Opinion Article Open Access
Land Use Impacts: The Effects of Non-Native Grasses on Marsh and Aquatic Ecosystems
Gulnihal Ozbay*, Kris Roeske, Lathadevi Karuna Chintapenta, Venu Kalavacharla, Matthew Stone and Laurieann PhalenDelaware State University, College of Agriculture and Related Sciences, USA
- *Corresponding Author:
- Gulnihal Ozbay
Delaware State University, College of Agriculture and Related Sciences
1200 North DuPont Highway, Dover, DE 19901 USA
Tel: 1+(302) 857 6476
E-mail: gozbay@desu.edu
Received date: November 10, 2014; Accepted date: November 10, 2014; Published date: November 17, 2014
Citation: Ozbay G, Roeske K, Chintapenta LK, Kalavacharla V, Stone M, et al.(2014) Land Use Impacts: The Effects of Non-Native Grasses on Marsh and Aquatic Ecosystems. J Ecosys Ecograph 4:151. doi:10.4172/2157-7625.1000151
Copyright: © 2014 Ozbay G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Ecosystem & Ecography
Foreword
Tidal marsh systems serve as great examples of dynamic ecosystems that can provide numerous lessons in restoration ecology. The ecosystem services which are provided by naturally functioning marsh habitats, in conjunction with the diverse assemblages of nekton which utilize those habitats, have long been underappreciated. Unfortunately, misplaced beliefs and unintentional but harmful actions persist in the face of some of the most large-scale and coordinated scientific efforts towards achieving the preservation, remediation, and reclamation of these spatially dynamic systems. An important aspect of the restoration process is to gauge the success of any management practices that have been employed. Our program focus over the past five years has been to look at the impacts of land use and land cover on aquatic health and marsh habitat. Progress towards any goals should be consistently monitored to ensure that responses to habitat management put the ecosystem on a satisfactory trajectory in terms of achieving desired structure and function.
As discussed by Dahl [1], the United States Department of Interior, in conjunction with the U.S. Fish and Wildlife Service concluded that 53% of the total wetland acreage in the continental United States has been lost over the past two centuries, with Delaware losing approximately 54% of its original wetland habitat. This loss cannot be reclaimed or fully restored to its historic structure and function in the near future, and represents the importance of persistent and ongoing management of these remaining native wetlands. Of particular concern, especially over the past several decades, is the rapid invasion of a nonnative subspecies of Phragmites australis [2]. The intrinsic value that Phragmites provides to the habitats in which it is found has often been determined from anecdotal reports of decreases in biodiversity and natural marsh function that in some areas have only recently been quantified using GIS and aerial imaging technology [3-6].
Naturally occurring variations in salt marsh topography will undoubtedly result in different flow dynamics, sedimentation rates, degrees of tidal inundation, and the availability of standing water on the marsh surface regardless of whether or not Phragmites is present [7]. However, it has been demonstrated in multiple cases that in areas where Phragmites is prevalent, the increased levels of biomass result in an altered soil chemistry, a shift in the availability of primary production, an elevated marsh surface, smoothed topography (i.e. the loss of rivulets), restricted flow to the marsh interior, an increase in shade and litter cover, and lowered or altered pathways for nutrient availability, especially nitrogen, for marsh nekton [8-12].Due to this apparent degradation of habitat quality with regard to marsh structure and function, Phragmites removal programs have been initiated with variable successes in regards to the restoration of so called pre- Phragmites biodiversity [13]. Some research has demonstrated that resident nekton displays no preference for Spartina over Phragmites in an experimental setup [14], but studies in natural habitats appear to contradict these findings [15,16].
Blackbird Creek, a largely forested watershed located in northern Delaware, is characterized by extensive salt marshes and large native populations of saltmarsh cord grass (Spartina alterniflora). It provides many recreational activities and is exposed to varying degrees of anthropogenic effects which disturb the ecosystem. Monitoring of ecosystem changes and established management practices has been ongoing since the mid-1970’s [17].
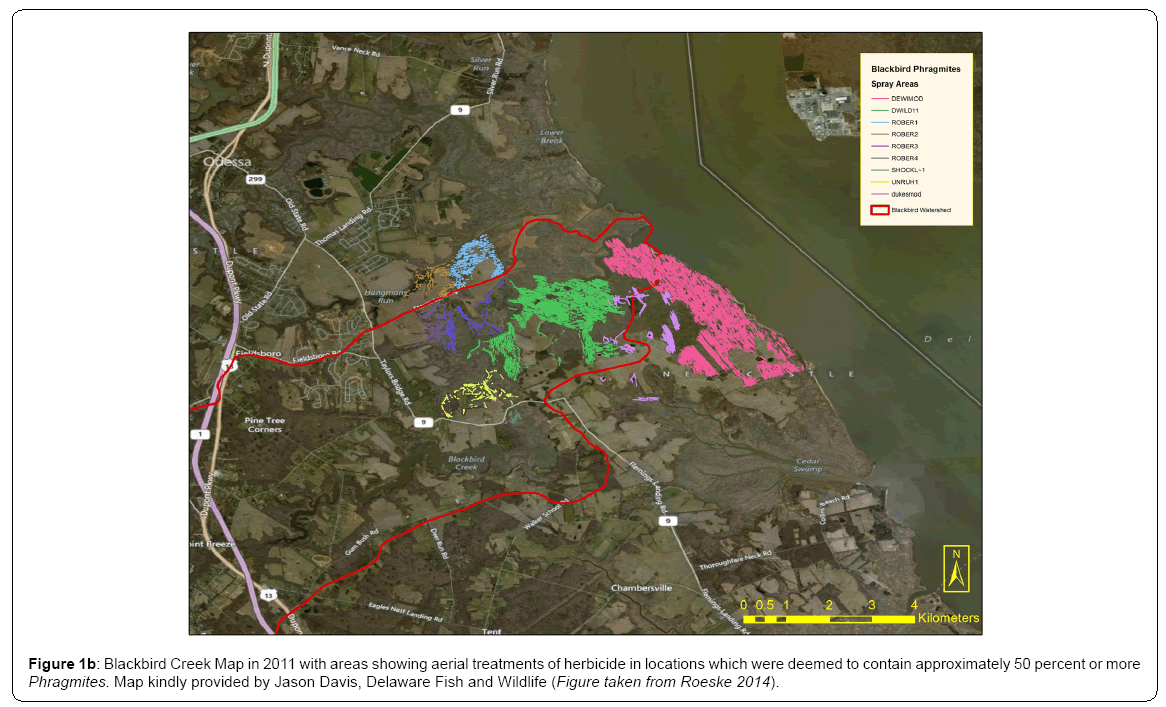
Blue crabs and various other species flourish among the Blackbird Creek marsh grasses, whose leaves, roots, and stems provide much needed food sources and shelter from predators. Many Delaware coastal wetlands, including Blackbird Creek, have been subject to a loss of biodiversity over the past several decades, largely due to the invasion of the common reed (Phragmites australis). It may be considered a highly disturbed ecosystem due to this invasion and to the intensive management (i.e. herbicide spraying) that has occurred since the early 1990’s (Figure 1a, 1b).
Healthy populations of submerged aquatic vegetation (SAV) and structured bottoms are two features which are severely lacking throughout the Delaware estuary [18]. The subtidal environment of Blackbird Creek can be described as muddy and unstructured, forcing juvenile crabs and fish to seek out other suitable refuge and nurseryproviding habitat. Thus, they generally benefit from the relatively shallow streams and creeks with accessible intertidal zones that, in their native form, consist of a mosaic of marsh vegetation which provides structure in comparison to the otherwise unstructured subtidal habitats. These microhabitats found within most tidal salt marshes in the region provide a means of protection from predation, safe mating and molting opportunities, and a source of nutrition [19,20]. With this knowledge in mind, it follows that any drastic changes to the marsh vegetation assemblages will result in subsequent alterations to the value of nearby habitats.
The generalist nature and widespread distribution of blue crabs (Callinectes sapidus) throughout the Delaware Bay, from both a temporal and spatial perspective, indicates the importance of these types of systems to sustain the health of blue crabs and other economically and ecologically important species in the face of anthropogenic and environmental stresses [21]. Some of the more prominent issues facing wetland habitats include past, present, and future changes in land use and land cover, such as habitat encroachment and destruction, agriculture and historic diking of estuarine habitat, shoreline hardening, shoreline erosion, and both direct and indirect anthropogenically induced alterations to the structure and function of the actual marsh surface such as hydrology, topography, vegetation community, nutrient retention, tidal flooding, detritus accumulation and availability to secondary producers. Long the subject of controversy, various studies have examined fish, macro-invertebrate, turtles and avian species diversities in the monocultures of Phragmites and the research outcomes have been highly variable regarding the direction and magnitude of impact of Phragmites australis on habitat suitability for various species. However, it is a different story for the birds and turtles where significant negative impacts of this deleterious species recorded on the diversity and abundance of birds (i.e. willet, seaside sparrow, sharp-tailed sparrow) and turtles [22,23]. Anecdotal reports of decreases in biodiversity and natural marsh function in some areas have only recently been quantified using GIS and aerial imaging technology [24]. Thus it would be pertinent for managers to maintain the health of these areas by preventing any further alterations, especially from the invasion of Phragmites australis.
The physical and mechanical methods for controlling Phragmites include the removal of dikes, the construction of controlled wetlands, raising the water table, modification of micro-topography [25,26], prescribed burning of selected marsh areas and rotary mowing where possible. The most effective management strategies have utilized a combination of the aforementioned removal techniques with the logic that constant stress through mowing, burning, herbicide treatment, increased salinity, etc. will encourage native plant colonization while pushing out Phragmites. It is also important to note that burning alone may actually reinvigorate the populations in subsequent years and thus should be utilized in a strategic manner over the course of several growing seasons if any permanent removal is to be accomplished [27].
Structure, function, and change are part of common terminology incorporated into landscape and habitat restoration jargon. If structure is restored, but function and/or change is neglected, restoration will not be achieved. This holds true for any combination of these three terms in that they are intimately connected. Without function, there cannot be any structure. Without change, you have a static environment, which is never the case in the natural environment. And if one thinks of the problem of the invasion of Phragmites as a landscape level problem, site based restoration will not suffice as it only addresses that particular habitat. Thus, all of the current and future management efforts must be viewed as “habitat restoration” and not “ecological restoration.” This has several important implications including the fact that while some species may benefit from the removal of Phragmites, other species might very well experience deleterious consequences, likely due to the management efforts themselves and not the actual removal or presence of Phragmites. In the case of Phragmites invasion, it appears that both biotic and physical thresholds have been crossed (Figure 2a, 2b, 2c). The dispersal of plants and animals is altered, and the physical functions of the affected marshes have been transformed to certain degrees.
Figure 2a: Dense monoculture of dead and living non-native Phragmites australis within Blackbird Creek. If left unmanaged, vast stretches of wetlands may become engulfed by this highly productive, genetically diverse, and hard to control subspecies, potentially resulting in the displacement of countless numbers of native ecologically and commercially important flora and fauna in the process (Picture taken by Roeske).
Figure 2b: Natural marsh absent of non-native Phragmites australis. Spartina alterniflora and Spartina cynosuroides are observed among several other typical native marsh plants and in most cases both plant and animal diversity is greater than in a marsh that is considered to be undergoing a late stage of non-native Phragmites invasion (Picture taken by Roeske).
The program focus over the last five years in Delaware Blackbird Creek has been to monitor blue crab and other aquatic species in relation to the land use, water quality, and salt marsh habitat. These efforts also include investigating microbial load and diversity associated with Spartina alterniflora- and Phragmites australis-dominated sites in Blackbird Creek. Bio-indicators including Protobacteria, Vesicular arbuscular mycorrhizae (VAM) fungi, and diatoms have also been investigated. Biological indicators such as a diatoms and soil microbes are species that respond to changes in environmental conditions and can be used to monitor the health of an environment or ecosystem. The microbial distribution of these bio-indicators in soil, roots, and leaves have been monitored and related to the physical and nutrient components of samples from different seasons in order to understand their effect on microbial distribution relative to plant growth and distribution since microbes are influenced strongly by plant phenology. Specific molecular analysis for Proteobacteria testing has been performed spatially and temporally to identify and compare microbial communities associated with two dominant marsh grasses (Phragmites australis and Spartina alterniflora). Vesicular arbuscular mycorrhizae (VAM) are fungi that effectively increase the nutrient availability to the host plants. Microscopic and molecular analysis of plant roots was carried out for the detection of the fungi and a seasonal study was conducted to determine their presence and abundance during the growing season and the death phase. Diatom population dynamics determined by genetic analysis in order to infer seasonal and spatial water quality trends.
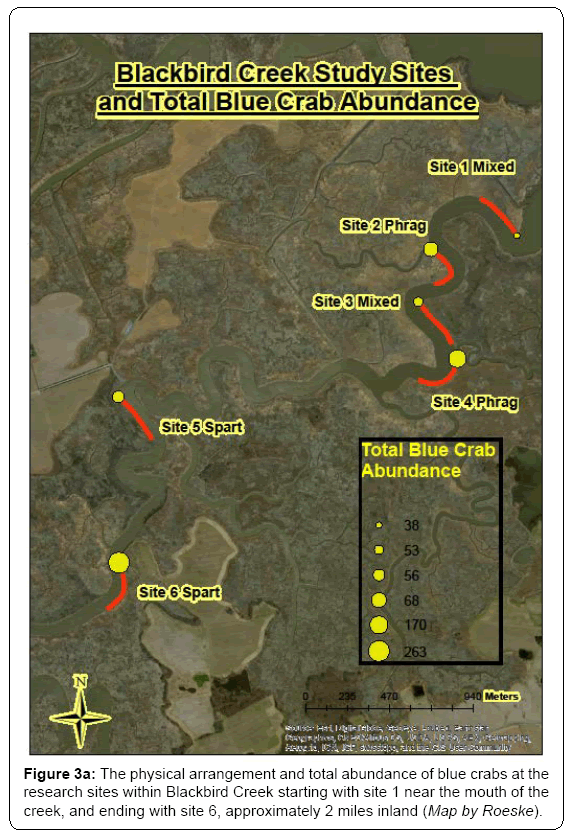
Comparable studies have suggested that changes in fish and invertebrate habitat may be the result of the invasion of Phragmites in the mid-Atlantic region. Low abundance of both male and female crabs are found near the mouth of Blackbird Creek, with peak abundances occurring at inland sites. Phragmites-dominated sites show a trend towards presence of larger crabs while Spartina-dominated sites show a trend toward smaller individuals and crabs in the pre-molt stage. A relatively high degree of molting was observed in mixed and Spartina designated sites. Fish abundance appears to be correlated with crab abundance across study sites. The Phragmites sites and Spartina sites displayed high relative abundance values (Figure 3a, 3b). Along with information about aquatic fauna, total phosphorous, nitrate, and nitrogen are high in areas dominated by Spartina and mixed marsh grass species, and Phragmites-dominated areas are not rich in any major nutrients. Marsh grass diversity seems to affect the soil nutrients and microbial community. VAM fungi were observed within the roots of Spartina alterniflora. These fungi were also observed in the invasive Phragmites when they exist along with Spartina. These fungi help the plants with uptake of nitrogen and phosphorus from the soil. The benthic diatom community differs among the sites studied, and a good correlation between the benthic diatom community and water quality was observed. Sites high in dissolved oxygen and moderate in nutrients provided diverse benthic diatom assemblages. The dominant diatom in Spartina and mixed marsh areas was Navicula phyllepta, where nitrate-nitrogen and total nitrogen were found in high concentrations. According to Underwood and Barnett [28] N. phyllepta is predominant in soils with nitrate-nitrogen and ammonium. In areas dominated by Phragmites, Cylindrotheca closterium, a diatom abundant in saline conditions [29] has been identified. Thalassiosira sp. is abundant in marsh areas near agricultural sites. Thus, microbial diversity varies with marsh habitat in which the soil nutrient composition is being impacted by land use and altering habitat quality for resident nekton.
Blue crabs are known to choose sites based on habitat quality.Some marshes may provide less value to actively feeding/molting crabs. This may have important implications for adults that utilize the marsh surface for feeding. The length of time of tidal inundation may be lower at Phragmites sites than Spartina sites because of the nature of the root structures which can result in increased detrital accumulation and raised marsh surfaces during late stage Phragmites invasion.
The effort to monitor marsh grass habitat provides enhanced insight into the importance of how marsh surface vegetation is utilized by blue crabs and resident fish and to advance the management and restoration efforts that have been, and are currently, in place for the elimination of P. australis. Also, the type of marsh grass affects the soil nutrients, and thereby influences the micro- and macroorganisms living in the marsh environment. Studying the microbial community with respect to native and invasive marsh grasses will help us identify stress tolerant microbes and microbial genes that play a role in helping plants deal with salt and other stresses, which may be potentially applied to terrestrial plants.
The Blackbird Creek watershed is comprised of only four percent urban development and thus provides an opportunity to study an area that has had little anthropogenic impact relative to other watersheds. Our research program is now focusing on how trophic dynamics can be affected as a function of various land uses. In Blackbird Creek, there are several areas designated as cropland. An understanding of crop rotation in these plots both within and between seasons, coupled with a complete survey of aquatic nekton would provide insight into whether changes in crops across years affect trophic interactions and food web dynamics. Additionally, this project delves into the effectiveness of riparian buffers as blockades for fertilizer runoff. With sites associated with buffers, it will be useful to identify different food web characteristics, especially if there are inconsistencies in the nutrient levels between those sites. While this is intriguing, it would be even more beneficial if a second watershed was considered-one with more urban areas and anthropogenic effects. If similar work was done in each of these watersheds, we may be able to discern the effects of different land uses on Delaware’s coastal waterways.
Acknowledgements
We would like to acknowledge the following individuals: Dr. Andrew Augustine, Mr. Michael Mensinger, fellow graduate student Brian Reckenbeil, and undergraduate interns Andrew Kluge, Amy Cannon, Akida Ferguson, Katie Ommanney, Eunice Handy, Eleanor Johnson, Deidre Carter, Kelly Clark, Connor Dixon, Sierra Schirtzinger for their contributions and efforts in this research program. This program is funded by various funding agencies including NSFEPSCOR, USDA-NIFA Capacity Building Grant, NOAA-ECSC, USDA Evans-Allen, and DuPont Clear into the Future Grants Programs.
References
- Dahl TE (1990) Wetlands Losses in the United States 1780’s to 1980’s. U.S. Department of the Interior, Fish and Wildlife Service, Washington, DC.
- Hazelton ELG, Mozdzer TJ, Durdick DM, Kettenring KM, Whigham DF (2014) Phragmitesaustralis management in the United States: 40 years of methods and outcomes. Am. J. Bot. 101: 1577-1584.
- Wilcox KL, Petrie SA, Maynard LA, Meyer SW (2003) Historical Distribution and Abundance of Phragmitesaustralis at Long Point, Lake Erie, Ontario. J. Great Lakes Research. 29: 664-680.
- Weis JS, Weis P (2003) Is the invasion of the common reed, Phragmitesaustralis, into tidal marshes of the eastern US an ecological disaster? Mar Pollut Bull. 46: 816-820.
- Artigas F, Pechmann IC (2010) Balloon imagery verification of remotely sensed Phragmitesaustralis expansion in an urban estuary of New Jersey, USA. Landscape Urban Plan.95: 105-112.
- Kiviat E (2013) Ecosystem services of Phragmites in North America with emphasis on habitat functions.
- Leonard LA, Wren PA, RL Beavers (2002) Flow Dynamics and Sedimentation in Spartinaalternifloraand Phragmitesaustralis Marshes of the Chesapeake Bay. Wetlands 22: 415-424.
- Stribling JM, Cornwell JC (1997) Identification of Important Primary Producers in a Chesapeake Bay Tidal Creek System Using Stable Isotopes of Carbon and Sulfur. Estuaries. 20: 77-85.
- Weinstein MP, Balletto JH (1999) Does the Common Reed, Phragmitesaustralis, Affect Essential Fish Habitat? Estuaries. 22:793-802.
- Meyerson LA,Saltonstall K,Windham L,Kiviat E,Findlay S (2000) A comparison of Phragmitesaustralisin freshwater and brackish marsh environments in North America. Wetlands Ecology and Management. 8: 89-103
- Windham L, Ehrenfeld JG (2003) Net Impact of a Plant Invasion on Nitrogen-Cycling Processes within a Brackish Tidal Marsh. Ecol Appl.13: 883-896.
- Arce MI, Gomez R, Vidal-Abarca MR, Suarez ML (2009) Effects of Phragmitesaustralisgrowth on nitrogen retention in a temporal stream. Limnetica. 28: 229-242.
- Ailstock MS, Norman CM, Bushmann PJ (2001) Common Reed Phragmitesaustralis: Control and Effects Upon Bio Diversity in Freshwater Nontidal Wetlands. Restor Ecol. 9: 49-59.
- Weis JS, Weis P (2000) Behavioral responses and interactions of estuarine animals with an invasive marsh plant: a laboratory analysis. Biological Invasions2: 305-314.
- Able KW, Hagan SM (2000) Effects of Common Reed (Phragmitesaustralis) Invasion on Marsh Surface Macrofauna: Response of Fishes and Decapod Crustaceans. Estuaries.23:633-646.
- Jivoff PR, Able KW(2003) Blue Crab, Callinectessapidus, Response to the Invasive Common Reed, Phragmitesaustralis: Abundance, Size, Sex Ratio, and Molting Frequency. Estuaries. 26: 587-595.
- DNREC-DNERR (2014) Delaware Coastal Programs. Blackbird Creek Reserve, USA.
- Beckman DW (2013) Marine Environmental Biology and Conservation. Jones and Bartlett Learning, USA.
- Peterson GW, Turner RE (1994) The value of salt marsh edge vs. interior as a habitat for fish and decapod crustaceans in Louisiana tidal marsh. Estuaries. 17: 235-262.
- Orth RJ, Montfrans JV (1990) Utilization of Marsh and Seagrass Habitats by Early Stages of Callinectessapidus: a Latitudinal Perspective. B Mar Sci.46: 126-144.
- Roeske KP (2014) Assessing the Impact of Phragmitesaustralis Subspecies australis on Blue Crab (Callinectessapidus) and Fish Population Dynamics in Blackbird Creek, Delaware.
- Fleet T (2014) Phragmitesaustralis - cryptic invasion of the Common Reed in North America. The Encyclopedia of Earth.
- Bolton RM, Brooks RJ(2010) Impact of the Seasonal Invasion ofPhragmitesaustralis(Common Reed) on Turtle Reproductive Success. Chelonian Conservation and Biology. 9: 238-243.
- Ozbay G,Augustine A, Fletcher R (2012) Remote Sensing of Phragmitesaustralis Invasion in Delaware Tidal Marsh Zones: Issues to Consider. J Geophys Remote Sensing. 1: 1-3.
- Philipp KR (2005) History of Delaware and New Jersey salt marsh restoration sites. Ecol Eng.25:214-230.
- Teal JM, Weishar L(2005) Ecological engineering, adaptive management, and restoration management in Delaware Bay salt marsh restoration. Ecol Eng.25: 304-314.
- Fish and Wildlife Service (2004) Comprehensive Conservation Plan.
- Underwood GJC, Barnett M(2006) What determines species composition in microphytobenthic biofilms? Microphytobenthos Symposium, Netherlands.
- Mirjana N, Maria B, Tamara D, Romina K (2005) The role of the diatom Cylindrothecaclosteriumin a mucilage event in the northern Adriatic Sea: coupling with high salinity water intrusions. J Plankton Res.27: 851-862.
Relevant Topics
- Aquatic Ecosystems
- Biodiversity
- Conservation Biology
- Coral Reef Ecology
- Distribution Aggregation
- Ecology and Migration of Animal
- Ecosystem Service
- Ecosystem-Level Measuring
- Endangered Species
- Environmental Tourism
- Forest Biome
- Lake Circulation
- Leaf Morphology
- Marine Conservation
- Marine Ecosystems
- Phytoplankton Abundance
- Population Dyanamics
- Semiarid Ecosystem Soil Properties
- Spatial Distribution
- Species Composition
- Species Rarity
- Sustainability Dynamics
- Sustainable Forest Management
- Tropical Aquaculture
- Tropical Ecosystems
Recommended Journals
Article Tools
Article Usage
- Total views: 15102
- [From(publication date):
December-2014 - Sep 20, 2024] - Breakdown by view type
- HTML page views : 10665
- PDF downloads : 4437