Review Article Open Access
Hereditary Nonpolyposis Colorectal Cancer (Lynch Syndrome)
| Jaime Ruiz-Tovar*, Antonio Arroyo and Rafael Calpena | |
| Coloproctology Unit, General University Hospital Elche (Alicante-Spain) | |
| Corresponding Author : | Jaime Ruiz-Tovar, MD, PhD Corazon de Maria, 64 7º J, 28002-Madrid (Spain) Tel: (0034) 63 053 4808 E-mail: jruiztovar@gmail.comu |
| Received November 26 , 2011; Accepted January 28, 2012; Published January 30, 2012 | |
| Citation: Tovar JR, Arroyo A, Calpena R (2012) Hereditary Nonpolyposis Colorectal Cancer (Lynch Syndrome). J Gastroint Dig Syst S6:003. doi:10.4172/2161-069X.S6-003 | |
| Copyright: © 2012 Tovar JR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Colorectal cancer (CRC) is the 3rd leading cause of cancer-related death in Western countries. Between 3-8% of CRC appear as hereditary forms, being the Lynch syndrome (hereditary nonpolyposis colorectal cancer) the most frequent one. The main features of Lynch syndrome are the presentation at young ages (mean 45 years), the preferent arisal in right colon (70% of the cases), the high incidence of synchronic (10%) and metachronic (30-50%) colorectal tumors, and the association with extra colonic neoplasms (mainly adenocarcinomas in endometrium, small bowel, ovarium, stomach, ureter and bladder). Clinical diagnosis is suspected because of the familial history and it is confirmed when identifying germline mutations in the DNA repair genes (mainly MLH1 and MSH2). These mutations determine a genomic instability state known as microsatellites instability, present in over 85% of tumours belonging to the Lynch syndrome and 10-15% of sporadic neoplasms. Identification of mutations and determination of microsatellites instability imply molecular technology, expensive and with limited availability. The immunohistochemical analysis of expression of mostly affected proteins (MLH1 and MSH2) can be an interesting option.
| Keywords |
| Hereditary nonpolyposis colorectal cancer; Lynch syndrome; MLH1; MSH2 |
| Introduction |
| Colorectal cancer (CRC) is the 3rd leading cause of cancer-related death in Western countries. Between 3-8% of CRC appear as hereditary forms, being the Lynch syndrome (hereditary nonpolyposis colorectal cancer) the most frequent one, representing between 2-4% of all the cases of CRC diagnosticated [1,2]. Lynch syndrome is an autosomic dominant disorder, characterized by a predisposition to develop certain types of neoplasms, being the most relevant ones colorectal and endometrial adenocarcinomas [3]. |
| Clinical Features |
| Despite patients with Lynch syndrome can develop colonic adenomas more frequently than general population, colonic polyposis is very unfrequent, because colorectal tumors do not usually arise on adenomatous polyps as happens in other cases of hereditary CRC (Familial adenomatous polyposis) [1,4]. It is estimated that between 50- 80% of patients diagnosed of Lynch syndrome will develop a CRC along their life [5]. |
| The main features of Lynch syndrome are the presentation at young ages (mean 45 years), the preferent arisal in right colon (70% of the cases), the high incidence of synchronic (10%) and metachronic (30-50%) colorectal tumors, and the association with extracolonic neoplasms (mainly adenocarcinomas in endometrium, small bowel, ovarium, stomach, ureter and bladder) [1,3,4]. |
| Histologically, the tumors are frequently poorly differentiated, mucinous and presenting an important lymphocytic infiltration. It is very frequent a high grade of microsatellites instability in the neoplastic cells, typical feature of tumors appearing as a consequence of mutations in DNA repair genes. It has been determined that those tumors with microsatellites instability present a better outcome than those that do not have [6]. |
| Subjects with Lynch syndrome tend to develop diverse neoplasms. Endometrial cancer is the most frequent extra colonic tumor, appearing in 40-60% of women with Lynch syndrome along their life. Moreover, 2% of all endometrial neoplasms are associate with Lynch syndrome [7]. Other related tumours are gastric, ovarian, bile duct, bladder, ureter, small bowel, cranial and pancreatic neoplasms [8,9] (Table 1). |
| Genetic Features |
| Lynch syndrome arises from germline mutations in DNA repair genes, especially MSH2, MLH1, MSH6 and PMS2. DNA repair system is essential for maintaining the genomic fidelity, solving errors appearing during the replication, mainly nucleotide changes in base pairs and insertions or deletions of fragments [1,2]. MSH2 & MLH1 mutations are responsible of up to 90% of cases of Lynch syndrome and MSH6 mutations represent 10% of cases, being PMS2 ones very unfrequent [8,10]. The different mutations have been associated with different clinical manifestations: MSH6 mutations are associated with a higher risk of developing endometrial cancer but lower risk of colorectal one, when compared with MSH2 and MLH1 mutations [11]. Carriers of PMS2 mutations present a risk of 15-20% of developing colorectal cancer, 15% of endometrial one and 25-32% of developing any of the other tumors associated with Lynch syndrome [12]. |
| Germline deletions in the gene codifying the adhesion molecule to epithelial cells (EpCAM) have been recently discovered in families with Lynch syndrome that did not present mutations in the DNA repair genes. These families developed an early beginning Lynch syndrome phenotype with multiple tumors. It is remarkable the presence of a hypermethilation of the MSH2 gene in the tumors arising in these subjects, a fact that is extremely unfrequent in colorectal cancer. Moreover, it has been observed in these families a germline deletion in the 3´ region of the EpCAM gene, resulting in a fusion EpCAM-MSH2 during the transcription. It has been estimated that this deletion may represent around 6% of cases of Lynch syndrome [13,14]. |
| Diagnosis |
| Actually, diverse tools allow the diagnosis of Lynch syndrome, including familial history analysis, tumoral analysis, predictive models of mutations and genetic evaluations. |
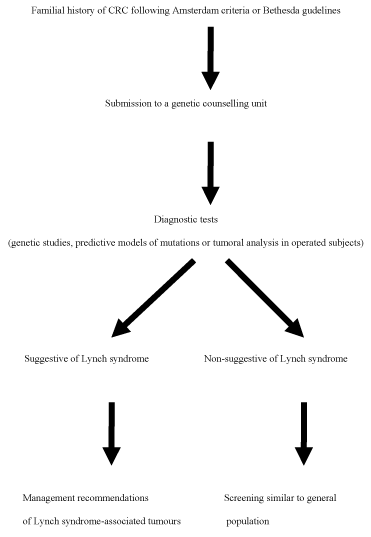
| It is quite important to obtain detailed information about personal and familial history. Some data may reflect that the patient presents a high risk to develop colorectal cancer and must be submitted to a genetic counselling unit. Amsterdam criteria were established to identify families susceptible to present a Lynch syndrome, but it has been observed that over 50% of the families do not meet these criteria. Therefore, Amsterdam Criteria II and Bethesda Guidelines were developed to increase the sensibility. Both, Amsterdam Criteria and Bethesda Guidelines, are actually used in the clinical practice to identify risky subjects to have a Lynch syndrome (Table 2) [1]. |
| There are commercial tests to analyze the distal segment of EpCAM and the 4 more relevant DNA repair genes. These genetic tests are performed when the patients present clinical or familial criteria of Lynch syndrome. Given that most mutations occur in MLH1 and MSH2, genetic tests begin with their analysis. Limits of these protocol are the high economic costs and low sensibility [1,2]. |
| Other identification method, with better cost-effectivity relationship, is the performance of a genetic study in the resected tumoral tissue in patients presenting the criteria of Bethesda Guidelines. This strategy begins with the determination of microsatellites instability and immunohistochemical analysis in colorectal tumours. Nearly 90% of CRC will present microsatellites instability, implying a high sensibility of this method. However, specificity is much lower, given that around 15% of sporadic CRC also presents microsatellites instability, as a result of the hypermethilation on the promoter region of MLH1 gene; in Lynch syndrome neoplasms the microsatellites instability is secondary to germ line mutations of the gene [1]. |
| Immunehistochemical methods use 4 specific antibodies for the proteins codified in MLH1, MSH2, MSH6 and PMS2 genes, determining a lack of these proteins. Sensibility of this method is similar to the microsatellites instability analysis [1]. |
| The analysis in endometrial neoplasms has demonstrated to be as effective as those performed in CRC [7]. Other tumors associated with Lynch syndrome present sometimes microsatellites instability and lack of proteins codified in DNA repair genes, but the efficiency of performing diagnostic tests in them has not been clearly established. Recently, Lynch syndrome predictive models have been developed, highlighting PREMM [1,2], MMRpro and MMRpredict. These models use clinical data of the patients and their familial history to estimate the probability of a subject to present a mutation in any DNA repair gene. These models have been validated in CRC and in endometrial tumours [15]. |
| Clinical Management |
| Early identification of subjects with Lynch syndrome is important because of the early beginning of the disease, the high penetrancy of neoplasms and the efficacy of screening for the different tumors. Close surveillance has demonstrated to reduce the incidence of CRC and the consequent diseases. |
| Performing a colonoscopy every 3 years has reduced the CRCrelated mortality over 50% [16]. This reduction is estimated to be higher when performing colonoscopies every 1-2 years. Moreover, continuous aspirin intake seems to decrease the risk of development of CRC in patients with Lynch syndrome and a recommendation can be therefore established [17]. Screening with colonoscopy must begin at age 20- 25 and be repeated every 1-2 years. When a neoplasm is diagnosed, subtotal colectomy with ileorectal anastomosis is recommended, with annual rectoscopy as follow-up [1]. |
| Prophylactic hysterectomy with bilateral salpingo-oophorectomy reduces the incidence of endometrial and ovarian cancer in females with Lynch syndrome and must be taken into consideration as an option in women who have completed their reproductive desires [18]. Screening of these tumours is also used, but is less effective than in CRC [19]. The measure with greater net benefit consists in an annual screening beginning at age 30 with prophylactic surgery at age 40 [19]. Despite there is little evidence about the effectiveness of screening for other tumours associated to Lynch syndrome, NCCN recommends some options depending on the clinical needs (Table 3) (Figure 1) [20]. |
References
- Jasperson KW, Tuohy TM, Neklason DW, Burt RW (2010) Hereditary and familial colon cancer. Gastroenterology 138: 2044-2058.
- Boland CR, Goel A (2010) Microsatellite instability in colorectal cancer. Gastroenterology 138: 2073-2087.
- Lichtenstein P, Holm NV, Verkasalo PK, Iliadou A, Kaprio J, et al. (2003) Environmental and hereditable factors in the causation of cancer- analysis of cohorts of twins in Sweden, Denmark and Finland. N Engl J Med 343: 78-85.
- Lynch HT, de la Chapelle A (2003) Hereditary Colorectal Cancer. N Engl J Med 348: 919-932.
- Stoffel E, Muklherjee B, Raymond VM, Tayob N, Kastrinos F, et al. (2009) Calculation of risk of colorectal and endometrial cancer among patients with Lynch syndrome. Gastroenterology 137: 1621-1627.
- Ribic CM, Sargent DJ, Moore MJ, Thibodeau SN, French AJ, et al. (2003) Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N Engl J Med 349: 247-257.
- Hampel H, Frankel W, Panescu J, Lockman J, Sotamaa K, et al. (2006) Screening for Lynch syndrome (hereditary nonpolyposis colorectal cancer) among endometrial cancer patients. Cancer Res 66: 7810-7817.
- Rustgi AK (2007) The genetics of hereditary colon cancer. Genes Dev 21: 2525-2538.
- Kastrinos F, Mukherjee B, Tayob N, Wang F, Sparr J, et al. (2009) Risk of pancreatic cancer in families with Lynch syndrome. JAMA 302: 1790-1795.
- Peltomaki P, Vasen H (2004) Mutations associated with HNPCC predisposition – update of ICG.HNPCC/INSIGHT mutation database. Dis Markers 20: 269-276.
- Plaschke J, Engel C, Krüger S, Holinski-Feder E, Pagenstecher C, et al. (2004) Lower incidence of colorectal cancer and later age of disease onset in 27 families with pathogenic MSH6 germline mutations compared with families with MLH1 or MSH2 mutations: the German Hereditary Nonpolyposis Coloretal Cancer Consortium. J Clin Oncol 22: 4486-4494.
- Senter L, Clendenning M, Sotamaa K, Hampel H, Green J, et al. (2008) The clinical phenotype of Lynch syndrome due to germline PMS2 mutations. Gastroenterology 135: 419-428.
- Kovacs ME, Papp J, Szentirmay Z, Otto S, Olah E (2009) Deletions removing the last exon of TACSTD1 constitute a distinct class of mutations predisposing to Lynch syndrome. Hum Mutat 30: 197-203.
- Niessen RC, Hofstra RM, Westers H, Ligtenberg MJ, Kooi K, et al. (2009) Germline hypermethylation of MLH1 and EPCAM deletions are a frequent case of Lynch syndrome. Genes Chromosomes Cancer 48: 737-744.
- Backes FJ, Leon ME, Ivanov I, Suarez A, Frankel WL, et al. (2009) Prospective evaluation of DNA mismatch repair protein expression in primary endometrial cancer. Gynecol Oncol 114: 486-490.
- Stupart DA, Goldberg PA, Algar U, Ramesar R (2009) Surveillance colonoscopy improves survival in a cohort of subjects with a single mismatch repair gene mutation. Colorectal Dis 11: 126-130.
- Järvinen HJ, Aarnio M, Mustonen H, Aktan-Collan K, Aaltonen LA, et al. (2000) Controlled 15-year trial on screening for colorectal cáncer in families with hereditary nonpolyposis colorectal cancer. Gastroenterology 118: 829-834.
- Schmeler KM, Lynch HT, Chen LM, Munsell MF, Soliman PT, et al. (2006) Prophylactic surgery to reduce the risk of gynecologic cancers in the Lynch syndrome. N Engl J Med 354: 261-269.
- Kwon JS, Sun CC, Peterson SK, White KG, Daniels MS, et al. (2008) Cost-effectiveness analysis of prevention strategies for gynecologic cancers in Lynch syndrome. Cancer 113: 326-335.
- National Comprehensive Cancer Network (NCCN) (2010) Clinical practice guidelines in oncology: colorectal cancer screening.
Tables and Figures at a glance
| Table 1 | Table 2 | Table 3 |
Figures at a glance
 |
| Figure 1 |
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 13345
- [From(publication date):
specialissue-2013 - Nov 24, 2025] - Breakdown by view type
- HTML page views : 8748
- PDF downloads : 4597
