Research Article Open Access
A Clinical Study on the Relationship Between Lymph Nodes Metastasis in Early Gastric Carcinoma and Clinicopathological Factors and Reasonable Treatment Options
Xin Zhang, Rui Zheng, Guo Wei, Jian-wei Bi and Ming-Ming Nie*
Department of General Surgery, Changhai Hospital, The Second Military Medical University, Xiangyin Road, Shanghai 200433, PR China
- Corresponding Author:
- Ming-Ming NIE
Department of General Surgery, Changhai Hospital
The Second Military Medical University
Xiangyin Road, Shanghai 200433, PR China
E-mail: niemm88@hotmail.com
Received Date: June 23, 2013; Accepted Date: August 09, 2013; Published Date: August 12, 2013
Citation: Zhang X, Zheng R, Wei G, Bi Jw, Nie MM (2013) A Clinical Study on the Relationship Between Lymph Nodes Metastasis in Early Gastric Carcinoma and Clinicopathological Factors and Reasonable Treatment Options. J Gastroint Dig Syst S1:007. doi: 10.4172/2161-069X.S1-007
Copyright: © 2013 Zhang X, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Objective: To investigate the rule of lymph node metastasis in early gastric cancer , in order to provide a reference for the reasonable treatment options.
Method: To retrospectively analyze the clinical data of 271 cases of EGC patients from October 2005 to October 2011.
Results: Both univariate analysis and multivariate analysis results show that tumor size, pathological classification and depth of invasion are correlated with lymph node metastasis. The further analysis of the rate of lymph node metastasis shows that lymph node metastasis cannot be found in the patients with differentiated tumor of which the size is below 3.0cm when tumor invades mucosal layer; at the same time, lymph node metastasis cannot be found in the patients with undifferentiated tumor whose size is below 2.0cm as well. However, lymph node metastasis occurs in all the sorts when tumor invades submucosal layer.
Conclusion: Tumor size, invasive depth, pathological classification, and lymphatic tube invasion were closely related to lymph node metastasis in patients with early gastric cancer, patients with early gastric cancer are recommended to have a preoperative Endoscopic Ultrasonographic (EUS) scan as well as pathological screenings for deciding the most appropriate type of surgery.
Keywords
Early gastric cancer; Lymph node metastasis; Pathological factors; Endoscopic resection
Introduction
Early gastric cancer (EGC) is defined as invasive gastric cancer that invades no more deeply than the submucosa, regardless of lymph node metastasis. Previous studies reported that the 5-year overall survival for EGC, with a rate of lymph node metastasis of 15.0-20.0% and a recurrence rate of 1.4-2.7%, was about 90.0% after surgery [1]. Lymph node involvement is an important factor that influences recurrence of EGC [2]. Thus, for patients with EGC, preoperative analysis of risk factors for lymph node metastasis not only indicates the prognosis, but could also help medical practitioners in choosing the most appropriate therapy regimen.
To date, the main treatment for EGC is laparotomy and laparoscopic operations. However, with the development of endoscopic techniques in the last few years, the techniques of full-thickness mucosal resection, with endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) as examples, have become increasingly mature and are now used extensively in Asian countries and are gradually becoming accepted in Western countries [3-4]. Compared with a laparotomy, endoscopic resection has advantages such as minimal invasion with little trauma and increased quality of life after surgery. However, there are also limitations of endoscopic resection for gastric cancer, especially in the resection of any possible metastasis to the lymph nodes around the stomach, which greatly increases the risk of recurrence after surgery. Furthermore, endoscopic resection for early gastric cancer has been controversial with regard to its lack of an accurate and effective assessment strategy for lymph node metastasis before surgery.
We performed a retrospective analysis of 271 patients with early gastric cancer in our department over the past 6 years. We investigated the association between clinical pathological characteristics and lymph node metastasis in early gastric cancer to provide relevant risk factors for lymph node metastasis and a basis for the selection of appropriate treatment for EGC.
Materials and Methods
Data screening and patient selection
In total, 221 (173 males, 98 females) with early gastric cancer admitted to our department between October 2005 and October 2011, aged between 25 and 87, were included. All patients above underwent laparotomy with D1 or D2 lymphadenectomy: 64 with D1 lymphadenectomy and 207 with D2 lymphadenectomy. The standard of surgical procedure and range of lymphadenectomy were classified according to Gastric Cancer Treatment Guidelines (3rd version) of the Japanese Gastric Cancer Association (JGCA) [5]. Additionally, one patient was underwent laparoscopic distal partial gastric resection. All patients underwent preoperative routine stomachoscopies and pathological biopsies with routine H&E staining.
Methods
Data on gender, age, tumor location, tumor size (cm), gross appearance, pathological classifications, and invasive depth were collected for analysis. Study subjects were divided into groups according to WHO age category definitions: young, middle-aged (< 60 years ≥ 60 years) and older groups. The sizes of tumor were measured by greatest diameter of surface. Gross types were divided into elevated, flat, or depressed types according to the Japanese Gastric Cancer Regulations (12th version). Pathological classifications were categorized into differentiated type (papillary adenocarcinoma, tubular adenocarcinoma and high-moderately differentiated adenocarcinoma) and undifferentiated type (differentiated adenocarcinoma, undifferentiated adenocarcinoma, mucinous adenocarcinoma, and signet-ring cell carcinoma) according to WHO criteria.
Statistical analysis
The chi-squared test was used to analyze enumeration data and multivariate analysis was performed through dichotomy nonconditional logistic regression analysis. All related factors with statistical significance were compared with rates of lymph node metastasis. Confidence intervals of overall rate were evaluated by binomial distribution. Statistical analyses were two-sided, and P values < 0.05 were considered to indicate statistical significance. All analyses were performed using the SPSS software (ver. 15.0).
Results
Clinicopathological features
Of all the 271 gastric carcinoma cases, the tumors comprised 179 cases with lesions located in the lower part of the stomach (66.1%), middle part of the stomach in 54 cases (19.9%), upper part of the stomach 38 cases (14.0%). In tumor size, less than 1cm in diameter in 13 cases (4.8%); 1 ~ 2cm in 99 cases (36.5%), 2 ~ 3cm in 95 cases (35.1%); larger than 3cm in 64 cases (23.6%). According to the Goseki classification, the tumour series included 27 type I tumours (9.9%), 194 type II tumours 71.6%, 50 type III tumours (18.5%). According to the WHO classification, three tumours were tubulous (1.1%), one were papillary (0.4%), 133 were moderate-differentiated (49.1%). 60 were low-differentiated (22.1%), four were mucinous (1.5%), 60 were signet ring cell (22.1%). According to the endoscopic and postoperative observation results, there were 195 cases with ulcer scar (72.0%) and 76 cases without ulcer scar (28.0%). And extent of tumor invasion the tumors comprised 161 cases with invasion in the mucosa (59.4%) and 110 cases in the submucosa (40.6%).
Univariate analysis results
Each factor for groups with and without lymph node metastasis was compared using the chi-squared test. There were statistically significant differences in the comparisons of lymph node metastasis rates for tumor size, histological classification, and invasive depth ≥ .2 Lycm,ph node metastasis rates were 42.2% for tumors which was significantly higher than the other two groups, with rates of 5.4% and 10.4% (P < 0.01). The lymph node metastasis rates in undifferentiated tumors (5.4%) were lower than in differentiated tumors (10.4%; P < 0.05). Metastasis rates of lymph nodes with submucosal invasion (32.7%) were significantly higher than with mucosal invasion (4.3%; P < 0.01; Table 1).
| Number of cases | With no lymph | With lymph | |||||
|---|---|---|---|---|---|---|---|
| nodes metastasis | nodes metastasis | χ2 | P | ||||
| % | % | ||||||
| Age | |||||||
| <60 | 147 | 123 | 83.7 | 24 | 16.3 | 0.051 | 0.882 |
| ≥ 60 | 124 | 105 | 84.7 | 19 | 15.3 | ||
| Gender | |||||||
| Male | 173 | 147 | 85.0 | 26 | 15.0 | 0.252 | 0.616 |
| Female | 98 | 81 | 82.7 | 17 | 17.3 | ||
| Location | |||||||
| Upper | 38 | 33 | 86.8 | 5 | 13.2 | 2.084 | 0.353 |
| Middle | 54 | 42 | 77.8 | 12 | 22.2 | ||
| Lower | 179 | 153 | 85.5 | 26 | 14.5 | ||
| Tumor size | |||||||
| <2 cm | 111 | 105 | 94.6 | 6 | 5.4 | 44.449 | <0.01 |
| 2~3 cm | 96 | 86 | 89.6 | 10 | 10.4 | ||
| ≥ 3 cm | 64 | 37 | 57.8 | 27 | 42.2 | ||
| Goseki’s type | |||||||
| I | 27 | 25 | 92.6 | 2 | 7.4 | 2.089 | 0.352 |
| II | 194 | 163 | 84.0 | 31 | 16.0 | ||
| III | 50 | 40 | 80.0 | 10 | 20.0 | ||
| WHO type | |||||||
| Differentiated | 137 | 123 | 89.8 | 14 | 10.2 | 6.621 | <0.05 |
| Undifferentiated | 134 | 105 | 78.4 | 29 | 21.6 | ||
| Uler scar | |||||||
| Yes | 195 | 162 | 83.0 | 33 | 17.0 | 2.499 | 0.114 |
| No | 76 | 56 | 73.7 | 20 | 26.3 | ||
| Depth of | |||||||
| invasion | |||||||
| Mucosa | 161 | 154 | 95.7 | 7 | 4.3 | 39.427 | <0.01 |
| Submucosa | 110 | 74 | 67.3 | 36 | 32.7 | ||
Table 1: Relation between clinicopathological factors of tumors and lymph nodes metastasis.
Multivariate analysis results
Lymph node involvement was considered a dependent variable (ymetastasis = 1 and yno_metastasis = 0). Tumor size, pathological classification, and invasive depth, which showed statistical significance in the univariate analysis, were considered as independent variables and multivariate non-conditional logistic regression analysis was performed χ. The regression model test (χ2 = 78.911, P < 0.01) showed that the model was statistically significant. Table 2 shows that factors influencing lymph node metastasis were tumor size, pathological classification, and invasive depth. An increase in lymph node metastasis of one class was associated with a 4.392-fold (95% CI: 2.438-7.911) increased risk over the former class. Compared with differentiated type, the risk of lymph node metastasis for an undifferentiated tumor was 4.708 (95% CI: 1.915-11.572) in pathological classification. Compared with mucosal invasion, the risk of lymph node metastasis with submucosal invasion was 11.310 (95% CI: 4.377-29.227).
| B | S.E. | Wald | P | OR | 95%CI.OR | ||
|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||
| Tumor size | 1.480 | 0.300 | 24.291 | 0.000 | 4.392 | 2.438 | 7.911 |
| Undifferentiated | 1.549 | 0.459 | 11.400 | 0.001 | 4.708 | 1.915 | 11.572 |
| Submucosal Invasion | 2.426 | 0.484 | 25.077 | 0.000 | 11.310 | 4.377 | 29.227 |
Table 2: Multivariate Logistic regression analysis on the lymph node metastasis in patients with EGC.
Analysis of lymph node metastasis rate
Taking tumor size, pathological classification, and invasive depth, which showed statistical significance as factors analyzed, and dividing patients into two subgroups, of mucosa and submucosa, then a further analysis of the lymph node metastasis rate (ratio between numbers of patients with lymph node metastasis and overall numbers of patients under the same condition) was performed in each group according to tumor size and pathological classification. Table 3 shows that when the tumor infiltrated the submucosa, no lymph node metastasis was present in patients with tumors < 3.0 cm and with undifferentiatedtype pathological classification (0/69, 95% CI 0-6%). Also, patients with tumors > 3.0 cm and undifferentiated-type pathological classification had no lymph node metastasis (0/22, 95% CI 0-15%). However, Table 4 shows that when the tumor infiltrated the submucosa, all patients with tumor < 2.0 cm and some in other categories had lymph node metastasis (6/110, 95% CI 0.2-32.3%).
| Tumor size | Lymph nodes metastasis rate (%) | |
|---|---|---|
| Differentiated | Undifferentiated | |
| <2.0 cm | 0.0(0/54) | 0.0(0/22) |
| 2.0-2.9 cm | 0.0(0/15) | 4.4(2/45) |
| ≥ 3.0 cm | 14.3(2/14) | 27.3(3/11) |
Table 3: Comparison of lymph nodes metastasis rate between differentiated and undifferentiated EGC in 161cases with tumor invasion in the mucosa.
| Tumor size | Lymph nodes metastasis rate (%) | |
|---|---|---|
| Differentiated | Undifferentiated | |
| <2.0 cm | 7.7(1/13) | 21.7(5/23) |
| 2.0-2.9 cm | 11.1(2/18) | 35.3(6/17) |
| ≥ 3.0 cm | 39.1(9/23) | 81.3(13/16) |
Table 4: Comparison of lymph nodes metastasis rate between differentiated and undifferentiated EGC in 110cases with tumor invasion in the submucosa.
Discussion
Gastric cancer is the second leading cause of cancer death worldwide after lung cancer [6] .Compared with advanced gastric cancer, early gastric cancer have obvious advantage in prognosis. However, although treatment on EGC can bring good prognosis, the diagnostic rate remains low, largely due to the lack of simple, inexpensive examination means and tumor markers with high specificity and sensitivity. Although the positive rate for the diagnosis of EGC reached 81.0% by the means of EUS [6], there still remains possibility of misdiagnosis or missed. In addition, most of the patients diagnosed EGC underwent laparoscopic surgery or laparotomy. Undoubtedly, these surgical treatments may reduce the risk of possible lymph nodes metastasis, but on the other hand, for those patients with EGC who have no lymph nodes metastasis, undergoing EMR or ESD might bring them lower risk of operations, trauma, smaller wounds, shorter time of postoperative recovery and decreased medical expenses.
Because only gastric mucosa and submucosa with lesions were removed through endoscopic resection, postoperative quality of life was obviously better than by laparotomy. That is, of course, why endoscopic resection has been popularized [4]. Lymph node involvement is the most important factor of prognosis for EGC and is significantly associated with survival rate, which is the main basis for the decision as to whether to use endoscopy to treat gastric cancer [7-12]. The principal indications in the Gastric Cancer Treatment Guidelines (ver.3, the “Guidelines”) for EMR and ESD are that the possibility of lymph node metastasis was extremely low and the tumor could be removed completely. The Guidelines also suggested that absolute indications for EMR and ESD were macroscopic mucosal cancers of less than 2 cm (cT1a) in diameter with a differentiated histological type (pap, tub1, tub2), regardless of gross appearance type without an ulcer lesion. Extensive indications for ESD have also been established. Although a number of reports about treatment of EGC with ESD have been published in recent years, related clinical evidence about long-term prognoses is insufficient. Thus the Guidelines note that related clinical studies should be performed to confirm the extensive indications for ESD. Several hospitals in our country have generally followed the example in the Guidelines when choosing a standard for endoscopic resection. Because controversy regarding the surgery indications in the Guidelines has continued, and large-scale screening has not been developed in our country, as it was in Japan, there is a lower diagnostic rate of early gastric cancer than in Japan and different problems are encountered clinically. Screening should be performed according to the Guidelines.
Lim et al. [13] reviewed 376 gastric cancers retrospectively and found that tumor size, invasive depth, pathological classification and lymphatic tube invasion were closely related to lymph node metastasis. Choi et al. [14] also reviewed 1717 patients and made the same findings. Our study also found that tumor size, invasive depth, pathological classification, and lymphatic tube invasion were closely related to lymph node metastasis in patients with early gastric cancer, consistent with previous reports. Moreover, when the tumor was confined to the mucosa with a diameter of less than 2 cm, patients with both differentiated and undifferentiated tumors did not show lymph node metastasis. When the tumor size was over 2 cm and less than 3 cm, patients with undifferentiated tumors did show lymph node metastasis. However, when the sub mucosa was infiltrated, lymph node metastasis was seen with all tumors, regardless of size.
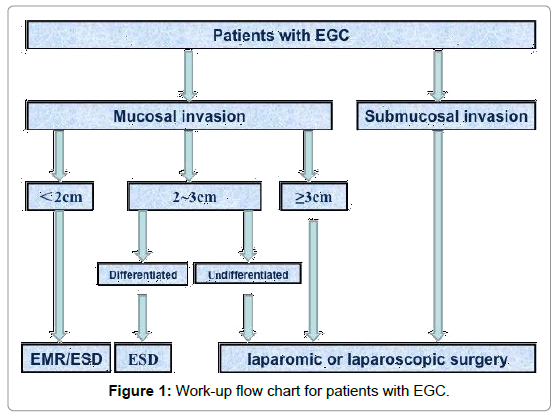
We provide a flow diagram about screening of treatment methods for early gastric cancer, based on our study (Figure 1). All examination data were collected through gastroscopy and ultrasound-guided gastroscopy and pathological biopsies were performed. Complication rates of 10.0-30.0% were estimated during endoscopic resection [15]. Thus, surgeons should have extensive gastroscopy and surgery experience to reduce the incidence of complications. Li et al. [16] considered that patients with a undifferentiated tumor < 2 cm and no lymphatic vessel invasion might be suggested to undergo EMR or ESD, consistent with the Guidelines and our study results. Additionally, we suggest that the indications for ESD should be restricted to mucosal tumors with a diameter < 2 cm or differentiated mucosal tumor with a diameter < 2 cm. Moreover, patients with tumors infiltrating the submucosa should undergo laparotomies without exception, which is a little different than the extensive indications in the Guidelines. Jee et al. [17] reported that the lymph node metastasis rate for tumors ≤ 3 cm with submucosal invasion was about 4.0%, resulting in a risk of incomplete treatment. Otherwise, the pathology department should collaborate closely with clinical departments so that patients undergo necessary and available pathological tests. Although little possibility of error was indicated in preoperative diagnoses [18-20], when it comes to endoscopic resection for gastric cancer, pathological diagnosis remains the gold standard for therapy indication. Only precise pathological results can allow accurate pathological screening and then different treatment methods can be chosen for different groups so that patients benefit. To date, multidisciplinary cooperation has greatly helped in the early diagnosis and treatment of gastric cancer.
In summary, we recommend that surgeons can choose proper surgical approaches for patients with EGC according to the preoperative EUS and pathological results, and control the indications of endoscopic resection strictly: For the patients with tumor size < 2 cm in diameter that invade to the mucosa, regardless of their pathology classification, EMR or ESD is recommended; For the patients with tumor size between 2 ~ 3 cm in diameter and are classified as differentiated tumors with no submucosal invasion, ESD is recommended; For the patients with tumor size ≥ 2 cm in diameter and are classified as undifferentiated tumor with no submucosal invasion, or patients with tumor ≥ 3 cm size but without submucosal invasion, regardless of tumor pathological classification mucosal layer, or the patients with submucosal invasion, Laparoscopic or laparotomic radical gastrectomy are recommended.
References
- Lai JF, Kim S, Kim K, Li C, Oh SJ, et al. (2009) Prediction of recurrence of early gastric cancer after curative resection. Ann Surg Oncol 16: 1896-1902.
- Sano T, Hollowood A (2006) Early gastric cancer: diagnosis and less invasive treatments. Scand J Surg 95: 249-255.
- Hotta K, Oyama T, Akamatsu T, Tomori A, Hasebe O, et al. (2010) A comparison of outcomes of endoscopic submucosal dissection (ESD) For early gastric neoplasms between high-volume and low-volume centers: multi-center retrospective questionnaire study conducted by the Nagano ESD Study Group. Intern Med 49: 253-259.
- Gotoda T (2007) Endoscopic resection of early gastric cancer. Gastric Cancer 10: 1-11.
- Japanese Gastric Cancer Association (2011) Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer 14: 113-123.
- Shimoyama S, Yasuda H, Hashimoto M, Tatsutomi Y, Aoki F, et al. (2004) Accuracy of linear-array EUS for preoperative staging of gastric cardia cancer. Gastrointest Endosc 60: 50-55.
- Saka M, Katai H, Fukagawa T, Nijjar R, Sano T (2008) Recurrence in early gastric cancer with lymph node metastasis. Gastric Cancer 11: 214-218.
- Kunisaki C, Makino H, Akiyama H, Otsuka Y, Ono HA, et al. (2008) Clinical significance of the metastatic lymph-node ratio in early gastric cancer. J Gastrointest Surg 12: 542-549.
- Nitti D, Marchet A, Olivieri M, Ambrosi A, Mencarelli R, et al. (2003) Ratio between metastatic and examined lymph nodes is an independent prognostic factor after D2 resection for gastric cancer: analysis of a large European monoinstitutional experience. Ann Surg Oncol 10: 1077-1085.
- Gotoda T, Iwasaki M, Kusano C, Seewald S, Oda I (2010) Endoscopic resection of early gastric cancer treated by guideline and expanded National Cancer Centre criteria. Br J Surg 97: 868-871.
- Kang HJ, Kim DH, Jeon TY, Lee SH, Shin N, et al. (2010) Lymph node metastasis from intestinal-type early gastric cancer: experience in a single institution and reassessment of the extended criteria for endoscopic submucosal dissection. Gastrointest Endosc 72: 508-515.
- Hoteya S, Yamashita S, Kikuchi D, Nakamura M, Fujimoto A, et al. (2011) Endoscopic submucosal dissection for submucosal invasive gastric cancer and curability criteria. Dig Endosc 23: 30-36.
- Lim MS, Lee HW, Im H, Kim BS, Lee MY, et al. (2011) Predictable factors for lymph node metastasis in early gastric cancer-analysis of single institutional experience. J Gastrointest Surg 15: 1783-1788.
- Choi J, Kim SG, Im JP, Kang SJ, Lee HJ, et al. (2011) Lymph node metastasis in multiple synchronous early gastric cancer. Gastrointest Endosc 74: 276-284.
- Yamamoto H (2007) Technology insight: endoscopic submucosal dissection of gastrointestinal neoplasms. Nat Clin Pract Gastroenterol Hepatol 4: 511-520.
- Li C, Kim S, Lai JF, Oh SJ, Hyung WJ, et al. (2008) Risk factors for lymph node metastasis in undifferentiated early gastric cancer. Ann Surg Oncol 15: 764-769.
- Jee YS, Hwang SH, Rao J, Park DJ, Kim HH, et al. (2009) Safety of extended endoscopic mucosal resection and endoscopic submucosal dissection following the Japanese Gastric Cancer Association treatment guidelines. Br J Surg 96: 1157-1161.
- Mouri R, Yoshida S, Tanaka S, Oka S, Yoshihara M, et al. (2009) Usefulness of endoscopic ultrasonography in determining the depth of invasion and indication for endoscopic treatment of early gastric cancer. J Clin Gastroenterol 43: 318-322.
- Kim GH, Park do Y, Kida M, Kim DH, Jeon TY, et al (2010) Accuracy of high-frequency catheter-based endoscopic ultrasonography according to the indications for endoscopic treatment of early gastric cancer. J Gastroenterol Hepatol 25: 506-511.
- Palmer G, Martling A, Cedermark B, Holm T (2011) Preoperative tumour staging with multidisciplinary team assessment improves the outcome in locally advanced primary rectal cancer. Colorectal Dis 13: 1361-1369.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 14700
- [From(publication date):
specialissue-2012 - Dec 23, 2025] - Breakdown by view type
- HTML page views : 9891
- PDF downloads : 4809