Review Article Open Access
Back to Basics: Regulation of the Gastrointestinal Functions
Ayman I Sayegh* and Martha C WashingtonGastroenterology Laboratory, Department of Biomedical Sciences, College of Veterinary Medicine, Tuskegee University, Tuskegee, Alabama, USA
- *Corresponding Author:
- Ayman I Sayegh
Gastroenterology Laboratory
Department of Biomedical Sciences
College of Veterinary Medicine
Tuskegee University
Tuskegee, Alabama, USA
E-mail: sayeghai@mytu.tuskegee.edu
Received date: December 12, 2012; Accepted date: December 14, 2012; Published date: December 17, 2012
Citation: Sayegh AI, Washington MC (2012) Back to Basics: Regulation of the Gastrointestinal Functions. J Gastroint Dig Syst 2:118. doi: 10.4172/2161-069X.1000118
Copyright: © 2012 Sayegh AI. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
The gastrointestinal tract is a tube like structure which extends from the mouth to the anus and supplies the various body systems, including itself, with nutrients, electrolytes and water through performing five functions. These functions include digestion, secretion, absorption, storage and excretion. To orchestrate these functions there are two control systems located inside and outside the gut wall. The intrinsic ones include the enteric nervous system and five hormones including secretion, gastrin, cholecystokinin, gastric inhibitory peptide and motilin and the extrinsic control system of the gut include the vagus and splanchnic nerves and the hormone aldosterone. This review describes the previous systems and their roles in the control of the various functions of the gut.
Keywords
Cholecystokinin; Enteric nervous system; Gastrin releasing peptide; Splanchnic nerve; Vagus nerve
Introduction
The digestive system consists of two parts, the gastrointestinal (GI) tract [1] and the major digestive accessory glands, which include the liver and pancreas. This review will only focus on the control systems that regulate the various functions of the gastrointestinal tract.
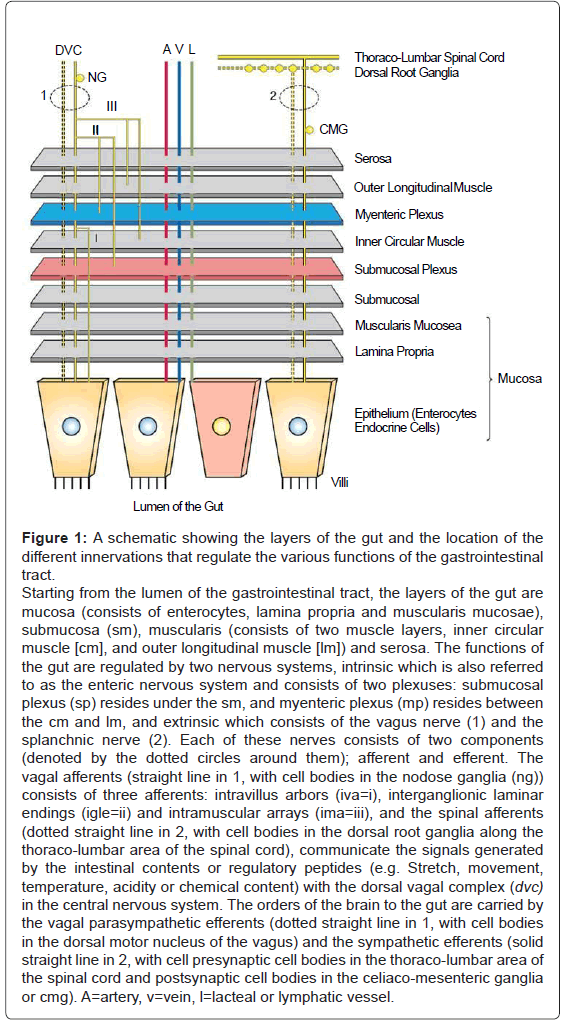
The gastrointestinal tract is a tube-like structure which extends from the mouth to the anus. Histologically (Figure 1), the gut consists of four main layers: [1] the mucosa, which comprises epithelial cells (enterocytes, endocrine cells and others), the lamina propria and the muscularis mucosae; [2] the submucosa; [3] two muscle layers, an inner thick circular layer and an outer thin longitudinal layer; and [4] a serosal layer. Functionally, the gastrointestinal tract supplies the body, including the gut itself, with nutrients, electrolytes and water by performing five distinct functions: motility, secretion, digestion, absorption and storage. The gut orchestrates these functions by two control systems, intrinsic and extrinsic. The intrinsic control system is located between the different layers of the gut, whereas the extrinsic control system resides outside the wall of the gut. Each of these systems consists of two components, namely, nerves and endocrine secretions. The intrinsic control system has two components: the Enteric Nervous System (ENS) and gut hormones, which include gastrin, Gastric Inhibitory Peptide (GIP), Cholecystokinin (CCK), secretin, and motilin. The extrinsic control system elements that regulate gut functions consist of the vagus and splanchnic nerves and the hormone aldosterone. The secretions of the intrinsic and extrinsic control systems of the gut reach their target tissues by four different routes: (A) endocrine - the secretions are deposited in the blood vessels of the gut, (B) paracrine - the secretions diffuse between the cells through the interstitial space, (C) autocrine - the secretions of one cell regulate the functions of the same cell and (D) neurocrine - the secretion of enteric neurons affects the nearby muscles, glands or blood vessels. In addition to the previously mentioned control systems, the gastrointestinal tract contains the highest number of immune cells and immune mediators in the body. Those cells and mediators interact with the intrinsic control system of the gut, both nerves and endocrine cells, to regulate some functions of the gastrointestinal tract, including motility and secretion. This review will provide the basic as well as the clinical scientist with a detailed outline on the intrinsic and the extrinsic control systems of the gut, including [1] the ENS, [2] the intrinsic endocrine system, [3] the immune system of the gut, [4] the vagus and splanchnic nerves and the hormone aldosterone.
Figure 1: A schematic showing the layers of the gut and the location of the different innervations that regulate the various functions of the gastrointestinal tract. Starting from the lumen of the gastrointestinal tract, the layers of the gut are mucosa (consists of enterocytes, lamina propria and muscularis mucosae), submucosa (sm), muscularis (consists of two muscle layers, inner circular muscle [cm], and outer longitudinal muscle [lm]) and serosa. The functions of the gut are regulated by two nervous systems, intrinsic which is also referred to as the enteric nervous system and consists of two plexuses: submucosal plexus (sp) resides under the sm, and myenteric plexus (mp) resides between the cm and lm, and extrinsic which consists of the vagus nerve (1) and the splanchnic nerve (2). Each of these nerves consists of two components (denoted by the dotted circles around them); afferent and efferent. The vagal afferents (straight line in 1, with cell bodies in the nodose ganglia (ng)) consists of three afferents: intravillus arbors (iva=i), interganglionic laminar endings (igle=ii) and intramuscular arrays (ima=iii), and the spinal afferents (dotted straight line in 2, with cell bodies in the dorsal root ganglia along the thoraco-lumbar area of the spinal cord), communicate the signals generated by the intestinal contents or regulatory peptides (e.g. Stretch, movement, temperature, acidity or chemical content) with the dorsal vagal complex (dvc) in the central nervous system. The orders of the brain to the gut are carried by the vagal parasympathetic efferents (dotted straight line in 1, with cell bodies in the dorsal motor nucleus of the vagus) and the sympathetic efferents (solid straight line in 2, with cell presynaptic cell bodies in the thoraco-lumbar area of the spinal cord and postsynaptic cell bodies in the celiaco-mesenteric ganglia or cmg). A=artery, v=vein, l=lacteal or lymphatic vessel.
The Enteric Nervous System (ENS)
The ENS [4] is considered the intrinsic nervous system of the gastrointestinal tract and a component of the Autonomic Nervous System (ANS) - the other two components of the ANS are the sympathetic and parasympathetic nervous systems. The ENS controls the majority of the gastrointestinal functions independent from the Central Nervous System (CNS: brain and spinal cord).
Anatomically, the ENS consists of two main ganglionated plexuses, termed the submucosal (Meissner) and myenteric (Auerbach) plexuses. The submucosal plexus is located under the submucosal layer of the gut, and the myenteric plexus resides between the inner circular muscle layer and the outer longitudinal muscle layer. The two plexuses communicate with each other by interneurons and with the central nervous system through vagal and splanchnic nerves.
The enteric neurons consist of three types (A) sensory (afferent) neurons, (B) interneurons, and (C) motor (efferent) neurons. Sensory input comes from mechanoreceptors within the muscular layers and chemoreceptors within the mucosa. Mechanoreceptors monitor distension of the gut wall, whereas chemoreceptors in the mucosa monitor chemical conditions in the gut lumen. Enteric motor nerves supply vascular muscle, gut muscle, and glands within the gut wall. Efferent neurons of the ENS may be stimulatory or inhibitory.
Unlike classical neurons, the enteric neurons release their neurotransmitter/neuromodulator molecules from vesicles located in swellings along often extensive branches of the axon, not just at the level of the distal synaptic terminals. These swellings are referred to as varicosities. These substances are secreted in response to action potentials, and they affect the activities of nearby smooth muscles or glandular cells. The presence of varicosities in the enteric neurons allow these neurons to activate a wider area in the vicinity of the axon compared with most other types of neurons, which release their neurotransmitters in a more focused and localized area at the synaptic terminal.
Depending on the species, the number of enteric neurons may reach 100 million. This number, in some cases, is more than the number of neurons in the spinal cord. However, to simplify the study of these neurons and to understand their physiological importance, four main classification methods have been utilized. These methods depend on the [1] morphology (different shapes) of enteric neurons, [2] the types of neurotransmitters or peptides that they may contain (also known as their chemical coding), [3] the electrical properties of the enteric neurons or electrophysiology, and [4] the function (e.g., sensory, motor, inhibitory and excitatory) of the enteric neurons).
On the basis of morphology the Russian histologist Alexander Dogiel described three types of enteric neurons in 1896 and 1899. These types are: (A) Dogiel type I, (B) Dogiel type II and (C) Dogiel type III neurons. Dogiel type I neurons have small, irregular cell bodies with multiple short dendrites. Dogiel type II neurons have large, oval-shaped cell bodies with one or two long dendrites. Dogiel type III neurons have large cell bodies with different shapes and multiple dendrites.
Electrophysiologically, there are two types of enteric neurons: (I) S-type (S stands for synaptic) which evokes a fast (e.g., millisecond) action potential and (II) AH-neurons (AH stands for after hyperpolarization) which evoke longer lasting action potential (e.g., seconds) compared with the S-type.
The enteric neurons contain many peptides and neurotransmitters, which can be detected by various immunohistochemical methods. The enteric neurons can be classified on the basis of these chemical contents. For example, some enteric neurons contain the neurotransmitter acetylcholine. Such neurons are called cholinergic neurons and are generally stimulatory to gut activities. Other enteric neurons contain adrenaline (also known as epinephrine). These neurons are called adrenergic neurons and are generally inhibitory to gut activities.
Finally, the enteric neurons can be classified on the basis of their function. The basic functions of enteric neurons are excitatory, inhibitory, sensory or motor. Excitatory neurons cause an increase in secretion if they innervate a gland or cause muscle contraction if they innervate a muscle. Inhibitory neurons cause a decrease in secretion or muscle relaxation. Sensory neurons detect luminal pH and pressure or temperature in the gut wall, while motor neurons innervate muscles and sphincters and cause contraction or relaxation.
In general, Dogiel type I neurons and S-type enteric neurons are considered motor neurons, whereas Dogiel type II and AH-neurons are considered sensory neurons. The excitatory enteric neurons of the gut, which act in the orad (toward the mouth) direction, contain acetylcholine (Ach) and substance P (sub P), while the inhibitory enteric neurons of the gut, which act in the aborad (toward the anus) direction, contain Vasoactive Intestinal Peptide (VIP) and Nitric Oxide (NO). The inhibitory enteric neurons of the gut are also referred to as guards (organizers or regulators). These neurons are required to control spurious excitation or contraction if either of these processes occurs.
In addition to the three neuronal components of the ENS, myenteric and submucosal plexuses and interneurons, there is a special type of cell in the gut that is referred to as the interstitial cell of Cajal or ICC. The ICC is considered a specialized smooth muscle cell which has the ability to generate spontaneous electrical activity, pacemaker-like activity, known as slow waves. Structurally, the ICC contains multiple arms or projections which spread the slow waves to adjacent smooth muscle cells and enteric neurons. The meeting between the slow waves which are generated by the ICCs and the action potentials which is generated by the enteric neurons causes smooth muscle contraction in the gut.
The Endocrine System of the Gut
The second component of the intrinsic control mechanisms of the gut is the endocrine system of the gastrointestinal tract. The gastrointestinal tract contains millions of epithelial cells, which are referred to as enterocytes, enterochromaffin cells and endocrine cells. The enterocytes have an absorptive function, whereas the enterochromaffin cells are secretory in nature. For a gut peptide to be called a hormone it must fulfill certain criteria. These criteria consist of five characteristics. First, the gut hormone must be secreted by one cell in the gut and must affect another cell. Second, the vehicle that transports gut hormones from the secreting cell to the target cell must be the blood (endocrine route). Third, the release of gut hormones must be stimulated by food. Fourth, it is not necessary for gut hormones to be secreted under neuronal control. Fifth, a synthetic form of the hormone (e.g., as synthesized by a drug company), must be capable of mimicking the actions of the natural hormone. If a gut peptide fulfills these criteria, it is termed a gut hormone; otherwise, it retains reference as a gut peptide. Therefore, all gut hormones are also considered gut peptides, whereas not all gut peptides are gut hormones. The gut peptides that fulfill the previous criteria are secretin, gastrin, Cholecystokinin (CCK), gastric inhibitory polypeptide (also known as Glucose-dependent insulinotropic peptide, GIP) and motilin. The following section will discuss each of the hormones individually in more detail.
Secretin
In 1902, Bayliss and Starling discovered secretin, the first gut peptide hormone to be identified. Secretin is secreted by the S-cells of the duodenum and upper jejunum in response to fat and protein, gastric acid, bile acids and herbal extracts. Functionally, secretin stimulates exocrine pancreatic and biliary secretions of water and bicarbonate, gastric mucus and pepsinogen, endocrine pancreatic secretions of insulin, glucagon and somatostatin and pancreatic growth. In addition, secretin inhibits gastric acid secretion (functioning as a sort of nature’s anti-acid), as well as motility of the intestine and gastric mucosal growth. The regulation of secretin functions is controlled by the action of hormones such as CCK or by hormonal-neuronal interaction, such as CCK and the vagus nerve. Pathologically, hypersecretinemia can be found in cases of duodenal ulcer, renal failure, chronic pancreatitis and esophageal small cell carcinoma.
Gastrin
Gastrin (G) was discovered in 1905 by John Sidney Adkins as a hormone that is secreted by the G cells of the gastric pylorus, antrum and duodenum. Gastrin is secreted in response to the presence of protein and gastric distention. Its major action is to increase gastric acid secretion. Depending on the number of amino acids in the peptide chain, gastrin has two different forms, G-17 (also known as little gastrin) and G-34 (also known as big gastrin). These forms are equipotent. The gastric antrum produces G-17 in response to food (90% of gastrin production in the gut is G-17), whereas the duodenum produces G-34 between meals. If the sixth tyrosine residue from the C-terminus is sulfated, then gastrin is called Gastrin II. However, if this residue is not sulfated, then the peptide is called Gastrin I.
Gastrin I and II bind to the cholecystokinin2 (CCK2) receptor (also called the CCK-B receptor), a G-protein coupled receptor, with an affinity similar to CCK. This binding results in stimulation of gastric acid secretion and hyperplasia of the Enterochromaffin like cells (ECL) - a type of endocrine cell of the gastric mucosa. Increased plasma levels of gastrin, independent of food or increased gastric acidity, can be used as a diagnostic tool for cases of pernicious anemia or gastrinoma (a tumor that secretes gastrin). In addition, gastrin increases acid secretion indirectly by stimulation of histamine release from the ECL, which can activate histamine-2 (H2) receptors on acid secreting gastric parietal cells. Therefore, one of the ways that the inhibition of gastric acid secretion can be affected is through drugs that block the H2 receptor, such as cimetidine.
Cholecystokinin (CCK)
In 1928, Ivy and Oldberg discovered CCK, which is a hormone that is secreted by the endocrine I cells and the enteric neurons of the duodenum and jejunum in response to fat and protein [3]. Its major action is to stimulate gallbladder emptying and the secretion of pancreatic enzymes. Cholecystokinin controls many gastrointestinal functions by activating two G-protein coupled receptors: CCK1, formerly known as CCK-A (alimentary) receptor, which is distributed mainly in the alimentary tract, and CCK2, formerly known as CCK-B (brain) receptor, which is distributed mainly in the central nervous system. Sulfation (adding a sulfate group) of CCK affects the binding of the peptide to its receptors. Sulfated CCK has a 100 to 1000 times greater binding affinity to CCK1 receptors than the non-sulfated form of CCK or gastrin. Sulfated CCK, non-sulfated CCK and gastrin bind to CCK2 receptors with equal affinity.
Physiologically, CCK controls many gastrointestinal-related functions. For example, CCK causes gallbladder and smooth muscle contraction while increasing pancreatic secretion it also inhibits gastric emptying. Along with gastrin releasing peptide [2], CCK is one of the classical peptides that reduce food intake.
Gastric inhibitory polypeptide
In 1969, Brown and his colleagues discovered Gastric Inhibitory Polypeptide (GIP). Gastric inhibitory polypeptide is referred to as an enterogastrone because of its ability to reduce the rate of stomach emptying. Enterogastrone is a collective term that refers to any hormone or regulatory substance that slows the movement of ingesta, particularly from the stomach to intestine. The name glucose-dependent insulinotropic peptide is also given to GIP because its secretion is stimulated by the presence of glucose in the duodenum and one of its actions is to stimulate the secretion of insulin from the endocrine pancreas. This hormone is secreted by the K cells of the proximal small intestine in response to fat and glucose. Functionally, GIP inhibits gastric acid secretion and stimulates insulin secretion.
Motilin
In 1973, Brown and colleagues discovered motilin. This peptide is secreted by the M or Mo cells of the duodenum and, to a lesser extent, by the jejunum. Motilin works on both muscles and nerves to regulate the Migrating Motor Complex (MMC), the basic pattern of intestinal motility present during the period between meals that is interrupted by feeding. In addition, motilin stimulates gastric emptying during the between-meal period and secretion of pepsinogen, a protein-digesting enzyme of the stomach. Clinically, drugs that mimic the actions of motilin are used to treat gut motility disorders (hypokinetic disorders) including delayed gastric emptying.
The Immune System of the Gut
The mucosa of the gut is exposed to numerous microorganisms and antigens (e.g., contaminated food or toxins). Such harmful agents require a local defense system (immune system) to control their numbers and to limit their access to the body. The majority of the immune cells in the body reside in the gut mucosa. These immune cells defend the gastrointestinal milieu in two ways. First, the immune cells of the gut respond to antigenic stimulation similar to any other immune cells in the body including the creation of antigenic memory, neutralization, and the synthesis of antibodies and recruitment of killer cells. Second, the immune cells of the gut respond by secreting inflammatory mediators such as prostaglandins, histamine, and cytokines, which interact directly with the ENS and GI endocrine and paracrine cells. This interaction results in the modulation of gut functions such as motility and secretion. For example, when bad food or toxins enter the gut, the immune cells become sensitized and start secreting prostaglandins, cytokines and other immune mediators. These substances will then interact directly with the ENS, endocrine, and paracrine systems of the gut to evoke responses such as increased fluid secretion, dilution of toxin, and increased motility in order to move the harmful material quickly through the system. Therefore, the microbe or toxin will be washed and will eventually pass in the feces. As a result of these actions, the gut is protected.
The Extrinsic Innervations of the Gut
In addition to the intrinsic control systems of the gastrointestinal tract, the ENS and the endocrine system, there are two extrinsic systems, nerves and hormones that also participate in the regulation of the various functions of the gut. The extrinsic innervations that control the functions of the gastrointestinal tract consist of vagus and splanchnic nerves, while the extrinsic hormonal system consists of one hormone, aldosterone. The following sections will discuss each of these systems in more detail.
The vagus nerve
Anatomically, the vagus nerve has two components, parasympathetic efferents (nerve fibers that send orders from the brain to the gut) and vagal afferents (nerve fibers that send information from the gut to the brain). Functionally, the vagus nerve consists of two general types of nerve fibers, afferents (sensory), which carry the signal from the organs to the central nervous system, and efferents (motor), which carry orders from the central nervous system to the organs. Specific vagal fiber types most relevant to the GI tract are: General and Special Visceral Afferents (GVA and SVA), which innervate the abdominal viscera, including the gastrointestinal tract, as well as the pharyngeal mucosa; General and Special Visceral Efferents (GVE and SVE) projecting from the central nervous system to parasympathetic ganglia near organs the pharynx, respectively.
The cell bodies of the GVA are located in the inferior vagal ganglion (nodose ganglion), and the cell bodies of the GVE and SVE are located in the Dorsal Motor Nucleus of the Vagus (DMV) and nucleus ambiguus, respectively. The DMV is located in the dorsal vagal complex of the hindbrain, along with the nucleus of the solitary tract (Figure 1). The nucleus of the solitary tract receives vagal GVA input from the GI tract.
The vagus nerve innervates the gastrointestinal tract via two main branches, the left and right vagi. The left vagus branches into celiac and left gastric nerves, whereas the right vagus branches into hepatic, right gastric and accessory celiac nerves. Vagotomy of some of these branches can be used as an optional treatment for gastric/peptic ulcers.
Furthermore, the vagus nerve communicates with the ENS of the gut, which also communicates with the dorsal vagal complex of the CNS, through vagal afferents. These GVAs are Intravillus Arbors (IVA), interganglionic laminar endings (IGLE) and intramuscular arrays (IMA) (Figure 1). The IVAs reach the villi of the gut mucosa and function as chemoreceptors, providing information to the CNS about the chemical condition of the gut lumen. The IMIs and IGLEs act as mechanoreceptors, and stretch or tension receptors, which provide the CNS with information about the physical situation in the gut. The vagal GVEs that communicate with the gut ENS are parasympathetic pre-ganglionic neurons that synapse on neurons of the submucosal or myenteric plexuses. In general, this parasympathetic efferent control is stimulatory (i.e., they increase gut blood flow, motility, and glandular secretions).
The splanchnic nerve
The splanchnic nerve supplies the gastrointestinal tract with both sympathetic efferent and spinal afferent innervations. The sympathetic pre-ganglionic neuronal cell bodies are located in the thoraco-lumbar region of the spinal cord, and the cell bodies of afferents to the spinal cord lie in the dorsal root ganglia (Figure 1). Post-ganglionic sympathetic cell bodies reside in the Celiaco-Mesenteric Ganglia (CMG), and synapse at the target organ. The CMG, solar in shape, consist of two major ganglia, celiac and cranial (superior) mesenteric, as well as smaller ganglia with no specific names. These ganglia reside between the celiac and cranial mesenteric arteries, representing branches of the aorta. In general, the sympathetic neurocrine secretions are inhibitory in nature.
Splanchnic nerves, carrying visceral and spinal afferents, are distributed in the mucosa, muscularis, serosa and mesentery of the gut, and they carry signals to the CNS regarding the presence of pathological conditions in the gut. Such signals include the distension of the gut wall, inflammation or the presence of noxious chemicals or substances in the lumen of the gut with associated colic or abdominal pain. These painful stimuli evoke sympathetic responses in the gastrointestinal tract, including the inhibition of gut motility and increased glandular secretions.
The Extrinsic Endocrine System of the Gut: Aldosterone
Only one known hormone is secreted outside the gastrointestinal tract but still participates in controlling some of the functions of the gastrointestinal tract: aldosterone.
Aldosterone
In 1953, Simon and Tait isolated aldosterone. This hormone is a steroid hormone (mineralocortecoid) that is secreted by the outer zona glomerulosa section of the adrenal cortex following stimulation by a low-salt (low sodium) diet, angiotensin, adrenocorticotropic hormone or high potassium levels. The main function of aldosterone is to act on the distal convoluted tubules and collecting ducts of the kidney causing secretion of potassium and reabsorption of sodium and water, with attending increase in blood pressure.
In the gastrointestinal tract, aldosterone stimulates sodium and water reabsorption from the gut and salivary glands in exchange with potassium ions. In addition, although it is species-dependent, aldosterone promotes increased absorption of water and sodium in the proximal colon and decreased absorption in the distal colon.
In summary, this review outlined the five systems which control the various functions of the gastrointestinal tract. The presentation should serve both basic and clinical professionals in the field of gastroenterology.
Acknowledgement
Supported by the National Institutes of Health (NIH 1SC1GM092285-01A1) and The Birmingham Racing Commission.
References
- Herdt TH, Sayegh AI (2013) Regulation of the gastrointestinal functions. In: Cunningham's Textbook of Veterinary Physiology, Klein BG. St. Louis, Missouri Saunders 263-273.
- Sayegh AI (2013) The role of bombesin and gastrin releasing peptides in the short-term control of food intake. In: Progress in molecular biology and translational science, Tao Y-X. Elsevier 343-370.
- Sayegh AI (2013) The role of cholecystokinin receptors in the short-term control of food intake. In: Progress in molecular biology and translational science, Tao Y-X. Elsevier, 277-316.
- Sayegh AI, Reeve JR Jr, Lampley ST, Hart B, Gulley S, et al. (2005) Role for the enteric nervous system in the regulation of satiety via cholecystokinin-8. J Am Vet Med Assoc 226: 1809-1816.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 31184
- [From(publication date):
December-2012 - Nov 27, 2025] - Breakdown by view type
- HTML page views : 25474
- PDF downloads : 5710