microorganisms [19]. These reactions produce several ROS, which generate molecules that enhance the atherosclerotic process [20]. |
| In hemodialysis, blood is pumped through a vascular access for an extracorporeal filter, called the dialyzer, which is composed of a semipermeable membrane that extracts the metabolic products and excessive water and then returns filtered blood to the patient [21]. Therefore, it is clear that the hemodialysis procedure exposes the patient to extracorporeal processes and non-biological materials that alter the balance of the oxidant system. Studies show that the mortality rate in patients on HD ranges from approximately 50-80%. One of the factors related to this prevalence is the pathogenesis of atherosclerosis, which is directly related to OS [22,23]. |
| The TAC values (Figure 1) show a non-significant decrease in the group 2 patients, which corroborates the study by Nakayama et al. that attributes these values to a possible loss of molecules during the hemodialysis process [24]. In contrast, Horoz et al. found higher TAC in the control patients compared with those who were HCV reactive and non-reactive on hemodialysis [25]. We noted that the presence of HCV in the hemodialysis patients establishes a difference in the TAC values, although not significant. |
| Unlike Tutal et al. who found higher MDA levels in healthy patients, our analysis presented a significant correlation only between groups 2 and 3, showing that lipid peroxidation in hemodialysis patients is higher if compared with patients only with HCV [26]. The increase in lipid peroxidation may be related to the biocompatibility of the dialyzer membrane, which corroborates the results of Samouilidou et al. [3]. |
| Although there was no significant association, another finding that was observed was a minor difference in the MDA levels in patients with HCV who were on HD. The presence of HCV, through mechanisms not yet well understood, seems to cause a protective effect on dialysis. However, more studies are needed to support this analysis. |
| When the MDA leves are analyzed in the plasma of patients, there is a significant association between the groups. These dosages were considerably higher in the group 4 patients. This result indicates that the dialysis process, which is responsible for both increased ROS production and a reduction of antioxidant agents [20], together with the hepatic tissue injuries caused by HCV, increases the lipid peroxidation process. One factor to be considered in this analysis is that a viral infection by HCV progresses with an inflammation process in hepatocytes, leading to an increase of ROS and decreased plasma concentrations of antioxidants. This situation enables viral replication and chronification, inefficiency of the immune response and an aggravated clinical situation [27]. |
| The HCV viral structures, such as the core protein and the non-structural protein 3 (NS3) seem to be sufficient to produce mitochondrial alterations and generate OS [7]. The non-structural protein NS3 of HCV can stimulate ROS generation by the activation of the phagocyte NADPH oxidase and trigger apoptosis of immune cells. This process decreases cytotoxicity, blocks viral clearance and contributes to the chronicity of the disease [28]. In addition to this mechanism, the NS3 protein can also induce the formation of nitric oxide, which accounts for changes in DNA strands and the inactivation of the electron transport chain, which contributes to an imbalance of the mitochondrial respiratory chain, generates ROS and causes cell damage [29]. |
| The core is associated with the endoplasmic reticulum and can cause a depolarization of the mitochondrial membrane potential due to the endoplasmic reticulum stress caused by increased calcium in the mitochondria. The increase of calcium is associated with the generation of ROS [30]. |
| It should be emphasized that elevated MDA concentrations increase the inflexibility and deformity of red blood cell membranes, which in turn increase their susceptibility to hemolysis [31]. The occurrence of these factors contributes to the development of anemia in renal patients [32]. |
| In both methodologies applied to the MDA dosages, we identified high MDA concentrations in the hemodialysis patients with HCV. As expected, these patients had a higher lipid peroxidation showing oxidative stress caused by the hemodialysis procedure and viral injuries. It is important to note that factors, such as uremic syndrome, heart failure, inflammation and compatibility with the dialyzer membrane, are closely related to the development process of inflammation in hemodialyzed patients [4]. |
| Final Considerations |
| Our results indicate that dialysis together with inflammatory processes in the liver tissue caused by the hepatitis C virus increase lipid peroxidation, allowing increased short and long term damage to proteins and lipids, which accelerates the process of atherosclerosis and decreases the quality of life of these patients. |
| References |
|
Tables and Figures at a glance
| Table 1 |
Figures at a glance
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 |
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 17526
- [From(publication date):
December-2015 - Aug 31, 2025] - Breakdown by view type
- HTML page views : 16672
- PDF downloads : 854