Papillary Thyroid Carcinoma with Prominent Hobnail Features Diagnosed Preoperatively by Fine Needle Aspiration and Demonstrating Constitutive Activation of mTOR Signaling Pathway: A Case Report
Received: 23-Oct-2013 / Accepted Date: 28-Nov-2013 / Published Date: 02-Dec-2013 DOI: 10.4172/2161-0681.1000152
Abstract
Papillary Thyroid Carcinoma (PTC) with prominent hobnail features is a recently recognized aggressive variant of PTC. The cytologic features and molecular characterization have not been fully investigated. We present a patient with a preoperative fine needle aspiration diagnosis of PTC with hobnail features that demonstrated constitutive activation of mTOR pathway with evidence of mTORC2 activation on immunohistochemical staining. These findings may be of value for clinical management, targeted therapy, and prognostication.
Keywords: Cytopathology, Fine needle aspiration, Hobnail variant, Immunohistochemistry, mTOR, Papillary thyroid carcinoma
309476Introduction
The hobnail variant of Papillary Thyroid Carcinoma (PTC) was recently recognized and named. It is a rare, moderately differentiated, aggressive variant of PTC. Asioli et al. 2010 [1] reported a mortality rate of up to 50%, which is significantly higher than the mortality rate of patients with conventional PTC. Besides the nuclear features of conventional PTC on histology, the tumor cells in the hobnail variant have characteristic features with the nucleus located in the apex of cytoplasm. The hobnail features usually appears with micropapillary growth pattern so this type of tumor is also called the hobnail/micropapillary variant [1-3]. The definition of this variant originally required that at least 30% of the tumor cells had hobnail features. However, a later study by the same group of pathologists showed that PTCs with even a smaller percentage of cells with hobnail features could be associated with an aggressive behavior [3]. To date, cytologic features of Fine Needle Aspiration (FNA) and molecular characterization of this tumor have not been fully investigated.
Among cancer signaling pathways, the mammalian Target of Rapamycin (mTOR) pathway has been increasingly studied because mTOR inhibitors as potential pharmaceutical agents have recently been identified and utilized in clinical or preclinical studies [4-6].
Here we present a case of papillary thyroid carcinoma with prominent hobnail features preoperatively diagnosed by FNA and immunostaining findings on mTOR pathway in the tumor.
Case Report
Clinical course
A 17-year-old woman presented with a palpable right thyroid mass. The patient otherwise was asymptomatic. Her thyroid peroxidase and thyroglobulin antibodies were both positive. A Fine Needle Aspiration (FNA) of the mass with immediate assessment for specimen adequacy was performed. The cytologic findings are described below. The final preoperative diagnosis was PTC with features suggestive of hobnail variant. A preoperative ultrasound examination further characterized the tumor as an oval and irregular, heterogeneous, coarsened hypoechoic and vascular mass and identified ipsilateral cervical lymphadenopathy in levels 3 and 4. The patient subsequently underwent a total thyroidectomy with central compartment nodal dissection and right neck level-2 through level-5 lymph nodal dissection. Postoperatively the patient received I131 treatment. One year after surgery, the patient was alive and well with a recurrence in a level-3 lymph node of right neck.
Cytologic findings and immunohistochemical studies
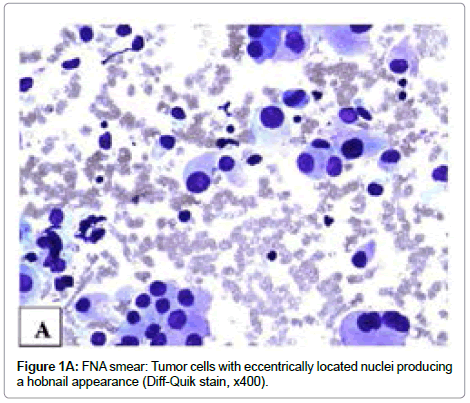
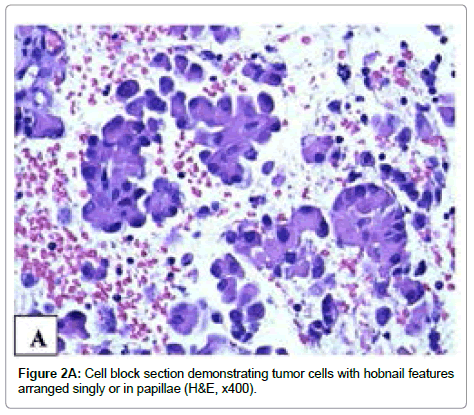
Three passes of FNA were performed using a 23-gauge needle with excellent acquisition. Smears were stained with Papanicolaou and Diff- Quick methods, respectively, and were hypercellular. The tumor cells had nuclear enlargement and moderate amount of granular cytoplasm and were arranged singly or in small to large papillary groups or follicles. The small papillary groups did not exhibit fibrovascular cores (micropapillae) while some large groups contained transversing vascular cores. More than 50% tumor cells had eccentrically located nuclei. These cells either were oval or had tailed-off cytoplasm, producing a hobnail appearance. The tumor cells covering the papillae with vascular cores showed apically placed nuclei. Intranuclear cytoplasmic inclusions were identified in some cells. On Diff-Quik stained smears, the cytoplasm was eosinophilic and granular (Figure 1A). On Papanicolaou stained smears, the nuclei were pale and powdery with small nucleoli and some cells had an irregular nuclear border and nuclear grooves (Figure 1B). The similar cytomorphology was present on H&E stained cell block sections (Figure 2A). Immunohistochemical stains performed on unstained cell block sections demonstrated that the tumor cells were diffusely positive for TTF-1 and thyroglobulin and focally positive for HBME-1. On the basis of the findings, a diagnosis of papillary thyroid carcinoma with features suggestive of hobnail variant was made preoperatively.
Gross and histological findings on surgical specimens
The specimens were composed of a total thyroidectomy with central compartment and right neck dissection specimens.
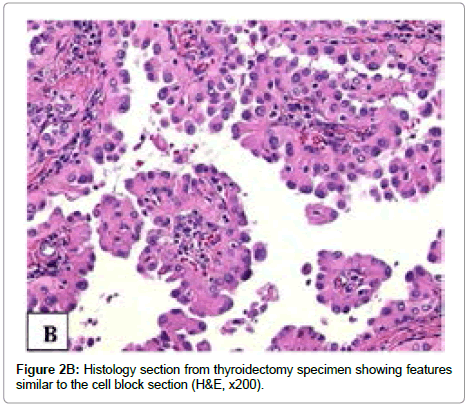
Serial sectioning the thyroid gland revealed a 3.2 × 2.0 × 1.4 cm tan-white heterogeneous well circumscribed mass extending through almost the entire right thyroid lobe. Microscopically, the tumor was present in a background of lymphocytic thyroiditis and extended into extrathyroidal tissue. The tumor showed a predominantly papillary pattern with scant psammoma bodies and focal lymphocytic infiltration, and no squamous metaplasia or significant stromal fibrosis. The tumor cells had eosinophilic granular cytoplasm, intranuclear inclusions, irregular nuclear border, and nuclear grooves. Over 70% tumor cells contained eccentrically/apically located nuclei (a hobnail appearance). The tumor cells with hobnail features were present in variably sized papillary structures with prominent vascular cores, formed micropapillae, or were singly detached from the papillae or follicles (Figure 2B). Mitoses were rare. Lymphovascular invasion was present. The carcinoma extended into perithyroidal adipose tissue and focally involved resection margin.
Metastatic carcinoma was found in 4 of 10 lymph nodes in the central compartment and 6 of 28 lymph nodes in the right neck dissection specimen. The metastatic carcinoma exhibited mixed papillary, follicular and solid patterns. The majority of tumor cells had a hobnail appearance.
Immunohistochemical study for mTOR pathway
Three primary antibodies were utilized for immunohistochemical staining to detect phosphorylated (p) antigens including p-mTOR (Ser2448), and p-Akt (Ser473) and also total IGF-1R (Tyr1165/1166) [Cell Signaling Technology, Beverly, MA and Genway Biotech, Inc, San Diego, CA, respectively]. The immunohistochemical stains were performed on formalin-fixed and paraffin-embedded unstained cell block sections of 4 μm thickness. The unstained sections were deparaffinized in xylene and rehydrated in a graded series of ethanol. Heat-induced epitope retrieval was performed. A semiautomatic method and rabbit monoclonal antibodies for p-mTOR and p-Akt and rabbit polyclonal antibody for IGF-1R were used against these antigens. Endogenous pigments were quenched with 3% H2O2 in methanol for 10 minutes and rinsed with Tris Buffered Saline with Tween 20 (TBST). A few drops of diluted normal blocking serum were placed on the tissue and incubated at room temperature. The serum was blotted off and the slides were incubated with primary antibody overnight at 4oC. The remaining procedure took place on a DAKO Autostainer programmed to treat each slide with diluted biotinylated secondary antibody for 30 minutes. The slides were rinsed and incubated with DAB (3,3’-diaminobenzidine chromogen solution, Envision+ System Kit, DAKO) for 10 minutes. The slides were rinsed again and counterstained with Gill II hematoxylin, treated with xylene, and cover-slipped. Appropriate positive and negative controls were obtained.
Chromogenic signal and subcellular expression pattern were assessed for each marker (IGF-1R, p-mTOR and p-Akt) by bright-field microscopy. The subcellular expression pattern was characterized as: nuclear, cytoplasmic, or plasmalemmal locations.
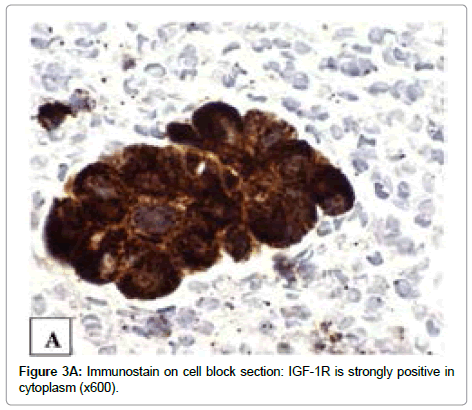
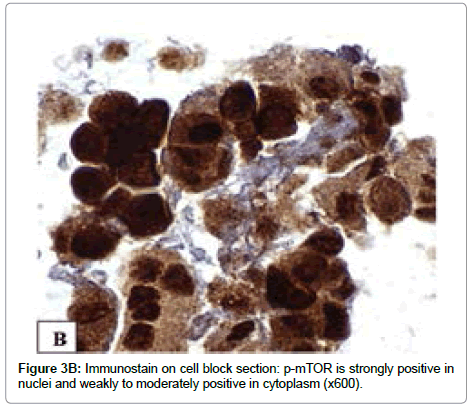
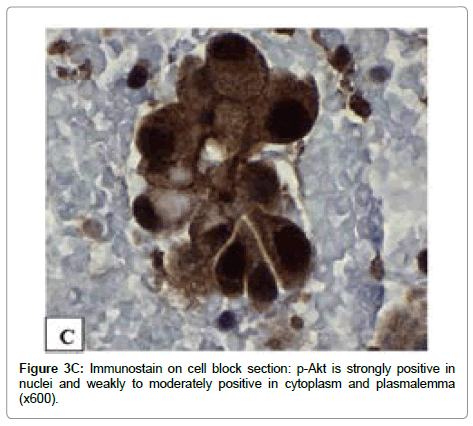
The immunoreactivity for IGF-1R (Tyr1165/1166), p-mTOR (Ser2448) and p-Akt (Ser473) is observed in all tumor cells. A strong (3+) immunoreactivity with IGF-1R (Tyr1165/1166) antibody is mainly present in cytoplasm (Figure 3A). The expression of p-mTOR and p-Akt were strong (3+) in all tumor cell nuclei, weak to moderate (1+ to 2+) in cytoplasm and focal in plasmalemma (Figures 3B and 3C).
Discussion
To our knowledge, this is the first case report of a patient whose PTC with prominent hobnail features was diagnosed preoperatively on FNA specimen, and the first report that the hobnail variant of PTC demonstrates constitutive activation of mTOR pathway.
In 2012, Bellevicine et al. [7] retrospectively reviewed the cytologic features of a PTC with prominent hobnail features. They described hobnail features as comet-like cell features and predicted that the preoperative recognition of PTC with prominent hobnail features on FNA would warrant more extensive neck surgery. Indeed, we performed a comprehensive neck dissection in our patient because of the preoperative diagnosis of PTC with hobnail features.
The hobnail appearance and micropapillary pattern in cytologic preparation are readily identified although the accurate percentage of hobnail-like cells in the entire tumor cannot be determined. Asioli S et al. 2013 [3] recently demonstrated that a PTC with even a small percentage of hobnail-like cells increased its aggressiveness while a percentage greater than 30% decreased patient survival significantly. The preoperative recognition of hobnail-like cells, therefore, provides valuable information for clinical management.
The current case has some clinical and pathologic features overlapping with those of diffuse sclerosing variant PTC, e.g., the younger age and the tumor arising from a background of lymphocytic thyroiditis. However, in contrast to the diffuse sclerosing variant PTC, this case had a distinct mass, no stromal fibrosis, no squamous metaplasia, scant psammoma bodies, and no significant lymphocytic infiltration in the tumor.
Although several prognostic clinicopathologic parameters have been proposed to predict the aggressiveness of thyroid cancers [8-10], the difference in tumorigenesis of aggressive thyroid cancers from the majority of those with an indolent behavior has not been elucidated.
The mTOR is a highly conserved protein kinase and a central controller of cell growth and metabolisms [11]. It plays an integral role in coordinating cell growth, cell proliferation, angiogenesis, and metabolism in response to growth factors, nutrients, and the energy status of the cells, promotes cell cycle progression and suppresses apoptosis in a variety of human cancers. The mTOR is found in two structurally and functionally distinct multiprotein complexes, mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2). The insulinlike growth factor (IGF)-1 receptor via insulin receptor substrate 1 (IRS-1) has been thought to be important in the upstream activation of mTOR [12]. Each of the mTORC signals via a different set of effector pathways (i.e. mTORC1 through p70S6K and mTORC2 through Akt Ser473, the latter of which is one of the upstream regulators of mTORC1 activation as well, collaborating in tumorigenesis [13,14]).
Basic research studies have shown that phosphorylation of two sites on Akt are associated with activation: one at Thr308 by PDK1 and the other at Ser473 mainly by mTORC2 [15-17]. These phosphorylation events can occur independently or interactively. Although phosphorylation of Akt at Thr308 alone empowers Akt with enough activity to phosphorylate many of its substrates, maximal Akt activity requires phosphorylation at both Thr308 and Ser473. It has been postulated that the phosphorylation of Akt at Ser473 stimulates Akt phosphorylation at Thr308 by PDK1 so that Ser473 is a critical regulatory site required for maximal Akt kinase activity [15,18].
mTORC1 contributes to cell proliferation and mTORC2 is thought to mediate cell survival. Rapamycin is an mTOR inhibitor to prevent mTOR-dependent downstream signaling transduction. mTORC1 is rapamycin sensitive while mTORC2 is relatively rapamycin insensitive. Therefore, it is important to identify the activation of mTOR pathway in tumors and recognition of mTORC2 activation is crucial. Moreover, there may be other implications of the mTORC2 activation in addition to the treatment, e.g., to study tumorigenesis, to identify aggressive tumors and to predict prognosis.
Our previous studies performed on thyroidectomy histology sections demonstrated constitutive activation of mTOR pathology in thyroid carcinomas with the evidence of mTORC2 activation in anaplastic thyroid carcinoma and some aggressive histological variants of PTC including tall cell, columnar cell and diffuse sclerosing variants [19,20]. The findings have provided additional information for the pathogenesis of aggressive thyroid carcinomas and targeted therapy. The activation of mTOR pathway in thyroid carcinomas is further supported by a recent study showing that treatment with metformin, an mTOR inhibitor, was associated with higher remission rate in diabetic patients with thyroid cancer [21].
In all our studies, we have used antibodies against phosphorylated (p)-mTOR (Ser2448) and p-Akt (Ser473) antigens because the phosphospecific probes directed against putative sites of activation of molecules [22]. The constitutive activation of mTOR pathway in the current case of PTC with prominent hobnail features is suggested by the coexpression of IGF-1R (Tyr1165/1166), p-mTOR (Ser2448) and p-Akt (Ser473). This is consistent with our previous observations of constitutive activation of mTOR signaling pathway in various types of thyroid cancers [19,20].
Rosner et al. 2010 [23] recently demonstrated that mTOR phosphorylated at Ser2448 bound to raptor and rictor, supporting the role of mTOR (Ser2448) phosphorylation in activation of molecules. Basic science studies have previously discovered that mTORC1 assembly occurred predominantly in the cytoplasm whereas mTORC2 is abundant in both cytoplasm and nucleus of non-transformed, nonimmortalized human diploid fibroblasts, Rosner and Hengstschläger 2008 [24]. Thus, the nuclear expression of p-mTOR (Ser 2448) indicates mTORC2 activation while cytoplasmic and plasmalemmal expression of p-mTOR (Ser 2448) may represent mTORC1 and/ or mTORC2 activation. The immunostaining pattern in the current case is predominantly nuclear, which is suggestive of mTORC2 overexpression.
The findings in this study are similar to those of our previous study of other aggressive variants of PTC—tall cell, columnar cell and diffuse sclerosing variants with regard to nuclear translocation of p-mTOR(Ser 2448) accompanied by a loss of plasmalemmal expression in some areas [20]. Moreover, the expression of p-Akt (Ser473) in the current case further supports the activation of mTORC2 pathway since Akt (Ser473) has been identified to be the downstream effector of mTORC2 [15,25,26].
Furthermore, expression of IGF-1R has been detected in a variety of cancers including thyroid cancers. Gydee et al. [27] found that differentiated thyroid carcinomas with expression of IGF-1R were more likely to show aggressive clinical features and to be persistent despite treatment. In our current study, we used antibody against IGF-1R (Tyr1165/1166) and demonstrated strong expression of IGF- 1R. Thus, the findings in this case support that the PTC with hobnail features is an aggressive variant.
The activation of mTORC2 pathway, conferring rapamycin resistance, has been reported in some malignant neoplasms [16]. Zakikhani et al. [12] recently observed that metformin stimulated phosphorylation of IRS-1 at Ser789 and down-regulated IGF-1R and IRS-1 levels and decreased Akt (Ser473) in breast cancer cells by in vitro assays and that maximal induction of apoptosis and inhibition of proliferation were greater for metformin than rapamycin. The findings suggest that metformin may be a treatment of choice for cancers with mTORC2 activation and rapamycin resistance. Therefore, our observation of the activation of the mTORC2 pathway in this case may be of therapeutic implications for PTC with hobnail features.
In summary, papillary thyroid carcinoma with prominent hobnail features has characteristic cytologic features. The concomitant nuclear expression p-mTOR (Ser 2448) and p-Akt (Ser473) indicates m-TORC2 activation in this tumor. This activation is probably related, in part, to downstream signaling from the IGF-1R pathway, which, at a molecular level, supports the clinical observation that papillary thyroid carcinoma with prominent hobnail features is an aggressive variant. The finding of mTOR, especially mTORC2, pathway activation in this tumor may serve as a potential therapeutic target.
Acknowledgements
The authors would like to thank Ms. Pamela K. Johnston, HT (ASCP) for her technical assistance and Ms. Bheravi Patel for secretarial and graphic design expertise.
References
- Asioli S, Erickson LA, Sebo TJ, Zhang J, Jin L, et al. (2010) Papillary thyroid carcinoma with prominent hobnail features: a new aggressive variant of moderately differentiated papillary carcinoma. A clinicopathologic, immunohistochemical, and molecular study. Am J Surg Pathol 34: 44-52.
- Motosugi U, Murata S, Nagata K, Yasuda M, Shimizu M (2009) Thyroid papillary carcinoma with micropapillary and hobnail growth pattern: a histological variant with intermediate malignancy? Thyroid 19: 535-537.
- Asioli S, Erickson LA, Righi A, Lloyd RV (2013) Papillary thyroid carcinoma with hobnail features: histopathologic criteria to predict aggressive behavior. Hum Pathol 44: 320-328.
- Galanis E, Buckner JC, Maurer MJ, Kreisberg JI, Ballman K, et al. (2005) Phase II trial of temsirolimus (CCI-779) in recurrent glioblastoma multiforme: a North Central Cancer Treatment Group Study. J Clin Oncol 23: 5294-5304.
- Agarwala SS, Case S (2010) Everolimus (RAD001) in the treatment of advanced renal cell carcinoma: a review. Oncologist 15: 236-245.
- Antonarakis ES, Carducci MA, Eisenberger MA (2010) Novel targeted therapeutics for metastatic castration-resistant prostate cancer. Cancer Lett 291: 1-13.
- Bellevicine C, Cozzolino I, Malapelle U, Zeppa P, Troncone G (2012) Cytological and molecular features of papillary thyroid carcinoma with prominent hobnail features: a case report. Acta Cytol 56: 560-564.
- Hay ID, Grant CS, Taylor WF, McConahey WM (1987) Ipsilatral versus bilateral lobar resection in papillary thyroid carcinoma: a retrospective analysis of surgical outcome using a novel prognostic scoring system. Surgery 102: 1088-1095.
- Byar DP, Green SB, Dor P, Williams ED, Colon J, et al. (1979) A prognostic index for thyroid carcinoma. A study of the E.O.R.T.C. Thyroid Cancer Cooperative Group. Eur J Cancer 15: 1033-1041.
- Cady B, Rossi R (1988) An expanded view of risk-group definition in differentiated thyroid carcinoma. Surgery 104: 947-953.
- Wullschleger S, Loewith R, Hall MN (2006) TOR signaling in growth and metabolism. Cell 124: 471-484.
- Zakikhani M, Blouin MJ, Piura E, Pollak MN (2010) Metformin and rapamycin have distinct effects on the AKT pathway and proliferation in breast cancer cells. Breast Cancer Res Treat 123: 271-279.
- Nojima H, Tokunaga C, Eguchi S, Oshiro N, Hidayat S, et al. (2003) The mammalian target of rapamycin (mTOR) partner, raptor, binds the mTOR substrates p70 S6 kinase and 4E-BP1 through their TOR signaling (TOS) motif. J Biol Chem 278: 15461-15464.
- Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307: 1098-1101.
- Sparks CA, Guertin DA (2010) Targeting mTOR: prospects for mTOR complex 2 inhibitors in cancer therapy. Oncogene 29: 3733-3744.
- Stephens L, Anderson K, Stokoe D, Erdjument-Bromage H, Painter GF, et al. (1998) Protein kinase B kinases that mediate phosphatidylinositol 3,4,5-trisphosphate-dependent activation of protein kinase B. Science 279: 710-714.
- Liu J, Brown RE (2010) Morphoproteomics demonstrates activation of mTOR pathway in anaplastic thyroid carcinoma: a preliminary observation. Ann Clin Lab Sci 40: 211-217.
- Liu J, Brown RE (2011) Morphoproteomics demonstrates activation of mammalian target of rapamycin pathway in papillary thyroid carcinomas with nuclear translocation of MTOR in aggressive histological variants. Mod Pathol 24: 1553-1559.
- Klubo-Gwiezdzinska J, Costello J Jr, Patel A, Bauer A, Jensen K, et al. (2013) Treatment with metformin is associated with higher remission rate in diabetic patients with thyroid cancer. J Clin Endocrinol Metab 98: 3269-3279.
- Brown RE (2005) Morphoproteomics: exposing protein circuitries in tumors to identify potential therapeutic targets in cancer patients. Expert Rev Proteomics 2: 337-348.
- Rosner M, Siegel N, Valli A, Fuchs C, Hengstschläger M (2010) mTOR phosphorylated at S2448 binds to raptor and rictor. Amino Acids 38: 223-228.
- Rosner M, Hengstschläger M (2008) Cytoplasmic and nuclear distribution of the protein complexes mTORC1 and mTORC2: rapamycin triggers dephosphorylation and delocalization of the mTORC2 components rictor and sin1. Hum Mol Genet 17: 2934-2948.
- Hresko RC, Mueckler M (2005) mTOR.RICTOR is the Ser473 kinase for Akt/protein kinase B in 3T3-L1 adipocytes. J Biol Chem 280: 40406-40416.
- Gydee H, O'Neill JT, Patel A, Bauer AJ, Tuttle RM, et al. (2004) Differentiated thyroid carcinomas from children and adolescents express IGF-I and the IGF-I receptor (IGF-I-R). Cancers with the most intense IGF-I-R expression may be more aggressive. Pediatr Res 55: 709-715.
Citation: Liu J, Brown RE, Rubenfeld S, Karni RJ (2013) Papillary Thyroid Carcinoma with Prominent Hobnail Features Diagnosed Preoperatively by Fine Needle Aspiration and Demonstrating Constitutive Activation of mTOR Signaling Pathway: A Case Report. J Clin Exp Pathol 4:152. DOI: 10.4172/2161-0681.1000152
Copyright: © 2013 Liu J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 18825
- [From(publication date): 1-2014 - Dec 23, 2025]
- Breakdown by view type
- HTML page views: 13835
- PDF downloads: 4990