Review Article Open Access
Presenilin 1 Mutation (A431V) Causing Features of Dementia with Lewy Bodies in a Chinese Family of Alzheimer��?s Disease
Yanan Qiao1, Dantao Peng2*, Miao Jin1 and Shuang Xue21Department of Neurology, China-Japan Friendship Hospital, Beijing 100029, PR China
2Department of Neurology, Center for Geriatric Medicine, China-Japan Friendship Hospital, Beijing 100029, PR China
- Corresponding Author:
- Dantao Peng
Department of Neurology
China-Japan Friendship Hospital
Beijing 100029, PR China
Tel: +8613910908579
E-mail: pengdantao@medmail.com.cn
Received date: February 16, 2017; Accepted date: February 25, 2017; Published date: February 28, 2017
Citation: Qiao Y, Peng D, Jin M, Xue S (2017) Presenilin 1 Mutation (A431V) Causing Features of Dementia with Lewy Bodies in a Chinese Family of Alzheimer’s Disease. J Alzheimers Dis Parkinsonism 7:307. doi:10.4172/2161-0460.1000307
Copyright: © 2017 Qiao Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Alzheimers Disease & Parkinsonism
Abstract
Aim: We reported a family with a presenilin 1 (PSEN1) gene mutation whose clinical manifestations are similar to the Dementia with Lewy bodies. Methods: We collected peripheral blood of the proband, his daughter and 100 normal Chinese individuals and extracted genomic DNAï¼�?PCR-sequencing of PSEN1 and microtubule associated protein tau (MAPT) were performed.We also gave them transcranial sonography test (TCS). Results: We found that the proband and his daughter were heterozygous for a mutation 1292nd base in exon 12 of PSEN1, causing the amino acid alanine substituded by valine at codon 431 (A431V), but this was not found in normal controlsï¼�?Meanwhile hyperechogenicity of bilateral substantia nigra could be seen in the two patients with the right-left asymmetry index >1.15. Conclusion: This study identified a mutation A431V in the PSEN1 gene in Chinese patients. We considered it might play an important role in familial Alzheimer’s disease leading clinical manifestations similar to DLB.
Keywords
Alzheimer’s disease; Dementia with lewy bodies; Mutation; Presenilin 1; Transcranial sonography
Introduction
Alzheimer’s disease (AD) and Dementia with Lewy bodies (DLB) are the most two common neurodegenerative diseases that cause progressive cognitive impairment. AD can be subdivided into sporadic type and familial type. The familial AD (FAD) is mostly early-onset, accounting for less than 5% of all AD cases, with Presenilin-1(PSEN1) and Presenilin-2 (PSEN2) as its genes, of which PSEN1 stands at the top [1-4]. Though, DLB is found mostly sporadic, without any confirmed genetic risk factor. Here, we describe a family presented with the clinical manifestations of DLB, who have a PSEN1 gene mutation.
Methods
Patients’ clinical data (Table 1)
| Patient | Age | Gender | Clinical | Presentations | Past history | MMSE | MOCA | |
|---|---|---|---|---|---|---|---|---|
| D | P | H | ||||||
| Ⅰ2 | dead | F | + | - | - | - | -- | -- |
| Ⅱ2 | 65 | F | + | + | - | HT | -- | -- |
| Ⅱ4 | 62 | F | + | + | + | - | -- | -- |
| Ⅱ5 | 58 | M | + | + | + | - | 4 | 3 |
| Ⅲ3 | 31 | F | + | - | - | - | 30 | 29 |
Table 1: Patients’ demographic and clinical data.
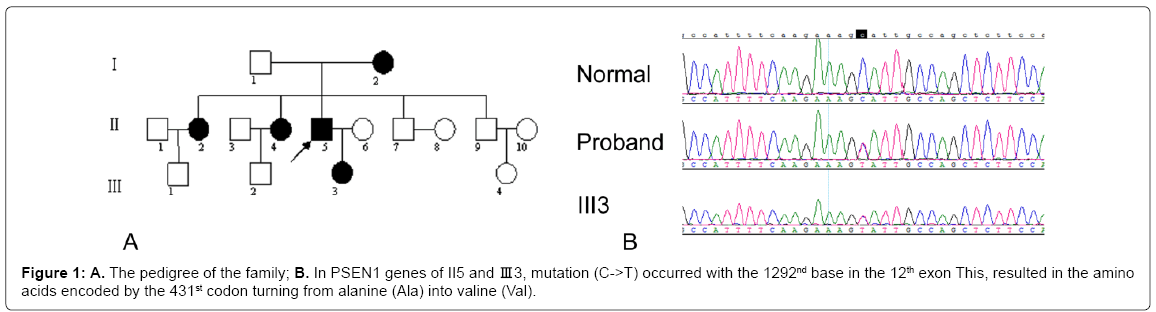
The patients come from a Chinese family, which has 16 members, 8 males and 8 females. The pedigree is shown in Figure 1A. According to the revised 2005 International Criteria of Dementia with Lewy bodies [5], the proband (member II5) conformed to the diagnosis criteria of probable clinical DLB, who is a 58 year old male and a National First-class Construction Engineer. He gradually developed cognitive decline and personality changes at the age of 54 years and began to exhibit Parkinsonian symptoms, hallucinations and significantly fluctuant cognitive impairment at 56 years old without any head trauma or toxic exposure or other past illness history. Mini-mental state examination (MMSE) was 4 points, Montreal cognitive assessment (MOCA) 3 points, and the third part of United Parkinson’s Disease Rate Scale (UPDRS) 19 points.
The member I 2 developed memory loss and personality changes at the age of over 50, who was dead now. The member II 3 and II 4 developed clinical symptoms similar as DLB, who got memory declining, slow movement and hallucinations at the age of their 50 years, both bed-ridden now. But they refused to take any physical examination and blood test.
The member III3, female, 31 years old, once healthy, is the daughter of the proband who did not show any memory loss, personality changes, Parinsonian symptoms and hallucination. Her MMSE scoring was 30 points, and MOCA 29 points (Figure 1B).
Genetic test
After complete description of the study to both of them, written informed consent was obtained from II 5 and Ⅲ 3. The study was approved by the local ethics committee at China-Japan Friendship Hospital. We took blood samples and tested microtubule associated protein tau (MAPT) and PSEN1 genes. A healthy control group of 100 members was also employed for comparison including 48 males and 52 females, with an average age of 52.1, who were tested PSEN1 gene mutation and all have signed the informed consent.
We used BigDye (Applied Biosystems;Axyprep DNA Gel EXraction kit) for test. The primer 3 online design (Sangon Biotech Beijing Co., Ltd.) was adopted. The polymerase chain reaction (PCR) amplification reaction system was No. 12 exon primer sequence: sense 5’-TCCAGATTGAATGAACGTCTGT-3’, antisense 5-AGACTTGGAAGGAAGCTGCA-3’. PCR products underwent electrophoresis on an ABI automated DNA sequencer (Perkin Elmer, Foster City, Calif). The data of electropherograms was analyzed using DNASTAR sequencing analysis software (Lasergene.v7.1).
Transcranial sonography (TCS) test
TCS was performed through the preauricular acoustic bone windows using a colour-coded phased-array ultrasound system equipped with a 2.5 MHz transducer (LOGIQ 9 ultrasound system). The ultrasound parameters were: penetration depth of 16 cm, dynamic range 50 dB. SN echogenic size measurements were performed on axial TCS scans automatically after manually encircling the outer circumference of substantia nigra (SN)’s echogenic area. SN echogenic sizes of less than 0.20 cm2 are classified as normal. Additionally, echogenicity of thalami, lenticular nucleus and heads of caudate nucleus was investigated and classified as hyperechogenic when it was more intense than the surrounding white matter. For estimation of the right-left asymmetry index of SN echogenic sizes (SN+ R-L index), the larger size of bilateral measurements was divided by the smaller size. All TCS examinations were performed by one experienced sonographer (S.X.) who was blinded to diagnosis and clinical data of the patients.
Results
Imaging and laboratory test data
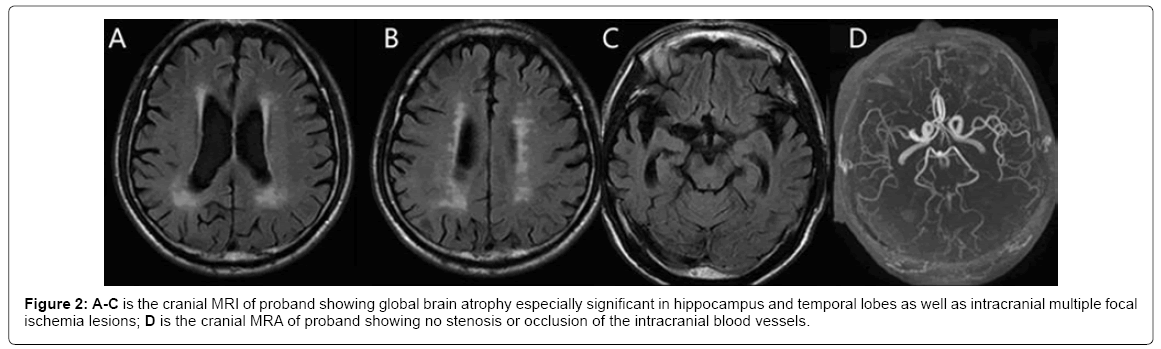
Proband II 5: Cranial magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA) showed (Figure 2) global brain atrophy especially in hippocampus and temporal lobes as well as intracranial multiple lacunar infarctions. Susceptibility weighted imaging (SWI) MRI showed no intracranial microbleeds. Single- Photon Emission Computed Tomography (SPECT) showed lower blood perfusion in temperal, frontal, parietal and occipital lobes. Electroencephalogram (EEG) showed diffusing and persistent complex slow wave in both waking and sleeping states. The result of apolipoprotein E (Apo E) gene was ε3/3. The 2-6 exons of notch homolog protein 3 (NOTCH3) genes had no pathogenic mutation. All baseline laboratory results, including examinations of peripheral blood and blood biochemistry (ureanitrogen and creatinine, serum vitamin B12 and folate levels), serologic tests for syphilis and for thyroid function, urine analysis and chest radiography, were within normal ranges.
The cranial MRI of III 3 had no obvious abnormality: She refused to do SWI, SPECT, EEG and serological laboratory examinations.
Genetic testing results
In PSEN1 genes of II5 and Ⅲ 3, mutation (C->T) occured with the 1292nd base in the 12th exon. This resulted in the amino acids alanine encoded by the 431st codon substituted by valine (A431V). The normal control group showed no similar PSEN1 mutation. II5 and Ⅲ 3 found no MAPT pathogenic mutations.
Trascranial sonography (TCS)
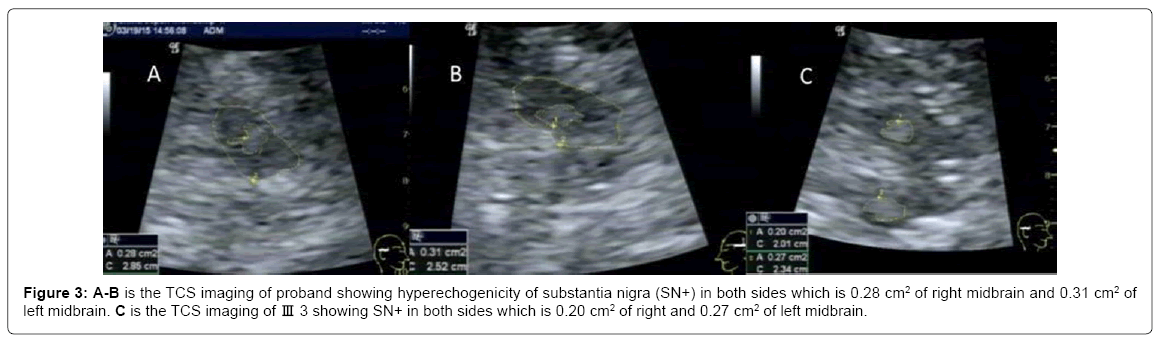
Hyperechogenicity of substantia nigra (SN+) in both sides which is 0.28 cm2 of right midbrain and 0.31 cm2 of left midbrain and abnormal hyperechogenicity in bilateral lentiform nucleus were found in Proband II5. The SN+ R-L index is 1.26 (Figures 3A and 3B).
The member III3 also had SN+ which is 0.20 cm2 of right midbrain and 0.27 cm2 of left midbrain .The SN+ R-L index is 1.35 (Figure 3C).
Discussion
About 5% of patients with AD are FAD with genetic changes. PSEN1 gene mutation accounts for 18%-55% in the FAD families [2]. PSEN1 gene is located at 14q24.2 with the product of 3.1KB and the encoding area composed of 10 exons. It encodes a protein of 467 amino acids about 42-43 ku long. So far, over 200 kinds of PSEN1 gene mutations have been found [6], of which the most are missense mutation at a single position. In our report, two patients of the family have C->T mutation of the 431st codon in the 12th exon at PSEN1 gene. Matsushita et al. [7] in 2002 found mutation at this position in an early-onset AD family, but not in normal controls. As a result, the mutation at this position was considered as a pathological mutation. Therefore, the family of our research was finally diagnosed as FAD caused by mutation of the 12th exon of PSEN1 gene. However, the patients of the family mentioned in Matsushita’s article showed no DLB symptoms. It might be related to the mild cognitive impairment at that time. But Matsushita did not describe the detail about the following up of the family. So whether the patients developed DLB symptoms at the late stage of the disease was not clear.
More and more researches [8-13] proved that the mutation of PSEN1 is widely involved in the AD pathological processes which include senile plaques (SPs), neurofibrillary tangles (NFT) and loss of neurons and synapses: (1) The mutation of PSEN1 can selectively cause the increasing of β-amyloid protein-42 by two or three times, and enhance the aggregation of pathological tau nerve filaments; (2) The abnormal modification of tau protein which relates to the formation of NFT has a positive correlation with the severity of dementia of AD. The mutation of PSEN1 can combine with microtubule to promote excessive phosphorylation of tau protein by glycogen synthase kinase- 3β (GSK-3β).
In this family, the proband II5 met the revised international clinical diagnosis criteria of DLB by MeKeith et al. [5] in 2005.Other members of the family (II2 and II4) also have the clinical manifestations similar to DLB. Though III3 has no DLB manifestations, her tested DNA also has PSEN1 gene mutation. This needs further follow-up. So far, there was no report about the mutation of PSEN1 gene (A431V) leading to DLB manifestations. Snider et al. [14] once reported DLB symptoms appearing in PSEN1 gene FS170 mutation. Ishikawa [15] reported a case that the deletion of the 440th position in PSEN1 leads to the loss of threonine, which also appears as DLB. It is not sure why the mutation of gene PSEN1 can lead to symptoms like DLB. Some cases of autopsy [14- 16] proved that in addition to AD pathological features, Lewy Bodies (LBs) which is the key pathological feature of DLB is also a very frequent coexistent pathologic abnormality in FAD. It can be found wide spread in the brainstem, limbic areas, nigra and neocortex. Meanwhile, these pathological changes of LBs occur more frequently in FAD related to gene PSEN1 mutation than FAD related to Presenilin-2 gene mutation and sporadic AD [17]. It was speculated that in the normal condition PSEN1 plays an important role in certain interations or interfuctions between β-amyloid andα-synuclein. This has been proved by Winslow [17] using immunoelectron microscopy found that PSEN1 together with α-synuclein and β-amyloid are closely related on the presynaptic vesicles, surfaces of mitochondria and plasma membrane. Aggregation of α-synuclein into Lewy bodies (LBs) can also be induced by PSEN1 mutation to an equal extent as β-amyloid pathology [15]. These above studies and assumptions provided proofs to the relationships between PSEN1 and α-synuclein and could state the mechanism of the AD patients of PSEN1 gene mutation presented with DLB clinical features.
It is noteworthy that by TCS hyperechogenicity of bilateral substantia nigra can be seen in the two patients of PSEN1 A431V mutation. TCS shows that 90% of the patients with Parkinson’s diseases (PD) have abnormally enlarged SN+ [18]. In addition to PD, corticobasal ganglionic degeneration (CBD), DLB and many other neurodegenerative diseases also have SN+. Walter et al. [19] believed that the features of SN+ of DLB are the areas of SN+ ≥ 0.25cm2, and the bilateral asymmetry index <1.15. It is suggested that the SN+ is related to iron metabolism rather than Lewy body accumulation. In our research, the asymmetry index of SN echogenic sizes of both II 5 and III 3>1.15 and II 5 also has hyperechogenicity of bilateral lentiform nucleus. These are inconsistent with TCS features of the previously reported DLB. It is considered that our family is early-onset AD with DLB clinical features, not sporadic DLB, so the SN+ is not similar as sporadic DLB. But in a sense, it may give a hint that asymmetry of bilateral SN+ may be one of characteristic features in differentiating FAD with DLB from sporadic DLB.
Conclusion
This research reported that the early-onset FAD of PSEN1 gene A431V mutation has the clinical features of DLB for the first time. It helps understanding clinical manifestations of FAD with PSEN1 mutation and suggests that when we meet with familial DLB, earlyonset FAD with PSEN1 mutation should be considered and TCS may be one of important methods to differentiate them. As a limitation of this research, the gene has not been detected in other family members, especially the other two patients. Furthermore, though positive in gene test, III3 has no symptoms. We will further follow up for more clinical data.
Acknowledgement
The authors are thankful to the participants, their families and healthy volunteers. The authors are also grateful to the members of Sangon Biotech Beijing Co., Ltd to examine the blood samples.
References
- Kamimura K, Tanahashi H, Yamanaka H, Takahashi K, Asada T, et al. (1998) Familial alzheimer's disease genes in Japanese. J Neurol Sci 160: 76-81.
- Campion D, Dumanchin C, Hannequin D, Dubois B, Belliard S, et al. (1999) Early-onset autosomal dominant alzheimer disease: Prevalence, genetic heterogeneity and mutation spectrum. Am J Hum Genet 65: 664-670.
- Finckh U, Müller-Thomsen T, Mann U, Eggers C, Marksteiner J, et al. (2000) High prevalence of pathogenic mutations in patients with early-onset dementia detected by sequence analyses of four different genes. Am J Hum Genet 66: 110-117.
- Athan ES, Williamson J, Ciappa A (2001) A founder mutation in presenilin 1 causing early-onset Alzheimer disease in unrelated caribbean hispanic families. JAMA 286: 2257-2263.
- McKeith IG, Dickson DW, Lowe J, Emre M, O'Brien JT, et al. (2005) Diagnosis and management of dementia with Lewy bodies: Third report of the DLB Consortium. Neurology 65: 1863-1872.
- Larner AJ (2013) Presenilin-1 mutations in Alzheimer's disease: An update on genotype-phenotype relationships. J Alzheimers Dis 37: 653-659.
- Matsushita S, Arai H, Okamura N, Ohmori T, Takasugi K, et al. (2000) Clinical and biomarker investigation of a patient with a novel presenilin-1 mutation (A431V) in the mild cognitive impairment stage of Alzheimer’s disease. Biol psychiatry 52: 907-910.
- Borchelt DR, Thinakaran G, Eckman CB, et al. (1996) Familial alzheimer’s disease-linked presenilin 1 variants elevate a beta 1-42/1-40 ratio in vitro and in vivo. Neuron 17: 1005-1013.
- Keller JN, Guo Q, Holtsberg FW, Bruce-Keller AJ, Mattson MP (1998) Increased sensitivity to mitochondrial toxin-induced apoptosis in neural cells expressing mutant presenilin-1 is linked to perturbed calcium homeostasis and enhanced oxyradical production. J Neurosci 18: 4439-4450.
- Takashima A, Murayama M, Murayama O, Kohno T, Honda T, et al. (1998) Presenilin 1 associates with glycogen synthase kinase-3beta and its substrate tau. Proc Natl Acad Sci U S A 95: 9637-9641.
- Weihl CC, Miller RJ, Roos RP (1999) The role of beta-catenin stability in mutant PS1-associated apoptosis. Neuroreport 10: 2527-2532.
- De Ferrari GV, Inestrosa NC (2000) Wnt signaling function in Alzheimer’s disease. Brain Res Brain Res Rev 33: 1-12.
- Gamblin TC, Chen F, Zambrano A, Abraha A, Lagalwar S, et al. (2003) Caspase cleavage of tau: Linking amyloid and neurofibrillary tangles in Alzheimer’s disease. Proc Natl Acad Sci U S A 100: 10032-10037.
- Snider BJ, Norton J, Coats MA, Chakraverty S, Hou CE, et al. (2005) Novel presenilin 1 mutation (S170F) causing Alzheimer disease with lewy bodies in the third decade of life. Arch Neurol 62: 1821-1830.
- Ishikawa A, Piao YS, Miyashita A, Kuwano R, Onodera O, et al. (2005) A mutant PSEN1 causes dementia with lewy bodies and variant Alzheimer’s disease. Ann Neurol 57: 429-434.
- Leverenz JB, Fishel MA, Peskind ER, Montine TJ, Nochlin D, et al. (2006) Lewy body pathology in familial Alzheimer disease: Evidence for disease and mutation specific pathologic phenotype. Arch Neurol 63: 370-376.
- Winslow AR, Moussaud S, Zhu L, Post KL, Dickson DW, et al. (2014) Convergence of pathology in dementia with lewy bodies and Alzheimer’s disease: A role for the novel interaction of alpha-synuclein and presenilin 1 in disease. Brain 137: 1958-1970.
- Berg D, Behnke S, Seppi K, Godau J, Lerche S, et al. (2013) Enlarged hyperechogenic substantia nigra as a risk marker for Parkinson's disease. Mov Disord 28: 216-219.
- Walter U, Dressler D, Wolters A, Wittstock M, Greim B, et al. (2006) Sonographic discrimination of dementia with Lewy bodies and Parkinson's disease with dementia. J Neurol 253: 448-454.
Relevant Topics
- Advanced Parkinson Treatment
- Advances in Alzheimers Therapy
- Alzheimers Medicine
- Alzheimers Products & Market Analysis
- Alzheimers Symptoms
- Degenerative Disorders
- Diagnostic Alzheimer
- Parkinson
- Parkinsonism Diagnosis
- Parkinsonism Gene Therapy
- Parkinsonism Stages and Treatment
- Stem cell Treatment Parkinson
Recommended Journals
Article Tools
Article Usage
- Total views: 4239
- [From(publication date):
February-2017 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 3261
- PDF downloads : 978