Research Article Open Access
Extraction of Diosgenin, a Bioactive Compound from Natural Source Dioscorea alata Var purpurae
Heena J Shah and Lele SS*
Food Engineering and Technology Department, Institute of Chemical Technology, Matunga, Mumbai, India
- *Corresponding Author:
- Professor of Biochemical Engineering
Food Engineering and Technology Department
Institute of Chemical Technology
Matunga, Mumbai, 400019, India
Tel: +91-22-33611111
Fax: +91-22-33611020
E-mail: shah.heena146@gmail.com, dr.smita.lele@gmail.com
Received date: May 26, 2012; Accepted date: August 21, 2012; Published date: August 27, 2012
Citation: Shah HJ, Lele SS (2012) Extraction of Diosgenin, a Bioactive Compound from Natural Source Dioscorea alata Var purpurae. J Anal Bioanal Tech 3:141. doi: 10.4172/2155-9872.1000141
Copyright: © 2012 Shah HJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
Diosgenin, a commercially important bioactive sapogenin, extracted from tubers belonging to Dioscorea species, is used as a precursor in the manufacturing of sex hormones, oral contraceptives and other pharmaceutically important steroidal drugs. Diosgenin content of two Indian variety viz., D. deltoidea and D. prazeri ranges from 0.32 - 1%. With the view to commercially exploit a new source of diosgenin, a lesser explored tuber D. alata Var purpurae (purple yam), from Western Ghats, India was studied. The diosgenin content was determined from tuber (fresh and dried) and callus of D. alata by acidic hydrolysis of the glycosides and extraction with an organic solvent, followed by the HPTLC analysis. HPTLC mobile phase was optimized to toluene: ethyl acetate 7:3 v/v using the PRISMA model and diosgenin content was found to be 0.078, 0.133 and 0.048% respectively after extraction for 8 hrs in alcohol.
Keywords
D. alata var purpurae; Acid hydrolysis; Solvent extraction; HPTLC; PRISMA model; Diosgenin
Introduction
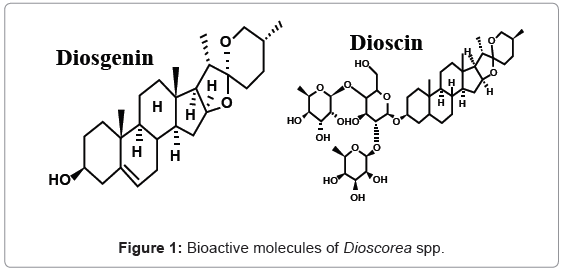
Diosgenin (Figure 1) is a bioactive steroidal sapogenin belonging to the triterpene group and is of great interest to the pharmaceutical industry [1]. It is the aglycone formed by the hydrolysis of saponin dioscin (Figure 1), a compound found in Dioscorea spp. [2]. It serves as an important starting material for the production of corticosteroids, sexual hormones, oral contraceptives as well as other steroidal drugs via hemisynthesis [3]. Different chemical and biological protocols are reported to extract diosgenin [4]. Past research shows various methods analyzing Diosgenin such as microscopy [5], spectrophotometeric [6], GLC [7], HPLC [8] and HPTLC [9].
In the present work, we studied extraction and estimation of Diosgenin content of the lesser explored tuber Diosocrea alata Var purpurae (purple yam), from Western Ghats, India [3]. We tried extracting diosgenin from fresh and dried tuber and callus developed by inoculating tuber as explant and optimizing the HPTLC method to analyze it.
Materials and Methods
Raw material
Dioscorea alata Var purpurae (purple yam) was procured from a fixed vendor in the local market of Mumbai. Tuber was dried in horizontal tray dryer and was pulverized to mesh size 40. Fresh tuber cortex was inoculated on Murashige and Skoog media to get callus by 8-10 days of inoculation.
Hydrolysis
Fresh tuber paste (5 gms), dried tuber powder (5 gms) and fresh callus powder (0.91 gms) were treated with slightly modified method described by Drapeau et al. [10]. Samples were hydrolyzed in 150 ml of refluxing 20% H2SO4 in 70% isopropanol for 8 h. The extract was filtered and extracted with hexane (50 ml×3). The three hexane extracts were combined and rinsed thrice times with 5% alkali and then rinsed thrice with distilled water. The extract was then passed through a column of Na2SO4 to eliminate any remaining water. The samples were concentrated to dryness by evaporating the solvent at 40°C in a Rotary evaporator. The dried crude extract was solubilized in 0.5 ml of chloroform prior to the quantitative determination of diosgenin by Thin layer chromatography. All the chemicals were of analytical grade from Himedia.
HPTLC analysis
Instrument used for HPTLC analysis was A CAMAG TLC system comprising of a Linomat-5 applicator and CAMAG TLC densitometer scanner of ANCHROM. Stationary phase used was silica gel 60 F254, 20×10 cm TLC plate obtained from Merck. Solvents Toluene (T), Ethyl Acetate (EA), Formic Acid (FA) and Glacial Acetic Acid (GAA) were of analytical grade from Himedia.
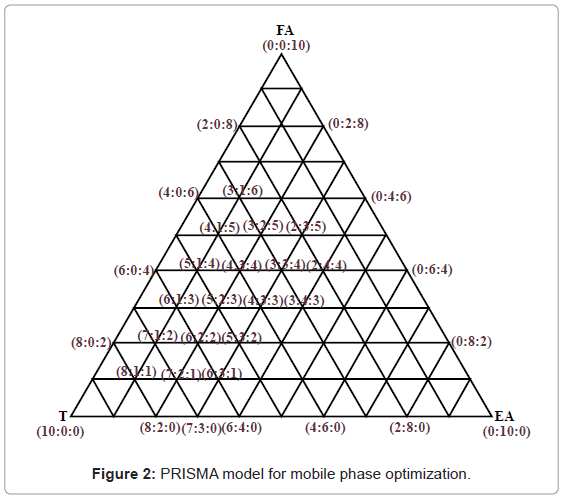
To begin the analysis, mobile phase T:EA:FA:GAA in the ratio 2:1:1:0.75 v/v reported by Kshirsagar et al. [9] was preferred. From the preliminary runs we came to the conclusion that GAA is not a significant component affecting the separation of Diosgenin. Remaining three mobile phase components where optimized by following the runs derived from PRISMA model (Figure 2).
For each run the factors kept constant were 1) Solvent front: 8 cm, 2) Time of run: 12-15 mins, 3) Developing reagent: Anisaldehyde in EA and conc. H2SO4, 4) Time of development: 15 min in oven at 60°C with air circulation, 5) Densitometry scan: 430 nm.
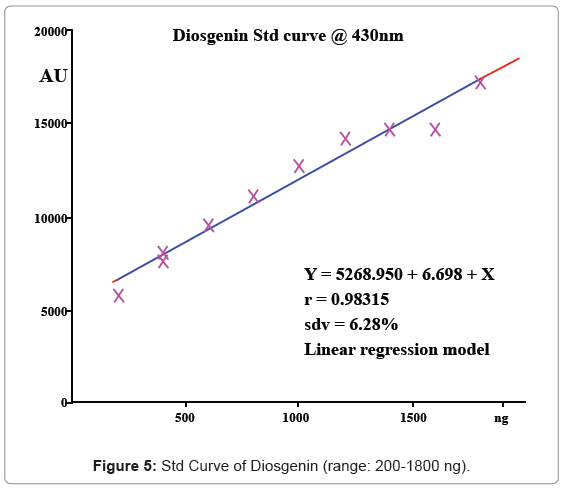
50 μgml-1 of working standard of Diosgenin (sigma-aldrich, 95% pure) prepared in chloroform was taken for the study. The calibration curve from 200-1800 ng/spot was prepared and checked for reproducibility, linearity and validating the method.
Validation
The developed method was validated in terms of linearity range, accuracy, limit of detection (LOD) and limit of quantification (LOQ).
Results and Discussion
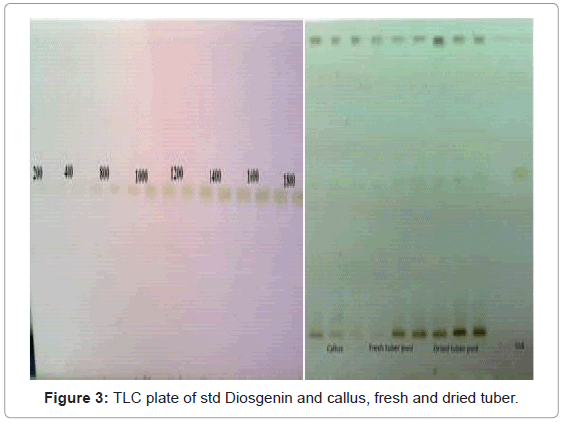
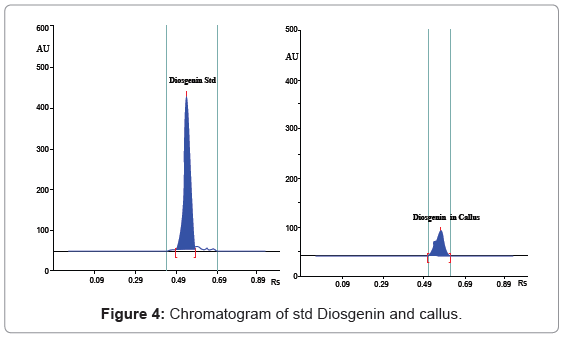
A solvent system giving clear sharp spots with Rf value in the range 0.45-0.65 was desired for quantification of Diosgenin (Figure 3). Table 1 shows that of all the mobile phase runs derived from PRISMA model, Toluene: Ethyl acetate (7:3 v/v) gave Rf value of 0.56 ± 0.01 for standard Diosgenin. Table 2 shows the densitometer scanning results at 430nm after development with the anisaldehyde reagent. Figure 4 is the chromatogram of standard and callus sample. The linear regression data (n=6) showed a good linear relationship over a concentration range of 200-1800 ng/spot with regression coefficient of 0.98315 and standard deviation of 6.28%. The calibration curve was prepared by plotting the concentration of Diosgenin versus average area of the peak (Figure 5).
| Sr. No. | Toluene | E A | F A | Mean Rf | SD | Comment |
|---|---|---|---|---|---|---|
| 1 | 3 | 2 | 5 | 0.69 | 0.00 | |
| 2 | 2 | 3 | 5 | 0.00 | 0.00 | Smudgy |
| 3 | 4 | 2 | 4 | 0.67 | 0.02 | |
| 4 | 3 | 3 | 4 | 0.85 | 0.00 | |
| 5 | 2 | 4 | 4 | 0.93 | 0.54 | Smudgy |
| 6 | 4 | 3 | 3 | 0.74 | 0.00 | |
| 7 | 3 | 4 | 3 | 0.92 | 0.01 | |
| 8 | 3 | 1 | 6 | 0.49 | 0.03 | Spread |
| 9 | 4 | 1 | 5 | 0.46 | 0.04 | |
| 10 | 5 | 1 | 4 | 0.43 | 0.01 | |
| 11 | 6 | 1 | 3 | 0.44 | 0.00 | |
| 12 | 5 | 2 | 3 | 0.59 | 0.00 | |
| 13 | 6 | 2 | 2 | 0.59 | 0.00 | |
| 14 | 7 | 1 | 2 | 0.43 | 0.01 | |
| 15 | 5 | 3 | 2 | 0.67 | 0.01 | Sharp |
| 16 | 8 | 1 | 1 | 0.44 | 0.08 | |
| 17 | 7 | 2 | 1 | 0.56 | 0.01 | Sharp |
| 18 | 6 | 3 | 1 | 0.62 | 0.00 | |
| 19 | 8 | 2 | 0 | 0.45 | 0.00 | |
| 20 | 7 | 3 | 0 | 0.56 | 0.01 | Sharp |
| 21 | 6 | 4 | 0 | 0.60 | 0.00 |
Table 1: PRISMA model runs.
| Track | Volume applied (μl) | Conc. (ng) | Area (AU) | X (calc., ng) | Remark |
|---|---|---|---|---|---|
| 1 | 4 | 200 | 5719 | ||
| 2 | 8 | 400 | 8060 | ||
| 3 | 12 | 600 | 9533 | ||
| 4 | 16 | 800 | 11063 | ||
| 5 | 20 | 1000 | 12745 | ||
| 6 | 24 | 1200 | 14241 | ||
| 7 | 28 | 1400 | 14727 | ||
| 8 | 32 | 1600 | 14733 | ||
| 9 | 36 | 1800 | 17186 | ||
| 10 | 5 | ** | 37600 | 36813.57 | Fresh tuber pwd |
| 11 | 5 | ** | 64113 | 63326.49 | Dried tuber pwd |
| 12 | 5 | ** | 4963 | 4176.67 | Fresh callus |
Table 2: Densitometer scanning results of std Diosgenin and samples.
The amount of Diosgenin in fresh and dried tuber as well as callus was computed from calibration curve and back calculated to infer the diosgenin content to be 0.078, 0.133 and 0.048% respectively. The developed method was validated as per ICH guidelines [11]. In Table 3 the percentage recovery was observed in the range from 96-98%. Other parameters of the validation study were 1) linearity range: 200-1800ng, 2) accuracy: ~97%, 3) limit of detection (LOD): 10 ng, 4) limit of quantification (LOQ): 20 ng.
| Sample | Diosgenin (ng) | STD added (ng) | Total Conc. | Actual Conc. | % Recovery |
|---|---|---|---|---|---|
| Fresh tuber pwd | 36813.57 | 100 | 36913.57 | 36909.75 | 96.18 |
| Dried tuber pwd | 63326.49 | 100 | 63426.49 | 63425 | 98.51 |
| Fresh callus | 4176.67 | 100 | 4276.67 | 4274.38 | 97.71 |
Table 3: Recovery study of Diosgenin.
Conclusion
he proposed extraction and HPTLC method resulted in a simple, faster and cost effective quantitative method for routine analysis of Diosgenin. From validation studies we concluded that the method is showing good performance. Dioscorea alata Var Purpurae, a lesser explored tuber from Western Ghats of India was confirmed as a probable source of Diosgenin. Diosgenin was quantified from the fresh tuber, dried tuber powder and its callus. Further increase in diosgenin content, by studying the effect of precursor, additive and elicitor in the callus, can be checked.
Acknowledgements
This work was supported by the “UNIVERSITY GRANTS COMMISSION - SPECIAL ASSISTANCE PROGRAMME (UGC-SAP)” India.
References
- Oncina R, Botia JM, Del Rio JA, Ortuno A (2000) Bioproduction of diosgenin in callus cultures of Trigonella foenum-graecum L. Food Chemistry70: 489-492.
- Olayemi JO, Ajaiyeoba EO (2007) Anti-inflammatory studies of yam (Dioscorea esculenta) extract on wistar rats. African Journal of Biotechnology 6: 1913-1915.
- Burkill IH (1960) The organography and the evolution of Dioscoreaceae, the family of the Yams. J Linn Soc (Bot) 56: 319-412.
- Zhang YQ, Liang JH, Fu EH, Li BX (2007) Effect of Modified Enzymatic Catalysis on the Extraction of Diosgenin from Dioscorea zingiberensisC. H. Wright. Chem Eng Technol 30: 1488-1494.
- Ravishankar GA, Grewal S (1991) A simple method for selection of high diosgenin yielding clones of Dioscorea deltoidea callus. Biotechnology Techniques5: 281-282.
- Sánchez GL, Medina Acevedo JC, Soto RR (1972) Spectrophotometric determination of diosgenin in Dioscorea compositafollowing thin-layer chromatography. Analyst 97: 973-976.
- Cooke BK (1970) Determination of diosgenin in Dioscorea deltoidea and Dioscorea sylvatica by using gas-liquid chromatography. Analyst 95: 95-97.
- Niño J, Jiménez DA, Mosquera OM, Correa YM (2007) Diosgenin quantification by HPLC in Dioscorea polygonoides tuber collection from Colombian flora. J Braz Chem Soc18: 1073-1076.
- Kshirsagar VB, Deokate UA, Bharkad VB, Khadabadi SS (2008) HPTLC method development and validation for the simultaneous estimation of Diosgenin and Levodopa in marketed formulation. Asian J. Research Chem1: 36-39.
- Drapeau D, Sauvaire Y, Blanch HW, Wilke CR (1986) Improvement of diosgenin yield from Dioscorea deltoideaplant cell culture by use of a non-traditional hydrolysis method. Planta Med 6: 474-478.
- ICH guidelines Q2A (1994) Text on Validation of Analytical Procedure. International Conference of Harmonization, Geneva, 1-5.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 26885
- [From(publication date):
August-2012 - Oct 19, 2025] - Breakdown by view type
- HTML page views : 21172
- PDF downloads : 5713