Review Article Open Access
Urine Formaldehyde: A Non-Invasive Marker for Alzheimer's Disease?
Zhiqian Tong*Alzheimer’s Disease Center, Beijing Institute for Brain Disorders, Capital Medical University, Beijing 100069, China
- Corresponding Author:
- Zhiqian Tong
Alzheimer’s Disease Center
Beijing Institute for Brain Disorders
Capital Medical University, Beijing 100069, China
Tel: +861083911987
E-mail: tzqbeida@ccmu.edu.cn
Received date: June 26, 2017; Accepted date: June 28, 2017; Published date: July 05, 2017
Citation: Tong Z (2017) Urine Formaldehyde: A Non-Invasive Marker for Alzheimer’s Disease? J Alzheimers Dis Parkinsonism 7:345. doi:10.4172/2161- 0460.1000345
Copyright: © 2017 Tong Z. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Alzheimers Disease & Parkinsonism
Abstract
Given the dramatic increase in Alzheimer’s disease (AD) cases globally, the identification of a suitable biomarker in easily collectable samples (e.g. plasma, blood, saliva and urine) for diagnosing AD is therefore of utmost importance. Previous studies indicated that excess formaldehyde contributes to Aβ aggregation and Tau hyper phosphorylation, both phenomena directly linked to the progress of AD. Our 7 year’s cross-sectional survey showed that morning urine formaldehyde levels were correlated positively with the severe degree of sporadic dementia, suggesting that urine formaldehyde measurement most likely acts as a suitable non-invasive method to support diagnostic purposes. In this a short review article, we provide a short overview of the animal and clinical studies on the possible mechanisms of exogenous and endogenous factors cause formaldehyde accumulation, which plays a critical role in the pathogenesis of both genetic dementia and sporadic dementia. Urine formaldehyde will be of significant value for the non-invasive diagnosis of cognitive ability in AD, but the more sensitive method for detecting formaldehyde concentrations and a longitudinal (long-term follow-up) study would be required to prove conclusively such a relationship between urine formaldehyde and dementia.
Keywords
Urine formaldehyde; Genetic dementia; Sporadic dementia; Alzheimer’s disease; Longitudinal study; Cross-sectional survey
Urine Formaldehyde Predicts Cognitive Decline in Patients with AD and Stroke
Although Alzheimer’s disease (AD) characterized by progressive deterioration in cognitive function was first described over 100 years ago, there is still no suitable biomarker for diagnosing AD in easily collectable samples (e.g. blood plasma, saliva and urine). Stroke patients often suffer from post-stroke cognitive impairment, or even from post-stroke dementia (PSD) [1]. Recently, excess formaldehyde accumulated in the hippocampi has been found in both AD patients and several transgenic AD-like animal models [2]. Surprisingly, excess formaldehyde not only induces the aggregation of Aβ and Tau proteins in vitro [3,4] and in vivo [5,6], but also leads to vascular damage in patients with stroke, AD [3,7] and PSD [8-10]. These data strongly suggest that accumulated formaldehyde plays a critical role in cognitive decline.
Urine levels of formaldehyde had been found to be inversely correlated with cognition in healthy aging individuals [11] and patients with dementia [2]. A cross-sectional survey for 7 years was carried out in China [12]. The cognitive abilities of the participants (n=577) were assessed by Mini Mental State Examination (MMSE). Morning urine formaldehyde concentrations were measured by high performance liquid chromatography with a fluorescence detector (Fluo-HPLC). The results showed that the optimal threshold of the level of morning urine formaldehyde as a predictive concentration for cognitive impairment was approximately 0.042 mM and formaldehyde levels in healthy control are about 0.02 mM. The findings suggest that morning urine formaldehyde may be a non-invasive marker for AD. More importantly, a non-invasive test which utilizes urine for analysis might potentially help primary-care physicians advice their patients on their long-term prognosis [13].
Remarkably, urine formaldehyde levels were also abnormally elevated in the patients with stroke, PSD [12], post-operative cognitive dysfunction (POCD) [14], multiple sclerosis (MS) [15] and AD [2], when their cognitions were in the process of deterioration. This phenomenon seemly affects that urine formaldehyde acts as a marker in AD. However, these mentioned above diseases have some identifiable clinical characters compared with AD and can be easily diagnosed by clinical doctors. Moreover, the main pathological feature of AD is progressive cognitive decline, urine formaldehyde was found to be inversely correlated with the scores of the Mini-Mental State Examination (MMSE), but neither the Activities of Daily Living (ADL) nor Clinical Dementia Rating (CDR) [2], therefore, urine formaldehyde still could act as a non-invasive marker for AD.
Exogenous and Endogenous Factors Induce Formaldehyde Accumulation
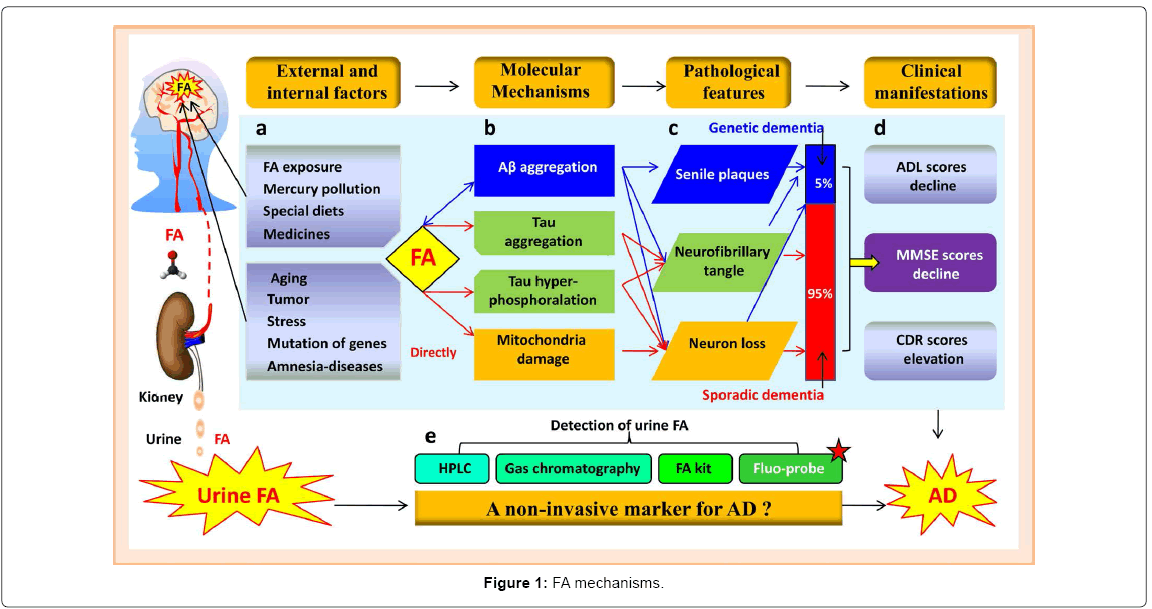
Although the physiological and pathological functions of endogenous formaldehyde in the brains are not clear until now, formaldehyde is present in the cytoplasm and nucleus of all cells of biological organism [16-19]. Using fluorescence-HPLC, we found that levels of brain formaldehyde are about 0.2~0.4 mM, similar to levels previously reported using gas chromatography/mass spectrometry [20]. Catalysis of the conversion of formaldehyde to formate via class III alcohol dehydrogenase (ADH3) and aldehyde dehydrogenase 2 (ALDH2) known as takes place in all tissues of the human body as a consequence of the regulation of endogenous formaldehyde [20]. Decades of research have established that both exogenous and endogenous factors can induce formaldehyde accumulation and lead to cognitive deficits (Figure 1(a)) [21].
Exogenous factors and Formaldehyde 1) Formaldehyde exposure. Epidemiological investigations indicate that exogenous formaldehyde exposure causes human cognitive decline and is associated with neurofilament protein changes and neuron demyelization [22-24]. 2) Mercury pollution. Environmental mercury, which some believe is a pathogenic factor for Alzheimer's disease [25], induces formaldehyde accumulation in vivo [26]. 3) Special diets. Formaldehyde participates in the “one-carbon cycle” [17], deficiencies of vitamin B12 or folate in the diet lead to dysfunction of one-carbon metabolism in Alzheimer's patients [27]. 4) Medicines. Endogenous formaldehyde is produced by microsomal cytochrome P-450 and is dependent upon oxidation of xenobiotics, including various drugs and environmental pollutants [28]. For example, abuse of formaldehyde-laced marijuana induces a high occurrence of dysmnesia [29].
Endogenous factors and Formaldehyde 1) Aging. Recent study shows that DNA demethylation leads to formaldehyde generation [30,31]. During aging, a decrease in global hippocampal 5-mC level [32] and an increase in 5hmC content have been observed in hippocampus [33]. Consistently, a wide global DNA demethylation associated with an abnormal high level of formaldehyde was found in autopsy samples from AD patients [34-36]. 2) Tumor. Cancer is known to be related to the occurrence of Alzheimer's disease [37] and cancer cells and tumor tissues release higher levels of formaldehyde than normal cells and tissues [38]. 3) Stress. Formaldehyde is also generated by lipid peroxidation (LPO) and oxidation stress [39,40], which may affect AD pathogenesis. 4) Mutations of formaldehyde metabolismrelated genes. Activities of semicarbazide-sensitive amine oxidase (SSAO, a blood formaldehyde-generating enzyme) are elevated in aged rats, as well as in patients suffering from AD [41-43]. Knockout of ALDH2 (a formaldehyde-degrading enzyme) induces memory loss and neurodegenerative disease [44]. In addition, ADH3 (a specific formaldehyde-degrading enzyme [45]) can defense neurodegenerative processes [46-48]. Knockout of ADH3 in Drosophila results in loss of visual memory [49]. 5) Aβ-mediated formaldehyde accumulation. Notably, excess formaldehyde was observed in APP-transgenic mice when memory started to decline on month 6 [50]. Aβ can inhibit alcohol-degrading enzyme activity [51], suggesting that inactivity of ADH3 leads to formaldehyde accumulation. This working hypothesis needs further investigation. 6) Some diseases associated with amnesia. Through clinical survey, an abnormal high level of formaldehyde has been found in the patients with stroke, PSD, POCD, MS [15] and AD [52]. These exogenous and endogenous factors play roles in formaldehyde accumulation and cognitive decline (Figure1(b)).
Excess Formaldehyde Promotes the Occurrence of both Genetic and Sporadic Dementia
Substantial clinical surveys of all kinds of dementia have shown that the occurrence of genetic dementia (which is closely related with mutations of APP and/or PSI) is only 5%, but 95% are a variety of pathological factors-mediated sporadic dementia, which is the most common form of AD and is not attributed to genetics (Figures 1(c) and 1(d)). Whether dominantly inherited variants of Alzheimer disease (AD) and ‘sporadic’ forms exhibit similar pathophysiological and biomarker signatures remains unresolved [53]. However, in our previous studies, there were abnormal high levels of formaldehyde in patients with these two kinds of dementia [2] and indeed caused memory deficits in mice and dementia in patients.
Formaldehyde and genetic dementia Increases in brain formaldehyde observed occurred at a similar time (from as early as 3 months of age) to the development of abnormal LTP levels in APP/ PS1 transgenic mice [54], Interestingly, reduced LTP parallels plaque appearance and increased Aβ levels and abnormal short-term memory (working memory). In APP-transgenic mice, brain formaldehyde also increased at the early stage (6 months old) in which Aβ starts to deposit in the brain, and typical senile plaques were detected in the brain of these two types of AD model [54,55]. A potential mechanism is that accumulated formaldehyde induces the aggregation of amyloid (Aβ) [3,4], as well as the hyperphosphorylation and aggregation of Tau proteins [5]. In return, Aβ can inhibit the activity of alcohol dehydrogenase [52], suggesting that inactivity of ADH3 leads to formaldehyde accumulation. In addition, a natural formaldehyde capturer [56], resveratrol, provides effective defence against cancer and neurodegenerative disease including AD [57,58]. Recent research has also shown that dietary supplementation with resveratrol reduces senile plaque (SP) pathology in a APP/PS1-transgenic model of AD [55]. These data indicate that excessive formaldehyde is involved in the pathogenesis of genetic dementia.
Formaldehyde and sporadic dementia our clinical survey showed that urine formaldehyde concentrations were markedly elevated in patients with sporadic dementia (not including genetic dementia) than healthy age-matched control [12]. The same results also were observed in the different kinds of animal models with sporadic dementia. For example, an abnormal high level of formaldehyde accumulates in the brains of SAMP8 mice (a sporadic age-related dementia model) and in the autopsied hippocampi from patients with genetic dementia-AD [2]. Another sporadic model of AD has been established by only knockout of formaldehyde-degrading enzyme gene-ALDH2 [59]. These ALDH2- /- mice are considered as an age-related model of cognitive deficits [60] and associated with AD-like pathologies including Tau hyperphosphorylation, brain atrophy and cognitive deficits [60]. Individuals carrying ALDH2 variants are vulnerable to neural damage and to develop AD [61]. These data indicate that excess formaldehyde can induce sporadic dementia.
Furthermore, we found that injection of formaldehyde into hippocampus can mimic the damage effects of excess formaldehyde, which directly induces neurons death and memory deficits [62]. Similarly, exposure of rats to gaseous formaldehyde causes formaldehyde accumulation [63], decreases the number of hippocampal neurons [64] and leads to memory decline [65]. These diverse lines of evidence indicate that excess formaldehyde in the brains leads to genetic dementia and sporadic dementia (Figure 1(b)).
More Sensitive Method for Detecting Urine Formaldehyde is required for Clinical Diagnosis
Interestingly, urine formaldehyde rather than blood formaldehyde is more suitable for reflecting the metabolism of endogenous formaldehyde in vivo [12]. As active formaldehyde is prone to react with serum proteins, blood formaldehyde (about 0.08 mM) are more stable than urine formaldehyde levels from healthy adult (about 0.02 mM). Previous study indicated that blood have an the equilibrium of reactions involving the one-carbon sources, therefore, blood formaldehyde concentrations are often relatively stable [66]. It's worth noting that urine contains very little residual proteins [12]. To rule out of the urine proteins interfering with the fluorescence signal of the formaldehydederivative by using Fluo-HPLC, a high speed of centrifugation of all urine samples (8,000 xg) was necessary for the precipitation of urine proteins from the supernatants [12].
Using this method of Fluo-HPLC, urine formaldehyde levels were detected in the range from 0.009 to 0.565 mM, which is consistent with previously reported data [67]. Although these existing approaches provide accurate and ultrasensitive assays for formaldehyde detection in biological samples, several disadvantages such as expense, sophisticated experimental procedures, and noxious analytical reagents, have limited their practical applications. Therefore, a simple, sensitive and efficient method for determining trace amounts of FA in biological samples is needed. Notably, recent years, some fluorescence probes for detecting formaldehyde have been established (Figure 1(e)) [68-73], these simple and quick methods of Fluo-probes should be used to measure urine formaldehyde in the further study.
Conclusion
The abnormally accumulation of endogenous formaldehyde is important for elucidating the mechanism of cognitive decline in the pathogenesis of genetic dementia and sporadic dementia. Excess formaldehyde enhances Aβ aggregation in genetic dementia, and directly induces neuron loss by damaging mitochondria in sporadic dementia. Tau hyerphosphoralation-induced by formaldehyde may be the common pathway of these two kinds of dementia (Figures 1(a)- 1(c)). Evaluation of endogenous formaldehyde in the urine has potential for use as a non-invasive and convenient method for investigation and diagnosis of dementia (Figures 1(d) and 1(e)). Particularly wish to point out that a longitudinal (long-term follow-up) study is very urgently required to prove conclusively such a relationship between urine formaldehyde and dementia. This finding also raises the possibility that urine formaldehyde could be used as a non-invasive marker for the assessment of therapeutics of AD.
References
- Pendlebury ST, Rothwell PM (2009) Prevalence, incidence and factors associated with pre-stroke and post-stroke dementia: A systematic review and meta-analysis. Lancet Neurol 8: 1006-1018.
- Tong ZQ, Zhang J, Luo WH, Wang WS, Li FX, et al. (2011) Urine formaldehyde level is inversely correlated to mini mental state examination scores in senile dementia. Neurobiol Aging 32: 31-41.
- Chen K, Kazachkov M, Yu PH (2007) Effect of aldehydes derived from oxidative deamination and oxidative stress on beta-amyloid aggregation: Pathological implications to Alzheimer's disease. J Neural Transm 114: 835-839.
- Kazachkov M, Chen K, Babiy S, Yu PH (2007) Evidence for in vivo scavenging by aminoguanidine of formaldehyde produced via semicarbazide-sensitive amine oxidase-mediated deamination. J Pharmacol Exp Ther 322: 1201-1207.
- Lu J, Miao J, Su T, Liu Y, He R (2013) Formaldehyde induces hyperphosphorylation and polymerization of Tau protein both in vitro and in vivo. Biochim Biophys Acta 1830: 4102-4116.
- Nie CL, Wang Y, Chen X, Liu YY, Dui W, et al. (2007) Formaldehyde at low concentration induces protein tau into globular amyloid-like aggregates in vitro and in vivo. PLoS ONE 2: e629.
- Yu PH, Deng YL (1998) Endogenous formaldehyde as a potential factor of vulnerability of atherosclerosis: Involvement of semicarbazide-sensitive amine oxidase-mediated methylamine turnover. Atherosclerosis 140: 357-363.
- Deng Y, Yu PH (1999) Simultaneous determination of formaldehyde and methylglyoxal in urine: Involvement of semicarbazide-sensitive amine oxidase-mediated deamination in diabetic complications. J Chromatogr Sci 37: 317-322.
- Gubisne-Haberle D, Hill W, Kazachkov M, Richardson JS, Yu PH (2004) Protein cross-linkage induced by formaldehyde derived from semicarbazide-sensitive amine oxidase-mediated deamination of methylamine. J Pharmacol Exp Ther 310: 1125-1132.
- Hernandez-Guillamon M, Solé M, Delgado P, García-Bonilla L, Giralt D, et al. (2012) VAP-1/SSAO plasma activity and brain expression in human hemorrhagic stroke. Cerebrovasc Dis 33: 55-63.
- Yu J, Su T, Zhou T, He Y, Lu J, et al. (2014) Uric formaldehyde levels are negatively correlated with cognitive abilities in healthy older adults. Neurosci Bull 30: 172-184.
- Tong Z, Wang W, Luo W, Lv J, et al. (2017) Urine formaldehyde predicts cognitive impairment in post-stroke dementia and Alzheimer's disease. J Alzheimers Dis 55: 1031-1038.
- Butcher J (2007) Urine tests for Alzheimer's disease--Are they fool's gold? Lancet Neurol 6: 106-107.
- Wang J, Su T, Liu Y, Yue Y, He R (2012) Postoperative cognitive dysfunction is correlated with urine formaldehyde in elderly non-cardiac surgical patients. Neurochem Res 37: 2125-2134.
- Khokhlov AP, Zavalishin IA, Savchenko Iu N, Dziuba AN (1989) Disorders of formaldehyde metabolism and its metabolic precursors in patients with multiple sclerosis. Zh Nevropatol Psikhiatr Im S S Korsakova 89: 45-48.
- Kalapos MP (1999) A possible evolutionary role of formaldehyde. Exp Mol Med 31: 1-4.
- Kalász H (2003) Biological role of formaldehyde, and cycles related to methylation, demethylation and formaldehyde production. Mini Rev Med Chem 3: 175-192.
- Trézl L, Csiba A, Juhasz S, Szentgyörgyi M, Lombai G, et al. (1997) Endogenous formaldehyde level of foods and its biological significance. Zeitschrift für Lebensmitteluntersuchung und -Forschung A 205: 300-304.
- Tyihák E, Albert L, Németh ZI, Kátay G, Király-Véghely Z, et al. (1998) Formaldehyde cycle and the natural formaldehyde generators and capturers. Acta biologica Hungarica 49: 225-238.
- Heck H, White EL, Casanova-Schmitz M (1982) Determination of formaldehyde in biological tissues by gas chromatography/mass spectrometry. Biomed Mass Spectrom 9: 347-353.
- Tong Z, Han C, Luo W, Wang X, Li H, et al. (2012) Accumulated hippocampal formaldehyde induces age-dependent memory decline. AGE 35: 583-596.
- Kilburn KH (1994) Neurobehavioral impairment and seizures from formaldehyde. Arch Environ Health 49: 37-44.
- Kilburn KH, Warshaw R, Thornton JC (1987) Formaldehyde impairs memory, equilibrium and dexterity in histology technicians: Effects which persist for days after exposure. Arch Environ Health 42: 117-120.
- Perna RB, Bordini EJ, Deinzer-Lifrak M (2001) A case of claimed persistent neuropsychological sequelae of chronic formaldehyde exposure: Clinical, psychometric and functional findings. Arch Clin Neuropsychol 16: 33-44.
- Ely J (2001) Mercury induced Alzheimer's disease: Accelerating incidence? Bull Environ Contam Toxicol 67: 800-806.
- Rétfalvi T, Németh ZI, Sarudi I, Albert L (1998) Alteration of endogenous formaldehyde level following mercury accumulation in different pig tissues. Acta Biol Hung 49: 375-379.
- Coppedè F (2010) One-carbon metabolism and Alzheimer's disease: Focus on epigenetics. Curr Genomics 11: 246-260.
- Retfalvi T, Nemeth ZI, Sarudi I, Albert L (1998) Alteration of endogenous formaldehyde level following mercury accumulation in different pig tissues. Acta Biol Hung 49: 375-379.
- Marceaux JC, Dilks LS, Hixson S (2008) Neuropsychological effects of formaldehyde use. J Psychoactive Drugs 40: 207-210.
- Patra SK, Patra A, Rizzi F, Ghosh TC, Bettuzzi S (2008) Demethylation of (Cytosine-5-C-methyl) DNA and regulation of transcription in the epigenetic pathways of cancer development. Cancer Metastasis Rev 27: 315-334.
- Wu SC, Zhang Y (2010) Active DNA demethylation: Many roads lead to Rome. Nat Rev Mol Cell Biol 11: 607-620.
- Igor PP, Vanyushin BF (2010) Age-related genomic hypomethylation. Epigenetics of Aging 11-27.
- Chen H, Dzitoyeva S, Manev H (2012) Effect of aging on 5-hydroxymethylcytosine in the mouse hippocampus. Restor Neurol Neurosci 30: 237-245.
- Mastroeni D, Grover A, Delvaux E, Whiteside C, Coleman PD, et al. (2010) Epigenetic changes in Alzheimer's disease: Decrements in DNA methylation. Neurobiol Aging 31: 2025-2037.
- Coppieters N, Dieriks BV, Lill C, Faull RL, Curtis MA, et al. (2014) Global changes in DNA methylation and hydroxymethylation in Alzheimer's disease human brain. Neurobiol Aging 35: 1334-1344.
- Tong Z, Han C, Qiang M, Wang W, Lv J, et al. (2015) Age-related formaldehyde interferes with DNA methyltransferase function, causing memory loss in Alzheimer's disease. Neurobiol Aging 36: 100-110.
- Burke WJ, McLaughlin JR, Chung HD, Gillespie KN, Grossberg GT, et al. (1994) Occurrence of cancer in Alzheimer and elderly control patients: An epidemiologic necropsy study. Alzheimer Dis Assoc Disord 8: 22-28.
- Tong Z, Luo W, Wang Y, Jiang Q, Han Y, et al. (2010) Tumor tissue-derived formaldehyde and acidic microenvironment synergistically induce bone cancer pain. PLoS ONE 5: e10234.
- Yu PH, Lai CT, Zuo DM (1997) Formation of formaldehyde from adrenaline in vivo: A potential risk factor for stress-related angiopathy. Neurochem Res 22: 615-620.
- Kang JE, Cirrito JR, Dong H, Csernansky JG, Holtzman DM (2007) Acute stress increases interstitial fluid amyloid-beta via corticotropin-releasing factor and neuronal activity. Proc Natl Acad Sci U S A 104: 10673-10678.
- Ferrer I, Lizcano JM, Hernandez M, Unzeta M (2002) Overexpression of semicarbazide sensitive amine oxidase in the cerebral blood vessels in patients with Alzheimer's disease and cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy. Neurosci Lett 321: 21-24.
- del Mar Hernandez M, Esteban M, Szabo P, Boada M, Unzeta M (2005) Human plasma semicarbazide sensitive amine oxidase (SSAO), beta-amyloid protein and aging. Neurosci Lett 384: 183-187.
- Mercier N, El Hadri K, Osborne-Pellegrin M, Nehme J, Perret C, et al. (2007) Modifications of arterial phenotype in response to amine oxidase inhibition by semicarbazide. Hypertension 50: 234-241.
- Ohta S, Ohsawa I (2006) Dysfunction of mitochondria and oxidative stress in the pathogenesis of Alzheimer's disease: On defects in the cytochrome C oxidase complex and aldehyde detoxification. J Alzheimers Dis 9: 155-166.
- Cinti DL, Keyes SR, Lemelin MA, Denk H, Schenkman JB (1976) Biochemical properties of rat liver mitochondrial aldehyde dehydrogenase with respect to oxidation of formaldehyde. J Biol Chem 251: 1571-1577.
- Jelski W, Sani TA, Szmitkowski M (2006) [Class III alcohol dehydrogenase and its role in the human body]. Postepy Hig Med Dosw (Online) 60: 406-409.
- Mori O, Haseba T, Kameyama K, Shimizu H, Kudoh M, et al. (2000) Histological distribution of class III alcohol dehydrogenase in human brain. Brain Res 852: 186-190.
- Galter D, Carmine A, Buervenich S, Duester G, Olson L (2003) Distribution of class I, III and IV alcohol dehydrogenase mRNAs in the adult rat, mouse and human brain. Eur J Biochem 270: 1316-1326.
- Hou Q, Jiang H, Zhang X, Guo C, Huang B, et al. (2011) Nitric oxide metabolism controlled by formaldehyde dehydrogenase (FDH, homolog of mammalian GSNOR) plays a crucial role in visual pattern memory in Drosophila. Nitric Oxide 24: 17-24.
- Tong Z, Zhang J, Luo W, Wang W, Li F, et al. (2011) Urine formaldehyde level is inversely correlated to mini mental state examination scores in senile dementia. Neurobiol Aging 32: 31-41.
- Lustbader JW, Cirilli M, Lin C, Xu HW, Takuma K, et al. (2004) ABAD directly links Abeta to mitochondrial toxicity in Alzheimer's disease. Science 304: 448-452.
- Khokhlov AP, Zavalishin IA, Savchenko IN, Dziuba AN (1989) Disorders of formaldehyde metabolism and its metabolic precursors in patients with multiple sclerosis. Zh Nevropatol Psikhiatr Im SS Korsakova 89: 45-48.
- Hampel H, Lista S (2012) Alzheimer disease: from inherited to sporadic AD-crossing the biomarker bridge. Nat Rev Neurol 8: 598-600.
- Trinchese F, Liu S, Battaglia F, Walter S, Mathews PM, et al. (2004) Progressive age-related development of Alzheimer-like pathology in APP/PS 1 mice. Ann Neurol 55: 801-814.
- Karuppagounder SS, Pinto JT, Xu H, Chen HL, Beal MF, et al. (2009) Dietary supplementation with resveratrol reduces plaque pathology in a transgenic model of Alzheimer's disease. Neurochem Int 54: 111-118.
- Tyihák E, Albert L, Németh ZI, Kátay G, Király-Véghely Z, et al. (1998) Formaldehyde cycle and the natural formaldehyde generators and capturers. Acta Biol Hung 49: 225-238.
- Saiko P, Szakmary A, Jaeger W, Szekeres T (2008) Resveratrol and its analogs: defense against cancer, coronary disease and neurodegenerative maladies or just a fad? Mutat Res 658: 68-94.
- Rocha-González HI, Ambriz-Tututi M, Granados-Soto V (2008) Resveratrol: A natural compound with pharmacological potential in neurodegenerative diseases. CNS Neurosci Ther 14: 234-247.
- Luo J, Lee SH, VandeVrede L, Qin Z, Aissa MB, et al. (2016) A multifunctional therapeutic approach to disease modification in multiple familial mouse models and a novel sporadic model of Alzheimer's disease. Mol Neurodegener 11: 35.
- D'Souza Y, Elharram A, Soon-Shiong R, Andrew RD, Bennett BM (2015) Characterization of Aldh2 (-/-) mice as an age-related model of cognitive impairment and Alzheimer's disease. Mol Brain 8: 27.
- Kamino K, Nagasaka K, Imagawa M, Yamamoto H, Yoneda H, et al. (2000) Deficiency in mitochondrial aldehyde dehydrogenase increases the risk for late-onset Alzheimer's disease in the Japanese population. Biochem Biophys Res Commun 273: 192-196.
- Tong Z, Han C, Luo W, Wang X, Li H, et al. (2013) Accumulated hippocampal formaldehyde induces age-dependent memory decline. Age (Dordr) 35: 583-596.
- Cui X (1996) Inhaled formaldehyde on the effects of GSH level and distribution of formaldehyde. China J PrevMed 3: 186.
- Gurel A, Coskun O, Armutcu F, Kanter M, Ozen OA (2005) Vitamin E against oxidative damage caused by formaldehyde in frontal cortex and hippocampus: Biochemical and histological studies. J Chem Neuroanat 29: 173-178.
- Malek FA, Möritz KU, Fanghänel J (2003) A study on the effect of inhalative formaldehyde exposure on water labyrinth test performance in rats. Ann Anat 185: 277-285.
- Conaway CC, Whysner J, Verna LK, Williams GM (1996) Formaldehyde mechanistic data and risk assessment: Endogenous protection from DNA adducts formation. Pharmacol Ther 71: 29-55.
- Szarvas SE, Volford J, Trézl L, Tyihák E, Rusznák I (1986) Determination of endogenous formaldehyde level in human blood and urine by dimedone-14C radiometric method. J Radioanal Nucl Chem 106: 357-367.
- Brewer TF, Chang CJ (2015) An Aza-Cope reactivity-based fluorescent probe for imaging formaldehyde in living cells. J Am Chem Soc 137: 10886-10889.
- Roth A, Li H, Anorma C, Chan J (2015) A reaction-based fluorescent probe for imaging of formaldehyde in living cells. J Am Chem Soc 137: 10890-10893.
- He L, Yang X, Liu Y, Kong X, Lin W (2016) A ratiometric fluorescent formaldehyde probe for bioimaging applications. Chem Commun 52: 4029-4032.
- Tang Y, Kong X, An Xu, Baoli Dong, Weiying Lin (2016) Development of a two-photon fluorescent probe for imaging of endogenous formaldehyde in living tissues. Angew Chem Int Ed Engl 55: 3356-3359.
- He L, Yang X, Ren M, Kong X, Liu Y, et al. (2016) An ultra-fast illuminating fluorescent probe for monitoring formaldehyde in living cells, shiitake mushrooms and indoors. Chem. Commun 52: 9582-9585.
- Bruemmer KJ, Brewer TF, Chang CJ (2017) Fluorescent probes for imaging formaldehyde in biological systems. Curr Opin Chem Biol 39: 17-23.
Relevant Topics
- Advanced Parkinson Treatment
- Advances in Alzheimers Therapy
- Alzheimers Medicine
- Alzheimers Products & Market Analysis
- Alzheimers Symptoms
- Degenerative Disorders
- Diagnostic Alzheimer
- Parkinson
- Parkinsonism Diagnosis
- Parkinsonism Gene Therapy
- Parkinsonism Stages and Treatment
- Stem cell Treatment Parkinson
Recommended Journals
Article Tools
Article Usage
- Total views: 4787
- [From(publication date):
August-2017 - Aug 29, 2025] - Breakdown by view type
- HTML page views : 3803
- PDF downloads : 984