Case Report Open Access
Primary Oral Melanoma: A Case Report with Immunohistochemical Findings
Juliana de Souza do Nascimento1, Adalberto Mosqueda Taylor2, Oslei Paes de Almeida3 and Bruno Augusto Benevenuto de Andrade4*1School of Dentistry, Fluminense Federal University (UFF), Nova Friburgo, Rio de Janeiro, Brazil
2Department of Health Care, Metropolitan Autonomous University Xochimilco, México
3Oral Pathology, School of Dentistry, State University of Campinas (UNICAMP), Piracicaba, São Paulo, Brazil
4Oral Pathology, Department of Oral Diagnosis and Pathology, School of Dentistry, Federal University of Rio de Janeiro (UFRJ), Rio de Janeiro, Brazil
- *Corresponding Author:
- Bruno Augusto Benevenuto de Andrade
DDS, PhD, Department of Oral Diagnosis and Pathology
School of Dentistry, Federal University of Rio de Janeiro (UFRJ)
Rio de Janeiro, Brazil
Tel: +55 21 25622071
Fax: +55 21 25622071
E-mail: augustodelima33@hotmail.com
Received date: September 30, 2014; Accepted date: October 16, 2014; Published date: October 19, 2014
Citation: Nascimento JS, Taylor AM, Almeida OP, Andrade BAB (2014) Primary Oral Melanoma: A Case Report with Immunohistochemical Findings. J Clin Exp Pathol 4:197. doi:10.4172/2161-0681.1000197
Copyright: ©2014 Nascimento JS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Clinical & Experimental Pathology
Abstract
Melanoma is a potentially aggressive and rare malign neoplasm of melanocytic origin. Only 1% occurs in oral mucosa, accounting for 0.5% of all oral malignancies being more aggressive when compared with the cutaneous counterpart. The tumor occurs more frequently in the hard palate and gingiva. Despite the advances in biological knowledge of melanoma, treatment and prognosis still have limitations, especially in head and neck melanomas, including oral melanomas. The aim of this study is to report a case of primary oral melanoma in a 54 year-old male patient. An asymptomatic pigmented lesion was located in the upper vestibular gingiva and an incisional biopsy was done. Cytomorphological findings revealed proliferation of pleomorphic epithelioid and plasmacytoid cells positive by immunohistochemistry for S-100 protein, HMB-45 and Melan-A, confirming the diagnostic of oral melanoma. This study showed the importance of the histopathological and immunohistochemical evaluation in order to determine the cytomorphologic aspects of oral melanoma to establish the final diagnosis.
Keywords
Oral melanoma; Immunohistochemistry; Histopathology
Introduction
Melanoma is a malignant tumor comprises of abnormal melanocytes, which are cells derived from the neural crest that constitute the melanin pigment in the basal layer of epithelium [1]. Over 90% of melanomas occur on the skin, but they may also arise from mucosal surface or at other sites wherein neural crest migrate, like oral and genital mucosa, nasal cavity and leptomeningeal area [1-3]. Primary oral melanomas are far less common than their cutaneous counterparts, comprising 0.4-1.8% of all melanomas and 0.5% of oral malignances [3-5]. The mean age of patients with oral melanoma is approximately 60 years, with a wide range varying from roughly 20 years to more than 90 years. There is a modest male preponderance [3-5]. It occurs most frequently in the hard palate and maxillary gingiva: other oral sites are mandibular gingiva, tongue, buccal mucosa, upper and lower lip [5-8]. Primary oral melanoma is initially asymptomatic and usually not noticed by the patients, which contribute to the delay on diagnosis [3,9,10].
Clinical features vary, but the most common presentation is a brown, dark blue, gray, purple or black macule, sometimes with erythema or ulceration [4,10,11]. A more advanced disease may take on nodular or irregular surface characteristics. They may exhibit asymmetric and irregular borders just like cutaneous melanomas. In the mouth, bony erosion is common [4].
The histopathologic appearance of oral melanoma is variable. It can show two principal patterns: an in situ pattern in which the neoplasm is limited to the epithelium and the epithelial-connective tissue interface, and an invasive pattern in which the neoplasm is found within the supporting connective tissue. A combined pattern of invasive melanoma with in situ component is typical for most advanced lesions [12]. Epithelioid, spindle and plasmacytoid tumor cells arranged in a sheet-like, organoid, alveolar, solid or desmoplasic configuration may be seen [4,5,12,13]. Melanomas with characteristic melanin-pigmented tumor cells are easy to diagnose, but the diagnosis can be difficult when the tumor is amelanotic [14].
Immunohistochemistry (IHC) has been the primary tool to distinguish melanomas from other tumors, for example, poorly differentiated carcinomas, neuroendocrine tumors, sarcomas and germ cell tumors [14-16]. The final diagnosis usually requires IHC with certain antibodies directed against melanocytic differentiation antigens. S-100, HMB-45 and Melan-A are currently the three most useful immunomarkers to identify melanocytes and melanomas [17-22].
In this case report, we examined the clinical, histopathological and immunohistochemical expression of S-100, HMB-45 and Melan-A in a primary oral melanoma located in the upper vestibular gingiva in a 54 year-old patient.
Case Report
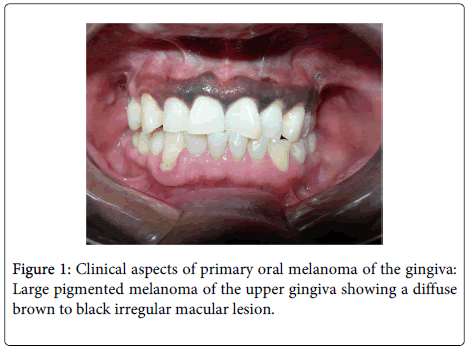
A 54 year-old male mexican patient, active smoker of 2 packets of cigarette/day for 20 years was referred for examination of a 6-months pigmented lesion in gingiva. In the physical exploration it was observed a diffuse asymptomatic pigmented macular lesion ranging from brown to black and extending across the upper vestibular gingiva of the teeth 14 to 24 without ulceration or erythema (Figure 1).
The associated teeth did not present mobility with panoramic and periapical radiograph not showing alteration. Upon clinical diagnosis of racial melanin pigmentation and oral melanoma, an incisional biopsy was done.
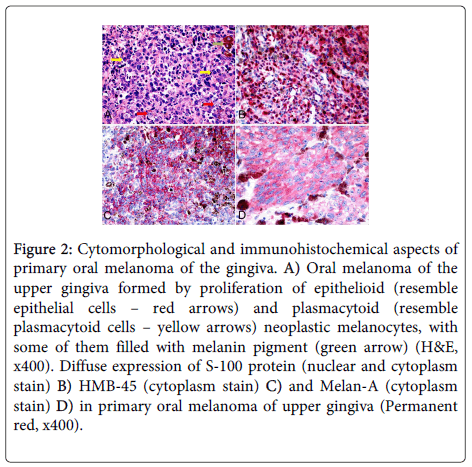
The material was preserved in 10% buffered formalin and paraffin-embedded tissue block. Histopathological findings revealed a proliferation of a polymorphous infiltrate of pleomorphic epithelioid and plasmacytoid cells containing large quantities of melanin invading the lamina propria and deep connective tissue. Mitoses, perivascular and perineural invasion were also observed. Immunohistochemical analysis for S100 protein, Melan-A and HMB-45 antibodies was performed. For immunohistochemical staining three-micrometer-thick sections were used. Briefly, after antigen retrieval with EDTA/Tris buffer (pH 9.0) in a microwave oven, endogenous peroxidase activity was blocked with 20% H2O2 for 5 cycles of 5 minutes each. Primary antibodies after overnight incubation were detected by secondary antibodies conjugated with polymer dextran marked with peroxidase (Dako EnVision Labelled Polymer; Dako, Glostrup, Denmark). The reaction was developed with Permanent Red (Permanent Red Substrate System, Dako). The preparations were lightly counterstained with Carazzi hematoxylin, mounted with Aquatex (MERCK, Germany) and examined by light microscope.
All antibodies showed a strong and diffuse positive staining of epithelioid and plasmacytoid cells, indicating the melanocytic origin of these cells (Figure 2). With the diagnosis of primary oral melanoma of the upper gingiva, surgical intervention was performed, with the resection of the gingival lesion. The patient died after six months of diagnosis by surgical complications of the lesion resection.
Figure 2: Cytomorphological and immunohistochemical aspects of primary oral melanoma of the gingiva. A) Oral melanoma of the upper gingiva formed by proliferation of epithelioid (resemble epithelial cells – red arrows) and plasmacytoid (resemble plasmacytoid cells – yellow arrows) neoplastic melanocytes, with some of them filled with melanin pigment (green arrow) (H&E, x400). Diffuse expression of S-100 protein (nuclear and cytoplasm stain) B) HMB-45 (cytoplasm stain) C) and Melan-A (cytoplasm stain) D) in primary oral melanoma of upper gingiva (Permanent red, x400).
Discussion
Primary oral melanoma is a rare neoplasm accounting for only 0.5% of all oral malignances and with an incidence of 1.2 cases per 10 million per year [4,10,13]. This lesion occurs more frequently in male than female with mean age of 50 years old [23]. There is a low incidence of this tumor in Europe and Australia [2]. However, higher percentages are reported in Japanese populations and in other nonwhite races like Uganda, India and North American Indian populations [6,7]. Among the Japanese, oral melanoma accounts for 11-14% of all cases of melanoma [7,8]. A definitive clinicopathological classification of oral melanoma has not yet been drawn up [10,12,24].
Clinically, oral melanoma commonly affect the hard palate, gingiva or alveolar ridge, but lips, tongue, floor of the mouth and uvula can also be involved. It can demonstrate a variable clinical presentation, such as macule, plaque or a mass. This lesion has a regular or irregular surface, with an intact or ulcerated mucosa. The pigmentation can be homogeneous from black to brown; but in some cases exhibit variation of color with black, brown, grey, purple or red; and sometimes no staining (amelanotic). Nonspecific signs and symptoms may include pain, paresthesia or anesthesia, bleeding, tooth mobility and bone loss [23,25,26]. The patient of this study showed an asymptomatic, not elevated and uniform pigmentation lesion without ulcerated areas.
Histopathological classification and staging of cutaneous melanomas using Breslow and Clark levels cannot be applied to oral mucosa tumors due to structural differences between skin and mucosa [13,27]. Oral melanoma differs from cutaneous melanoma in several aspects. Established risk factors for cutaneous melanoma, such as sun damage, a family history of melanoma, or atypical nevi do not apply to oral melanoma. Likewise, oral melanocytic tumors need to be studied separately from those occurring on the skin [16]. Prasad et al. [28] proposed a classification based on the level of melanoma invasion as microinvasive (level I, cell clusters in the superficial lamina propria), invasive (level II, cell invasion into the lamina propria), and deep invasive (level III, invasion into skeletal muscle, bone or cartilage), and this classification was employed in our case. Our case showed level III of invasion, characterized by invasion of tumor cells into the lamina propria and deep connective tissue. The advanced stage in the moment of diagnostic may be a consequence of delay in the diagnosis of pigmented lesions in the oral cavity. Lourenço et al. [13] in their series of 35 primary oral melanomas, 28 (82.35%) were diagnosed as level III of invasion and only 1 case (2.94%) as level I, while the other 6 cases were classified as level II. Prasad et al. [28] suggested that one of the most important histopathological predictor or worst prognosis is tumor histological level, being level III tumors trends for worst outcome.
A proliferation of pleomorphic epithelioid and spindle cells containing inside a significant amount of melanin were observed in this case. Pleomorphic cells have been described in oral mucosal melanomas and this classification seems to have no impact in prediction of tumor behavior [5,12,13]. However, Lourenço et al. [13] observed in their series that polymorphous tumors had a slightly higher incidence of vascular infiltration and necrosis.
Melanoma has a wide spectrum of histologic features which mimic epithelial, mesenchymal and neural tumors being the immunohistochemistry the primary tool to distinguish melanoma from these other tumors [14-16]. Yu et al. [17] and Andrade et al. [29] studied the expression of melanocytic differentiation markers. The first one founded 6 primary oral melanomas with 100% positive rate for HMB-45 compared with an 83% positive rate for S-100 and a 67% positive rate for Melan-A in six primary oral melanomas, suggesting that both HMB-45 and S-100 may be good markers for immunohistochemical diagnosis of primary oral melanoma. The second study showed 22 primary oral melanomas marked by HMB-45 and S-100, while Melan-A was not expressed in 3 cases. In this study, S100 protein, Melan-A and HMB-45 antibodies were positives for epithelioid and plasmacytoid cells, confirming that they were melanocytic cells. Prasad et al. [16] assessed the expression of HMB-45, S-100 and Melan-A in 48 primary sinonasal and oral melanoma by IHC, and found a positive rate of 85%, 96% and 94% respectively, which indicate that S-100 and Melan-A are better markers than HMB-45 for immunohistochemical diagnosis [15-17].
Conclusion
This study showed the importance of the histopathological and immunohistochemical evaluation in order to determine the morphological aspects of oral melanoma to establish the final diagnosis.
References
- Femiano F, Lanza A, Buonaiuto C, Gombos F, Di Spirito F, et al. (2008) Oral malignant melanoma: a review of the literature. J Oral Pathol Med 37: 383-388.
- Hashemi Pour MS (2008) Malignant melanoma of the oral cavity: a review of literature. Indian J Dent Res 19: 47-51.
- Gu GM, Epstein JB, Morton TH Jr (2003) Intraoral melanoma: long-term follow-up and implication for dental clinicians. A case report and literature review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 96: 404-413.
- Hicks MJ, Flaitz CM (2000) Oral mucosal melanoma: epidemiology and pathobiology. Oral Oncol 36: 152-169.
- Meleti M, Rene´ Leemans C, Mooi WJ, Vescovi P, van der Waal I (2007) Oral malignant melanoma: a review of the literature. Oral Oncol 43: 116-121.
- Rapini RP (1997) Oral melanoma: diagnosis and treatment. Semin Cutan Med Surg 16: 320-322.
- Wagner M, Morris CG, Werning JW, Mendenhall WM (2008) Mucosal melanoma of the head and neck. Am J Clin Oncol 31: 43-48.
- Manolidis S, Donald PJ (1997) Malignant mucosal melanoma of the head and neck: review of the literature and report of 14 patients. Cancer 80: 1373-1386.
- Umeda M, Komatsubara H, Shigeta T, Ojima Y, Minamikawa T, et al. (2008) Treatment and prognosis of malignant melanoma of the oral cavity: preoperative surgical procedure increases risk of distant metastasis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 106: 51-57.
- Garzino-Demo P, Fasolis M, Maggiore GM, Pagano M, Berrone S (2004) Oral mucosal melanoma: a series of case reports. J Craniomaxillofac Surg 32: 251-257.
- Younes MN, Myers JN (2004) Melanoma of the head and neck: current concepts in staging, diagnosis, and management. Surg Oncol Clin N Am 13: 201-229.
- Barker BF, Carpenter WM, Daniels TE, Kahn MA, Leider AS, et al. (1997) Oral mucosal melanomas: the WESTOP Banff workshop proceedings. Western Society of Teachers of Oral Pathology. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 83: 672-679.
- Lourenço SV, A MS, Sotto MN, Bologna SB, Giacomo TB, et al. (2009) Primary oral mucosal melanoma: a series of 35 new cases from South America. Am J Dermatopathol 31: 323-330.
- Banerjee SS, Harris M (2000) Morphological and immunophenotypic variations in malignant melanoma. Histopathology 36: 387-402.
- Ohsie SJ, Sarantopoulos GP, Cochran AJ, Binder SW (2008) Immunohistochemical characteristics of melanoma. J Cutan Pathol 35: 433-444.
- Prasad ML, Jungbluth AA, Iversen K, Huvos AG, Busam KJ (2001) Expression of melanocytic differentiation markers in malignant melanomas of the oral and sinonasal mucosa. Am J Surg Pathol 25: 782-787.
- Yu CH, Chen HH, Liu CM, Jeng YM, Wang JT, et al. (2005) HMB-45 may be a more sensitive maker than S-100 or Melan-A for immunohistochemical diagnosis of primary oral and nasal mucosal melanomas. J Oral Pathol Med 34: 540-545.
- Moore BW (1965) A soluble protein characteristic of the nervous system. Biochem Biophys Res Commun 19: 739-744.
- Salama I, Malone PS, Mihaimeed F, Jones JL (2008) A review of the S100 proteins in cancer. Eur J Surg Oncol 34: 357-364.
- Busam KJ (2004) The use and application of special techniques in assessing melanocytic tumours. Pathology 36: 462-469.
- Gown AM, Vogel AM, Hoak D, Gough F, McNutt MA (1986) Monoclonal antibodies specific for melanocytic tumors distinguish subpopulations of melanocytes. Am J Pathol 123: 195-203.
- Carlson JA, Ross JS, Slominski AJ (2009) New techniques in dermatopathology that help to diagnose and prognosticate melanoma. Clin Dermatol 27: 75-102.
- Lourenço SV, Fernandes JD, Hsieh R, Coutinho-Camillo CM, Bologna S, et al. (2014) Head and neck mucosal melanoma: a review. Am J Dermatopathol 36: 578-587.
- Chidzonga MM, Mahomva L, Marimo C, Makunike-Mutasa R (2007) Primary malignant melanoma of the oral mucosa. J Oral Maxillofac Surg 65: 1117-1120.
- Alawi F (2013) Pigmented lesions of the oral cavity: an update. Dent Clin North Am 57: 699-710.
- Sun CZ, Chen YF, Jiang YE, Hu ZD, Yang AK, et al. (2012) Treatment and prognosis of oral mucosal melanoma. Oral Oncol 48: 647-652.
- Clark WHJr (1966) A classification of malignant melanoma in man correlated with histogenesis and biologic behavior. In: montagna W, Hus (Ed). Advances in the biology of the skin. Volume 8. The pigmentary system, New York: Pergamon Press : 621.
- Prasad ML, Patel SG, Huvos AG, Shah JP, Busam KJ (2004) Primary mucosal melanoma of the head and neck: a proposal for microstaging localized, Stage I (lymph node-negative) tumors. Cancer 100: 1657-1664.
- de-Andrade BA, Toral-Rizo VH, León JE, Contreras E, Carlos R, et al. (2012) Primary oral melanoma: a histopathological and immunohistochemical study of 22 cases of Latin America. Med Oral Patol Oral Cir Bucal 17: e383-388.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 17818
- [From(publication date):
December-2014 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 12948
- PDF downloads : 4870