Significance of Diagnostic Paracentesis in the Preoperative Diagnosis of Strangulated Small Bowel Obstruction
Received: 20-Jun-2018 / Accepted Date: 27-Jun-2018 / Published Date: 30-Jun-2018 DOI: 10.4172/2161-069X.1000568
Abstract
Objective: This study aimed to clarify the significance of diagnostic paracentesis (DPC) and ascites analysis in the preoperative diagnosis of strangulated small bowel obstruction (SBO). Methods: Between June 2008 and May 2018, 38 patients underwent DPC and ascites analysis based on a preoperative diagnosis of strangulated SBO. The patients were divided into two groups based on the intraoperative macroscopic bowel condition: the strangulated (26 patients) and simple (12 patients) groups. The following factors were investigated and compared between the two groups: patient characteristics, preoperative vital signs, blood test results, ascites analysis results white blood cell [WBC] and red blood cell [RBC] counts, and lactate dehydrogenase and creatinine phosphokinase levels), and computed tomography (CT) findings. Results: No procedure-related complications were noted. The vital signs did not differ significantly between the two groups. The arterial base excess (BE) level was significantly lower in the strangulated group than in the simple group, and a BE level of ≤ -1.1 mmol/L had a sensitivity of 65%, specificity of 92%, and overall accuracy of 74% for differentiating the two groups. The ascites RBC count was significantly higher in the strangulated group than in the simple group, and an ascites RBC count of ≥ 20,000/mm3 had a sensitivity of 81%, specificity of 83%, and overall accuracy of 82% for differentiating the two groups. Regarding the distribution of the patients, 24 (92%) patients of the strangulated group were present in the area of BE level of ≤ -1.1 mmol/L or ascites RBC count of ≥ 20,000/mm3. Strangulation sign on CT, besides accumulation of ascites, was more frequently and significantly detected in the strangulated group than in the simple group. Conclusion: Our results suggest that DPC and ascites analysis, particularly ascites RBC count, are significant in the preoperative diagnosis of strangulated SBO when combined with CT findings and BE level.
Keywords: Ascites analysis; Ascites red blood cell count; Base excess; Computed tomography; Diagnostic paracentesis; Simple small bowel obstruction; Strangulated small bowel obstruction
Introduction
Strangulated small bowel obstruction (SBO) is defined as a mechanical blockage of bowel lumen accompanied with compromised vascular supply [1], and is one of the most emergent and severe acute abdomens, of which prognosis greatly depends on early diagnosis and treatment. However, accurate preoperative diagnosis of strangulation is often difficult because per the literature [2], even the judgment of the senior attending surgeon was a sensitivity of 48%, specificity of 83%, and overall accuracy of 70%. It is very important to differentiate strangulated SBO from simple SBO accurately and quickly, because strangulated SBO requires emergency operation, whereas a simple SBO may simply require a planned operation following conservative treatment. Many authors [1-15] have investigated the optimal diagnostic parameters including patient’s complaints, abdominal physical findings, vital signs, blood test results, and computed tomography (CT) findings in an attempt to differentiate them; however, this method remains controversial. From our experiences, we have found that bloody ascites are frequently encountered at laparotomy for strangulated SBO. Kobayashi et al. [1] reported the effectiveness of diagnostic paracentesis (DPC) and ascites analysis for suspected strangulation, and in this study, we attempted to expound upon their work in order to clarify the significance and the role of DPC in the preoperative diagnosis of strangulated SBO.
This study was approved by the institutional review board of the Funabashi Central Hospital of Japan Community Health Care Organization, and details that might disclose the identity of the subjects under this study were carefully omitted.
Methods
Between June 2008 and May 2018, 56 patients underwent an emergency operation under the preoperative diagnosis (including suspicion) of strangulated SBO at our institution. Among the 56 patients, 38 patients, receiving preoperative ultrasound, showed sufficient amount of ascites to puncture at the liver surface, Morison’s pouch, around the spleen, or around the urinary bladder. These 38 patients underwent DPC and were enrolled in this study. The patients were then divided into two groups based on the intraoperative macroscopic bowel condition: the strangulated (26 patients) and simple (12 patients) groups. The following factors were investigated and compared between the two groups: patient characteristics (age, sex, and previous laparotomy), preoperative vital signs (body temperature, heart rate, arterial partial pressure of carbon dioxide, and systolic blood pressure), blood test results (white blood cell [WBC] and red blood cell [RBC] counts, lactate dehydrogenase [LDH], creatinine phosphokinase [CK], C-reactive protein [CRP], and arterial base excess [BE] levels), ascites analysis results (WBC and RBC counts, and LDH and CK levels), and strangulation sign on CT findings (mesenteric fluid, bowel wall thickening, closed loop sign, whirl sign, conversion of the mesenteries, or reduced bowel wall enhancement). In each group, all continuous data are expressed as medians (25th to 75th percentile) and categorical data are expressed as number (%), and statistical analysis was performed using SPSS 11.0TM. Differences between continuous variables were calculated using the Mann- Whitney U test, those between categorical variables were calculated using the chi-squared test, and a p-value of less than 0.05 (two-sided) was considered significant.
DPC was performed within 1 hour prior to the laparotomy as follows: after written informed consent was obtained from each patient, the site of puncture was determined by ultrasound and site marking was done on the skin. After antiseptic preparation of the skin and local anesthesia, blind paracentesis using a 23-gauge fine needle was performed, and 5 to 10 mL of ascites was aspirated and examined.
Results
No procedure-related complications such as hemorrhage, intestinal puncture, or infection were noted. Median age, sex distribution, and number of previous laparotomies were not significantly different between the two groups. The causes of SBO in the strangulated group were fibrous band, internal hernia, volvulus, and non-occlusive mesenteric ischemia. While in the simple group, the causes of SBO were intra-abdominal adhesion associated with prior laparotomies, food impaction, and enterocolitis (Table 1).
| Category | Strangulated (n=26) | Simple (n=12) | p-value |
|---|---|---|---|
| Age | 73.5 (61.8-85.0) | 77.5 (63.0-84.5) | 0.659 |
| Male patients | 9 (35) | 5 (42) | 0.675 |
| Previous laparotomy | 14 (54) | 8 (67) | 0.457 |
| Cause of SBO | Fibrous band 11 (42) | Adhesion 9 (75) | - |
| Internal hernia 7 (27) | Food impaction 2 (17) | - | |
| Volvulus 5 (19) | Enterocolitis 1 (8) | - | |
| NOMI 3 (12) | - | - |
Table 1: Patient characteristics; Continuous data are expressed as medians (25th to 75th percentile) and p-values were calculated using the Mann-Whitney U test. Categorical data are expressed as number (%) and p-values were calculated using the chi-squared test, SBO: Small bowel obstruction, NOMI: Non-occlusive mesenteric ischemia.
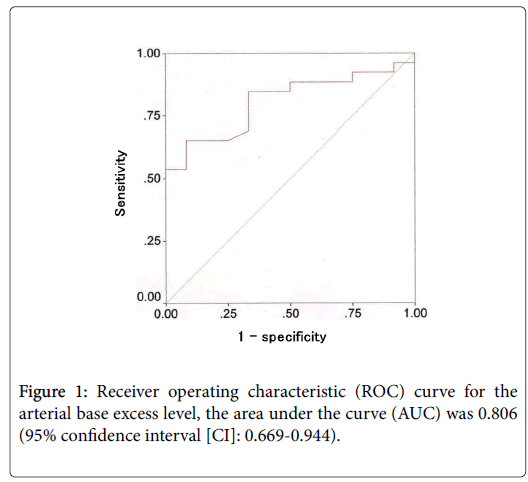
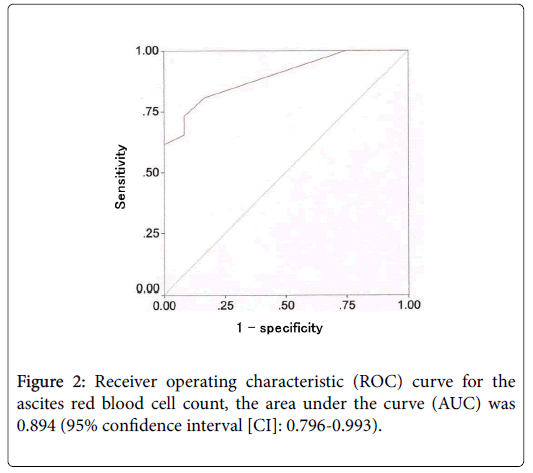
The vital signs did not differ significantly between the two groups (Table 2). Among the blood test results, only the BE level was significantly lower in the strangulated group than in the simple group (Table 2). We tried to determine the cut-off value of BE level using a receiver operating characteristic (ROC) curve, and a BE level of ≤ -1.1mmol/L had a sensitivity of 65%, specificity of 92%, and overall accuracy of 74% for differentiating the two groups (Figure 1).Among the ascites analysis results, only the ascites RBC count was significantly higher in the strangulated group than in the simple group (Table 2). We tried to determine the cut-off value of ascites RBC count using a ROC curve, and an ascites RBC count of ≥ 20,000/mm3 had a sensitivity of 81%, specificity of 83%, and overall accuracy of 82% for differentiating the two groups (Figure 2).Regarding the distribution of the patients, 24 (92%) patients of the strangulated group were present in the area of BE level of ≤ -1.1mmol/L or ascites RBC count of ≥ 20,000/mm3.
| Category | Strangulated (n=26) | Simple (n=12) | p-value |
|---|---|---|---|
| Vital sign | |||
| BT (°C) | 36.5 (35.9-36.8) | 36.9 (36.6-37.7) | 0.073 |
| HR (b.p.m.) | 83 (67-100) | 85 (66-100) | 0.875 |
| PaCO2 (Torr) | 35.7 (33.8-39.4) | 38.0 (34.6-43.4) | 0.192 |
| sBP (mmHg) | 120 (110-138) | 140 (123-154) | 0.053 |
| Blood | |||
| WBC (× 102/mm3) | 113 (61-149) | 102 (57-136) | 0.638 |
| RBC (× 104/mm3) | 433 (403-478) | 432 (357-481) | 0.888 |
| LDH (IU) | 203 (189-272) | 204 (163-224) | 0.33 |
| CK (IU) | 60 (33-117) | 54 (34-65) | 0.499 |
| CRP (mg/dL) | 1.7 (0.2-13.3) | 1.5 (0.2-11.1) | 0.888 |
| BE (mmol/L) | -2.1 (-4.1-1.0) | 2.8 (-0.3-4.5) | 0.003 |
| Ascites | |||
| WBC (× 102/mm3) | 11.0 (4.8-107.0) | 14.0 (5.3-27.8) | 0.46 |
| RBC (× 104/mm3) | 6.5 (2.0-14.0) | 1.0 (0.3-1.0) | <0.001 |
| LDH (IU) | 330 (234-930) | 283 (264-807) | 0.74 |
| CK (IU) | 83 (35-153) | 83 (47-189) | 0.757 |
| CT findings | |||
| Strangulation sign | 19 (73) | 1 (8) | <0.001 |
| Continuous data are expressed as medians (25th to 75th percentile), and p-values were calculated using the Mann-Whitney U test. Categorical data are expressed as number (%), and p-values were calculated using the chi-squared test. BT: Body temperature, HR: Heart rate, PaCO2: Arterial partial pressure of carbon dioxide, sBP: systolic blood pressure, WBC: White blood cell, RBC: Red blood cell, LDH: Lactate dehydrogenase, CK: Creatinine phosphokinase, CRP: C-reactive protein, BE: Arterial base excess, CT: Computed tomography. | |||
Table 2: Comparison of patients with and without bowel strangulation.
Strangulation sign on CT, besides accumulation of ascites, was more frequently and significantly detected in the strangulated group than in the simple group (Table 2).Preoperative CT had a sensitivity of 73%, specificity of 92%, and overall accuracy of 79% for differentiating the two groups. Bowel resection was conducted in 17 (65%) patients of the strangulated group with the median length of 80 (58-138) cm resected necrotic bowel.Two patients in the strangulated group died of septic shock, and one patient in the simple group died of acute deterioration of basal chronic heart failure. However, no postoperative morbidity was found in the other 35 patients.
Discussion
Differential diagnosis of strangulated and simple SBO is a frequently contended issue, and many authors [2-6] have investigated the optimal diagnostic parameters including intensity of abdominal pain, presence of signs of peritoneal irritation, vital signs, presence of systemic inflammatory response syndrome, and blood test results (leukocytosis, elevated levels of LDH, CK, and CRP, and metabolic acidosis). However, it remains controversial, and a definitive consensus on a proper differentiation method has not been established. On the other hand, many authors [3,6-15] have reported the usefulness of CT in diagnosing strangulation, and accumulation of ascites, mesenteric fluid, bowel wall thickening, closed loop sign, whirl sign, conversion of the mesenteries, reduced bowel wall enhancement, increased CT value of fluid accumulated in the bowel lumen, and high-density intraabdominal free fluid are commonly identified as strangulation signs which are sufficient to confirm strangulation on CT.
Due to the recent advances in the CT model performance, the role of contrast-enhanced CT in diagnosing strangulation is becoming more viable. However, it has been reported that these CT findings represent the structural or reactive changes of the small bowel at a relatively late stage of strangulation [1], and although its specificity is as high as 90%-100%, sensitivity is only 70%-80% [7,8]. Therefore, although CT is sufficient for definitive diagnosis of strangulation, it may not be always effective for early diagnosis. In addition, considering disparity in physician’s ability to read CT, the diagnosis by CT tends to be the subjective judgment of the reader, as it was reported that even the judgment of the radiology specialists had an accuracy of approximately 80% for diagnosing strangulation [9,10]. Moreover, owing to the model performance differences between each CT device, as well as clinical limitations of each institution, it seems difficult to claim that CT is universally sufficient for early diagnosis of strangulation among all physicians. Because of these reasons, we investigated the optimal strategy of diagnosing strangulation using DPC and ascites analysis for the patients who cannot be definitively diagnosed by CT.
In the present study, when the BE level of -1.1 mmol/L was set as a cut-off value, strangulated and simple SBO could be well differentiated with high accuracy. Takahashi et al. [3] also reported that the mean level of BE showed -1.84 mmol/L in strangulated SBO, which was significantly lower than that in simple SBO (0.52 mmol/L). BE level can be easily and less invasively examined by arterial blood sampling only. Therefore, it was suggested that BE level may be one of the most useful predictors for diagnosing strangulation. However, it is concerned that the decrease of the BE level may depend on the length of the bowel necrosis. Actually in the present study, 9 strangulated cases showed BE levels of higher than -1.1 mmol/L, and 4 of them could avoid bowel resection because the intraoperative macroscopic bowel condition was still congestion, not yet transmural necrosis. In the other 5 patients, the length of resected necrotic bowel was as short as 25, 40, 45, 65, and 70 cm, respectively. Postoperative courses of all of the 9 patients were uneventful. Therefore, we hypothesize that the BE level has limitation for diagnosing strangulated SBO of which length of bowel necrosis is short. As an alternative predictor of metabolic acidosis, arterial lactate level is well known, and Tanaka et al. [4] reported that lactate level of higher than 2.0 mmol/L is a useful predictor of bowel necrosis. In the present study, due to the model performance limitations of the blood gas analyzer in our institution, lactate level was only examined for the 20 most recent cases. However, lactate level may still be a useful predictor for diagnosing strangulation, and should be investigated hereafter.
It has been reported that ascites appears in 75%-100% of cases of strangulated SBO; however, it also appears in 25%-50% of cases of simple SBO [7,9,11]. Therefore, it is hard to differentiate strangulated and simple SBO simply with the appearance of ascites itself. However, the property of ascites is very different between them. The mechanism of the appearance of ascites in strangulated SBO is mesenteric compression followed by venous occlusion, congestion, edema, and finally the break of intestinal microcirculation. Therefore, the ascites in strangulated SBO is often bloody [1,12,13]. On the other hand, in simple SBO, the ascites is serous because the appearance of ascites is caused by lymphatic leakage from the intestinal wall due to increased intraluminal pressure of the dilated intestine. Therefore, by investigating whether the accumulated ascites is bloody, the preoperative differentiation between strangulated and simple SBO may be possible, and DPC may be significant on this point. In an area of emergency medicine, DPC was initially introduced for the diagnosis of intestinal injury in patients of blunt abdominal trauma [16]. Kobayashi et al. [1] applied this technique for diagnosing strangulation and reported that macroscopic serosanguinous ascites and ascites RBC count above 20,000/mm3 are statistically significant for the diagnosis. Also in the present study, ascites RBC count was significantly higher in the strangulated group, and when the ascites RBC count of 20,000/mm3 was set as a cut-off value, strangulated and simple SBO could be well differentiated with high accuracy. This result was fortuitously similar to that of Kobayashi et al. [1], and the propriety of their work was confirmed. In addition, ascites RBC counts were above 20,000/mm3 in 7 of 9 strangulated cases of which BE levels were higher than -1.1 mmol/L in the present study, and thus we hypothesize that the appearance of RBC in ascites may be an early local sign of bowel vascular compromise, which may occur even when the length of bowel necrosis is short. As a result, we consider the significance and the role of DPC to be its ability to complement CT findings and BE level in diagnosing strangulation.
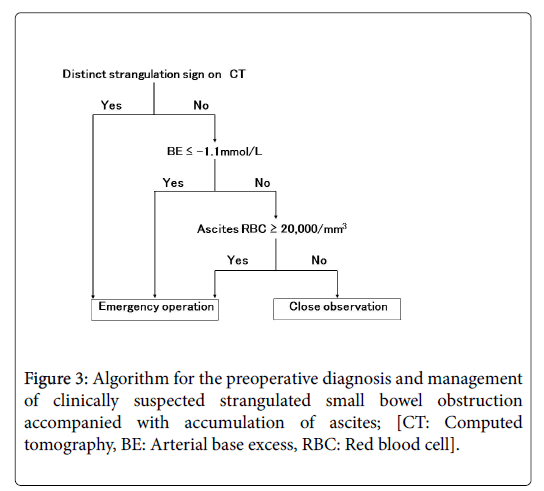
Based on the results in the present study, we attempted to create an algorithm for the preoperative diagnosis and management of clinically suspected strangulated SBO accompanied with accumulation of ascites (Figure 3). For the patients with distinct strangulation sign on CT, an emergency operation should be performed. For the patients who cannot be definitively diagnosed by CT, BE level should be examined first, and if it is below -1.1 mmol/L, emergency operation should be performed. On the other hand, if the BE level is higher than -1.1 mmol/L, DPC should be attempted subsequently, and if the ascites RBC count is above 20,000/mm3, emergency operation should be performed. However, if the ascites RBC count is lower than 20,000/ mm3, the case is most likely simple SBO; therefore, close observation with standby for emergency operation may be considered. Assuming that all of the 26 strangulated cases in the present study could not be definitively diagnosed by CT, and when they were applied to this algorithm, 24 (92%) patients were judged as requiring an emergency operation, and 2 (8%) patients (above mentioned patients who could avoid bowel resection) was judged as indication of close observation. We would like to continue the evaluation for the patients with clinically suspected strangulated SBO according to this algorithm hereafter.
The present study has several limitations as follows: 1) it was a single-center, retrospective, and observational study, 2) The obtained results were based on the small number of cases, 3) The time interval between blood gas sampling and DPC was not adjusted. A further evaluation of the present study is to investigate whether DPC may be a universal diagnostic method for diagnosing strangulation, and elevation of the evidence levels of indication, safety, other significant parameters, a cut-off value of ascites RBC count, and accuracy is essential. In order to achieve this purpose, accumulation of further cases and a multi-center prospective study may be necessary.
Conclusion
Ascites RBC count was significantly higher in the strangulated SBO than in the simple SBO, and when ascites RBC count is above 20,000/mm3, the diagnosis is most likely strangulated SBO. Our results suggest that DPC and ascites analysis, particularly ascites RBC count, are significant in the preoperative diagnosis and management of clinically suspected strangulated SBO accompanied with accumulation of ascites when combined with CT findings and BE level.
References
- Kobayashi S, Matsuura K, Matsushima K, Okubo K, Henzan E, et al. (2007) Effectiveness of diagnostic paracentesis and ascites analysis for suspected strangulation obstruction. J Gastrointest Surg 11: 240-246.
- Sarr MG, Bulkley GB, Zuidema GD (1983) Preoperative recognition of intestinal strangulation obstruction: Prospective evaluation of diagnostic capability. Am J Surg 145: 176-182.
- Takahashi R, Akagi Y, Tanaka T, Kaibara A, Kajiwara S, et al. (2014) Clinicopathological evaluation of anoxic mucosal injury in strangulation ileus. BMC Surg 14: 79.
- Tanaka K, Hanyu N, Iida T, Watanabe A, Kawano S, et al. (2012) Lactate levels in the detection of preoperative bowel strangulation. Am Surg 78: 86-88.
- Tsumura H, Ichikawa T, Hiyama E, Murakami Y, Sueda T (2004) Systemic inflammatory response syndrome (SIRS) as a predictor of strangulated small bowel obstruction. Hepatogastroenterology 51: 1393-1396.
- Huang X, Fang G, Lin J, Xu K, Shi H, et al. (2018) A prediction model for recognizing strangulated small bowel obstruction. Gastroenterol Res Pract 2018: 7164648.
- Ha HK, Kim JS, Lee MS, Lee HJ, Jeong YK, et al. (1997) Differentiation of simple and strangulated small-bowel obstructions: Usefulness of known CT criteria. Radiology 204: 507-512.
- Mallo RD, Salem L, Lalani T, Flum DR (2005) Computed tomography diagnosis of ischemia and complete obstruction in small bowel obstruction: A systematic review. J Gastrointest Surg 9: 690-694.
- Kim JH, Ha HK, Kim JK, Eun HW, Park KB, et al. (2004) Usefulness of known computed tomography and clinical criteria for diagnosing strangulation in small-bowel obstruction: Analysis of true and false interpretation groups in computed tomography. World J Surg 28: 63-68.
- Sheedy SP, Earnest F 4th, Fletcher JG, Fidler JL, Hoskin TL (2006) CT of small-bowel ischemia associated with obstruction in emergency department patients: Diagnostic performance evaluation. Radiology 241: 729-736.
- Zalcman M, Sy M, Donckier V, Closset J, Gansbeke DV (2000) Helical CT signs in the diagnosis of intestinal ischemia in small-bowel obstruction. AJR Am J Roentgenol 175: 1601-1607.
- Matsushima K, Inaba K, Dollbaum R, Cheng V, Khan M, et al. (2016) High-density free fluid on computed tomography: A predictor of surgical intervention in patients with adhesive small bowel obstruction. J Gastrointest Surg 20: 1861-1866.
- Balthazar EJ, Liebeskind ME, Macari M (1997) Intestinal ischemia in patients in whom small bowel obstruction is suspected: Evaluation of accuracy, limitations, and clinical implications of CT in diagnosis. Radiology 205: 519-522.
- Ohira G, Shuto K, Kono T, Tohma T, Gunji H, et al. (2012) Utility of arterial phase of dynamic CT for detection of intestinal ischemia associated with strangulation ileus. World J Radiol 4: 450-454.
- Millet I, Boutot D, Faget C, Pages-Bouic E, Molinari N, et al. (2017) Assessment of strangulation in adhesive small bowel obstruction on the basis of combined CT findings: Implications for clinical care. Radiology 285: 798-808.
- Sloop RD (1978) The dominant role of paracentesis technics in the early diagnosis of blunt abdominal trauma. Am J Surg 136: 145-150.
Citation: Ohtsuka Y, Takahara Y, Uno H, Nishida T, Shida T et al. (2018) Significance of Diagnostic Paracentesis in the Preoperative Diagnosis of Strangulated Small Bowel Obstruction. J Gastrointest Dig Syst 8: 568. DOI: 10.4172/2161-069X.1000568
Copyright: © 2018 Ohtsuka Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 9537
- [From(publication date): 0-2018 - Dec 10, 2025]
- Breakdown by view type
- HTML page views: 8565
- PDF downloads: 972