Research Article Open Access
Magnetic Resonance Imaging in Assessment of Anorectal Fistulae and its Role in Management
Manar T Alaat El Essawy*
Department of Radiology and Diagnostic Imaging, Damietta Oncology Institute, Damietta, Saudi Arabia
- *Corresponding Author:
- Manar T Alaat El Essawy
Department of Radiology and Diagnostic Imaging
Damietta Oncology Institute
P.O. Box 361680, Riyadh 11313
Saudi Arabia
Tel: 00966114709173, 00966500471079
E-mail: manar970@hotmail.com
Received date: August 14, 2013; Accepted date: September 16, 2013; Published date: September 18, 2013
Citation: Alaat El Essawy MT (2013) Magnetic Resonance Imaging in Assessment of Anorectal Fistulae and its Role in Management. J Gastroint Dig Syst 3:139. doi:10.4172/2161-069X.1000139
Copyright: © 2013 Alaat El Essawy MT. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Objectives: This study was conducted to assess the diagnostic accuracy of magnetic resonance imaging (MRI) in perianal fistula in comparison to the operative findings with a description of the technique, illustration of relevant normal anatomy, and examples of various fistula types. This study is based on a retrospective analysis of 56 patients with a clinical diagnosis of perianal fistula. MRI finding was correlated with the surgical finding in 52 patients which is highly significant (P=0.003). Fallacies of MRI were observed in 4 patients with recurrent fistulae. Conclusions: MRI is useful in successful treatment of perianal fistulae by reliable assessment of fistula anatomy and correct assessment of the extent of the disease and its relation to the sphincter complex.
Keywords
Perianal fistula; Anal canal; Levatorani
Introduction
A perianal fistula is an inflammatory condition that affects the region around the anal canal with a presence of a fistulous tract across the anal sphincters. Perianal fistula has a high tendency to recur because of undetected infection at surgery, causing significant morbidity and often requiring repeated surgical treatments [1].
Failure to recognize occult lesions result in recurrent fistula and convert a simple fistula into a complex fistulizing process .Once the fistulizing process becomes complex , the chance of healing is greatly reduced [2].
Anatomy
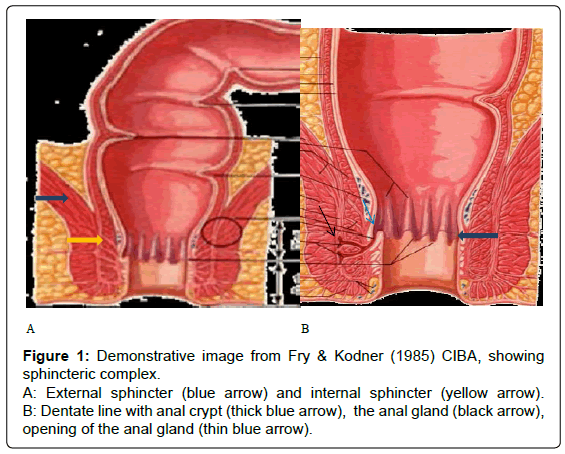
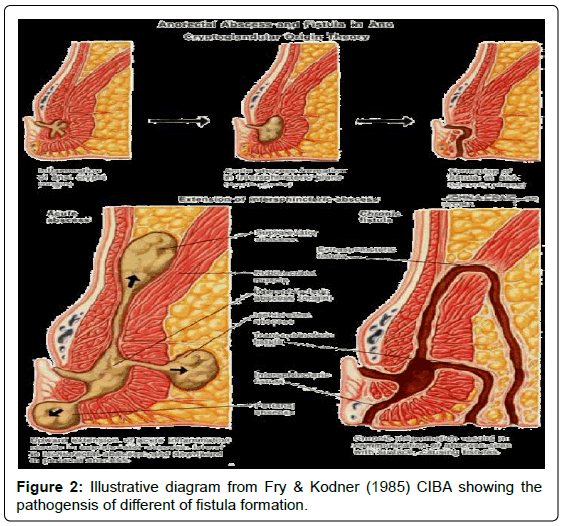
Knowledge of the anatomy of the anal sphincter complex and surrounding spaces is crucial for image interpretation. The anal canal extends from the levatorani muscle cranially to the anal verge caudally and is surrounded by the internal and external anal sphincters. The internal sphincter is the inferior extension of the inner circular smooth muscle of the rectum and is primarily responsible for resting involuntary anal continence. The external sphincter is composed of striated skeletal muscle, which is contiguous with both the levatorani and puborectalis muscles superiorly and is primarily responsible for voluntary continence (Figure 1A) [3]. The mucosal surface of the upper half of the anal canal shows longitudinal mucosal folds which are the anal columns of Morgagni. They end in a small semi lunar fold (the anal valves), which form pockets (crypts) of Morgagni, located at the dentate line which is about 2 cm far from the anal verge [4].The dentate line is an important landmark, as being the site of opening of the anal glands (Figure 1B); it is considered the initial site of infection which initiates the fistula formation. The anal glands being deeply located in the intersphincteric space are claimed for abscess formation secondary to any obstruction of their ducts’ outlet openings. The infection process thereafter, spreads through the intersphincteric space, downward or outward delineating the different types of fistula (Figure 2) [5].
Normal MRI anatomy of the anal sphincter
The external anal sphincter (a striated muscle) is clearly visualized on MRI. It is hypo intense on T1W, T2W, and fat-suppressed T2W images, and is bordered laterally by the fat in the ischioanal fossa.
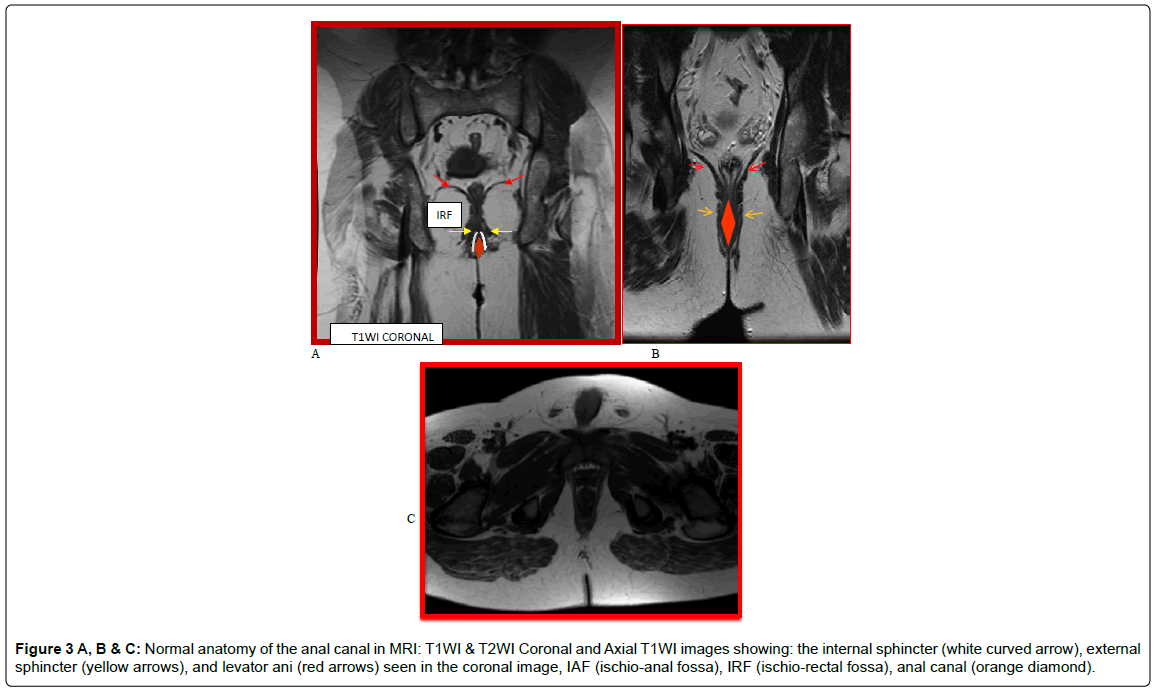
The internal sphincter (a smooth muscle) is hypo intense on T1W and T2W TSE images and is relatively hyper intense on fat-suppressed T2W images (Figures 3A,B and C). It shows enhancement on postgadolinium T1W images
Figure 3 A, B & C: Normal anatomy of the anal canal in MRI: T1WI & T2WI Coronal and Axial T1WI images showing: the internal sphincter (white curved arrow), external sphincter (yellow arrows), and levator ani (red arrows) seen in the coronal image, IAF (ischio-anal fossa), IRF (ischio-rectal fossa), anal canal (orange diamond).
The coronal images depict the levatorani muscle (Figure 3A), the identification of which is important to distinguish supralevator from infralevator infections.
Classification of the perianal fistulae
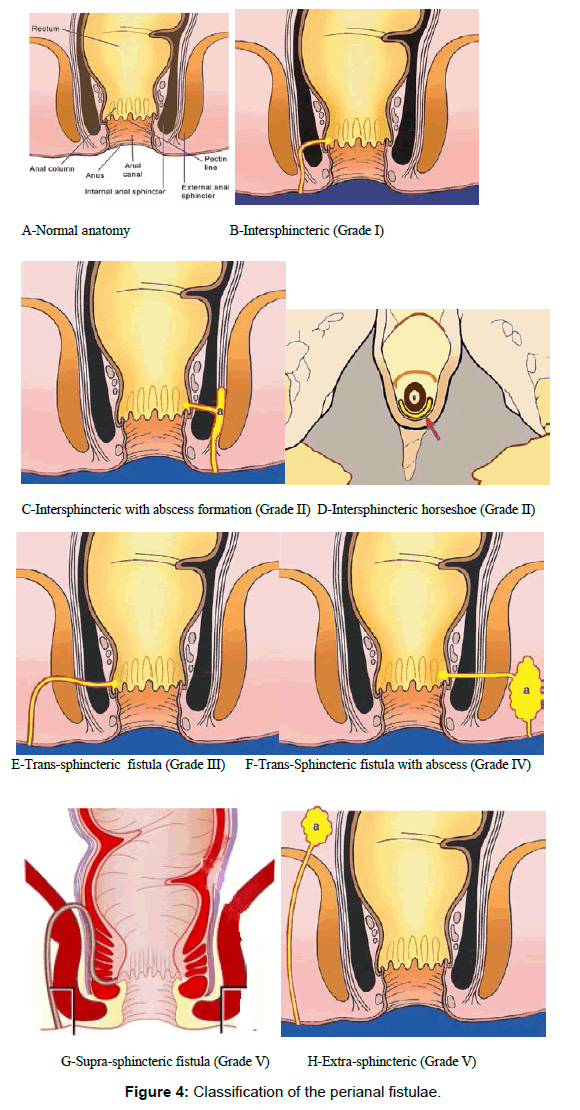
The anatomic course of an anal fistula will be dictated by the location of the infected anal gland. There will usually be an internal opening in the anal canal at the level of the dentate line. In most cases this is at the 6-o’clock position, because anal glands are more abundant posteriorly (radial positions around the anus are referenced with respect to a clock face, with 12 o’clock being directly anterior) [6]. The fistula can reach the perianal skin by a variety of routes [1]. Fistulas may thus be classified according to the route taken by this “primary tract” that links the internal and external openings (Figure 4A) [2].
Initial classification of perianal fistulas was based on surgical anatomy described by Parks et al. [7]. Morris et al. [8] subsequently modified the classification system on the basis of radiologic anatomy on pelvic MRI, which is known as the St. James’ University Hospital Classification of perianal fistula is divided as:
Grade 1: Simple linear intersphincteric fistula
A grade 1 fistula arises from the anal canal, penetrates the internal anal sphincter, and extends caudally through the intersphincteric space to its cutaneous opening (Figure 4B). There are no secondary tracts, cranial extension above the levatorani, or associated abscess. The tracts are confined by the external sphincter with no disruption of the external sphincter.
Grade 2: Intersphincteric fistula with intersphincteric abscess (Figure 4C) or secondary fistulous tract confined by the external sphincter. The “horse-shoe” fistula is a subtype (Figure 4D) that crosses the midline and surrounds both sides of the internal sphincter.
Grade 3: Transsphincteric fistula
A grade 3 fistula arises from the anal canal, penetrates both the internal and external anal sphincters, and extends through the ischiorectal or ischioanal fossae in its pathway to its cutaneous opening (Figure 4E). There is no associated secondary tract, abscess, or extension cranial to the levatorani.
Grade 4: Transsphincteric fistula with abscess (Figure 4F) or secondary tract within the ischioanal or ischiorectal fossa.
Grade 5: Supralevator and extrasphincteric fistula
Grade 5 fistulas encompass a variety of complex tracts characterized by their extension above the sphincteric complex. Suprasphincteric fistulas (Figure 4G) arise from the anal canal, penetrate the internal sphincter, and then ascend within the intersphincteric space into the supralevator space. The fistula then pierces the levatorani as it descends through the ischiorectal fossae en route to its cutaneous opening. Extrasphincteric fistulas (Figure 4H) are a separate entity caused by primary pelvic disease (e.g., Crohn disease, diverticulitis, or carcinoma) that extends caudally through the levator plate, traverses the ischiorectal fossa, and terminates at the cutaneous opening. Most importantly, an extrasphincteric fistula does not involve the internal or external anal sphincters [3].
Materials and Methods
We included in this study all patients, who had been admitted to colorectal unit with peri-anal fistula in 2009. After preoperative clinical examination, MR imaging was done using a 1.5 and 3 Tesla superconducting magnet with external coil. Imaging was done using inversion recovery (STIR), T2WI, T1WI and T1WI post contrast with fat suppression sequences in axial, coronal and sometimes sagittal planes. MRI fistulogram with instillation of contrast (mixture of 1 ml of 0.01 mmol gadolinium with 20 ml saline) or saline through the external opening was performed in some cases. Slice thickness was 4 mm, F.O.V. was 25×25. Imaging matrix was 256×256. The images were evaluated for the presence of the primary fistulous tract, Internal opening and its relations to the sphincters. Secondary extensions, any abscesses or collections also were recognized. The fistula is identified as hyper intense tubular structures on T2WI and abscess appears as fluid filled cavities either high or low signals on T2WI.
These findings were compared to the operative findings.
Results
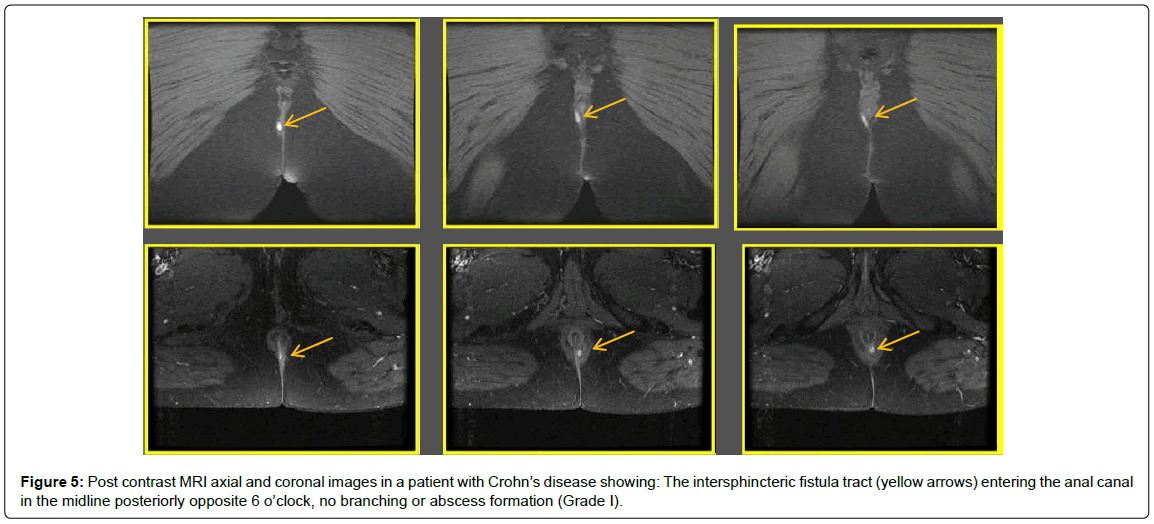
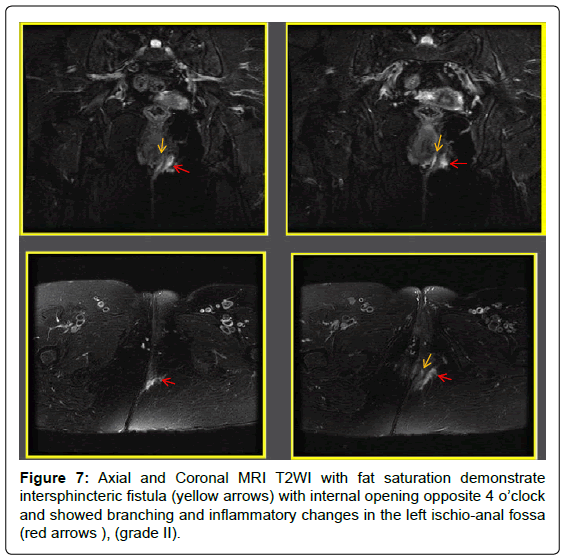
56 patients were included in the study, 46 males and 10 females with mean age 39, 3 years +/-11, 44 SD (Range between 14-56 years). Primary fistulae were recorded in 40 cases and 16 cases were recurrent. MRI findings were as follow (Table 1) inter-sphincteric (33-58.9% cases) (Figures 5-8), trans-sphincteric (14-25%-cases) (Figures 9-11), suprasphincteric (Figure 12 and 13), and extrasphincteric (7-12.5%- case) (Figures 14 and 15). With MRI, horse-shoe extension was detected in 5(8.9%) cases (Figure 8) and abscess cavity was seen in 20(35.7%) cases (15 ischiorectal and 5 supralevator) (Figures 11,14 and 15). MRI findings were concordant with the surgical findings in 52 patients (92.8%) which is highly significant (P=0.003). Fallacies of MRI were observed in 4(7.1%) patients who had a history of Crohn’s disease and recurrent fistulae (Table 1).
| Abnormality Classifications | MR Imaging Findings | Surgical Findings Reference Standard | |||
|---|---|---|---|---|---|
| True-positive | False-negative | False-positive | True-negative | ||
| Primary Track | |||||
| Intersphincteric | 31 | 2 | 0 | 33 | |
| Transsphincteric | 14 | 0 | 2 | 16 | |
| Suprasphincteric | 4 | 0 | 0 | 4 | |
| Extrasphincteric | 3 | 0 | 0 | 3 | |
| Detection Total | 52 | 2 | 2 | 54 | 56 |
| Abscess | |||||
| Intersphincteric | 15 | 0 | 0 | 15 | |
| Supralevator | 5 | 0 | 0 | 5 | |
| Horseshoe Fistula | |||||
| Infralevator (ischiorectal or Intersphincteric fossa) | 4 | 0 | 0 | 4 | |
| Supralevator | 1 | 0 | 0 | 1 | |
| Secondary internal opening | 18 | 18 | |||
Table 1: MRI findings compared to surgical assessment.
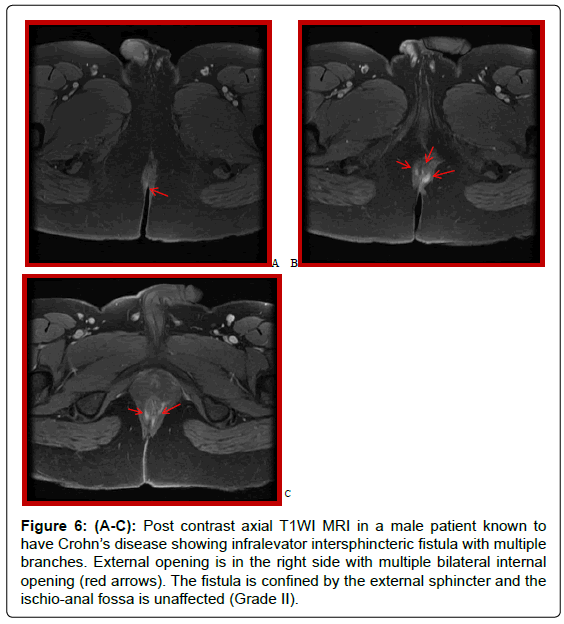
Figure 6: (A-C): Post contrast axial T1WI MRI in a male patient known to have Crohn’s disease showing infralevator intersphincteric fistula with multiple branches. External opening is in the right side with multiple bilateral internal opening (red arrows). The fistula is confined by the external sphincter and the ischio-anal fossa is unaffected (Grade II).
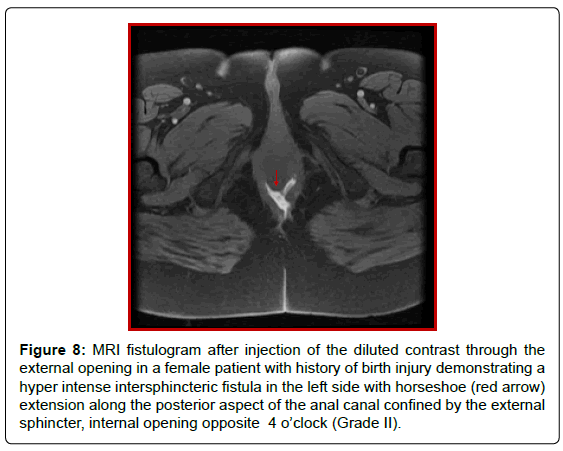
Figure 8: MRI fistulogram after injection of the diluted contrast through the external opening in a female patient with history of birth injury demonstrating a hyper intense intersphincteric fistula in the left side with horseshoe (red arrow) extension along the posterior aspect of the anal canal confined by the external sphincter, internal opening opposite 4 o’clock (Grade II).
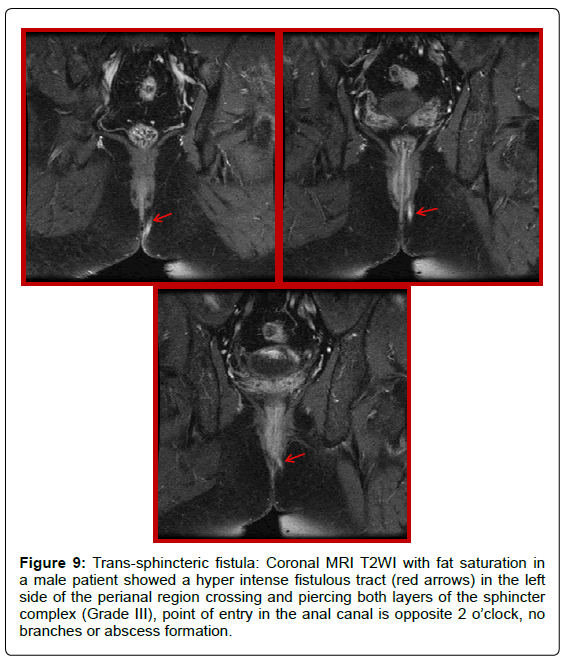
Figure 9: Trans-sphincteric fistula: Coronal MRI T2WI with fat saturation in a male patient showed a hyper intense fistulous tract (red arrows) in the left side of the perianal region crossing and piercing both layers of the sphincter complex (Grade III), point of entry in the anal canal is opposite 2 o’clock, no branches or abscess formation.
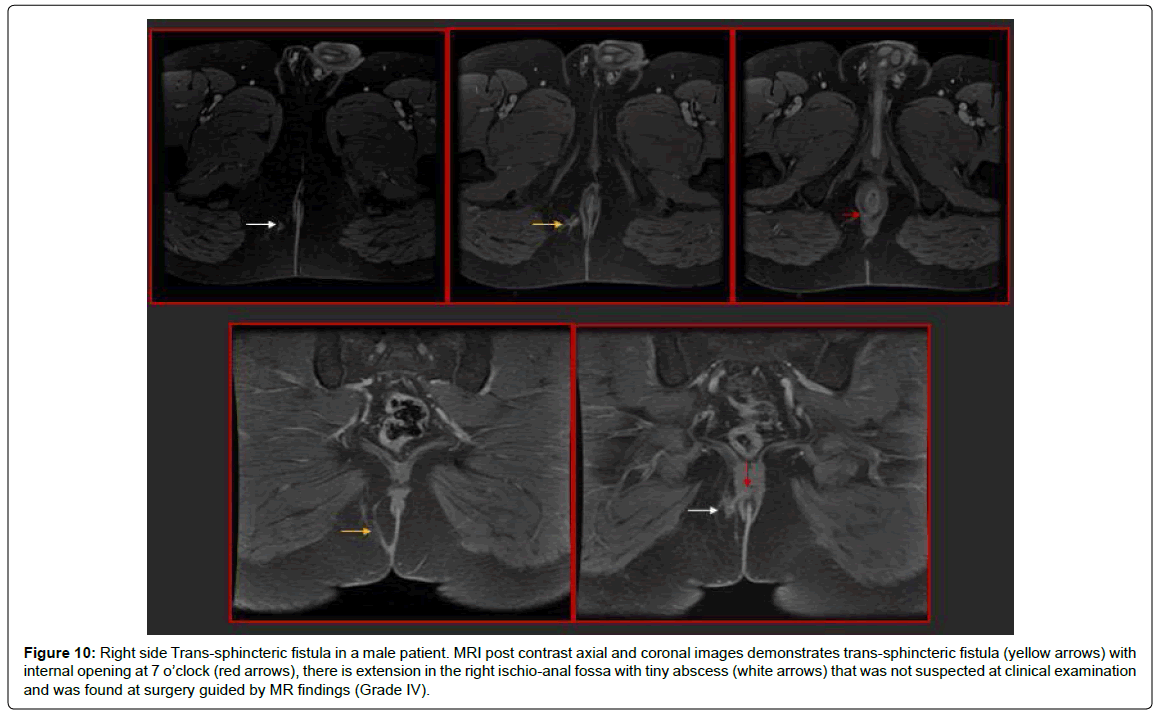
Figure 10: Right side Trans-sphincteric fistula in a male patient. MRI post contrast axial and coronal images demonstrates trans-sphincteric fistula (yellow arrows) with internal opening at 7 o’clock (red arrows), there is extension in the right ischio-anal fossa with tiny abscess (white arrows) that was not suspected at clinical examination and was found at surgery guided by MR findings (Grade IV).
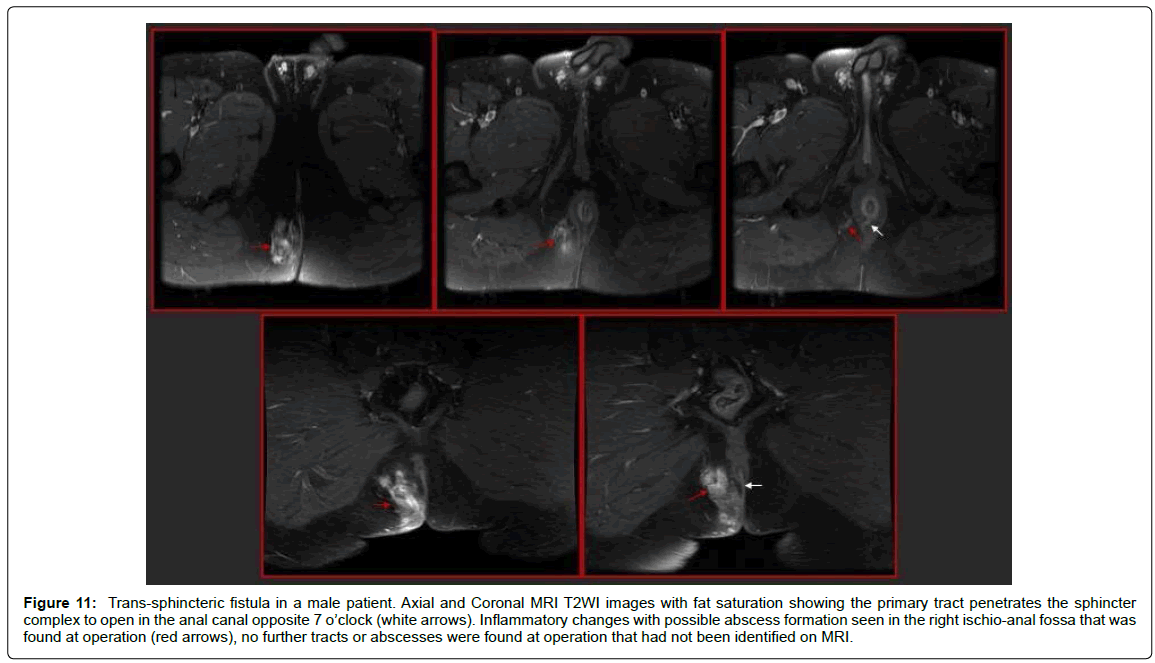
Figure 11: Trans-sphincteric fistula in a male patient. Axial and Coronal MRI T2WI images with fat saturation showing the primary tract penetrates the sphincter complex to open in the anal canal opposite 7 o’clock (white arrows). Inflammatory changes with possible abscess formation seen in the right ischio-anal fossa that was found at operation (red arrows), no further tracts or abscesses were found at operation that had not been identified on MRI.
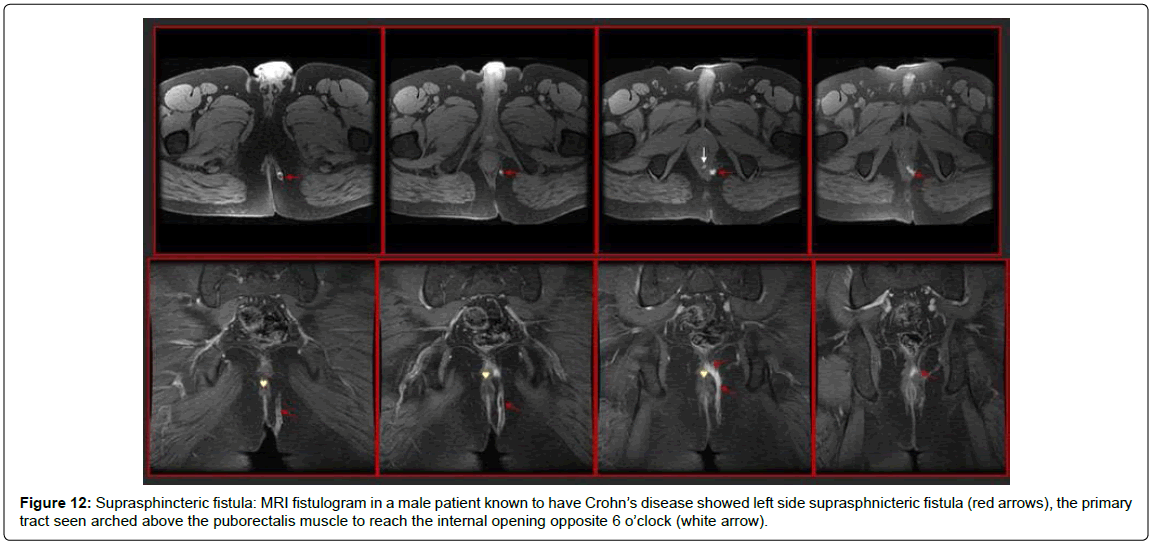
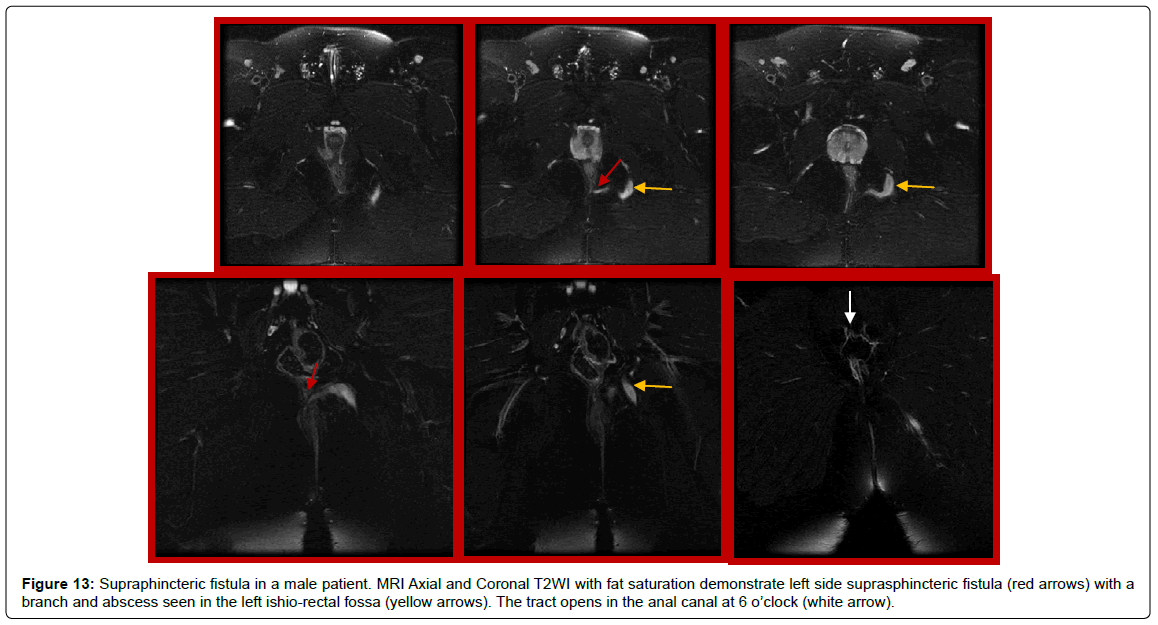
Figure 13: Supraphincteric fistula in a male patient. MRI Axial and Coronal T2WI with fat saturation demonstrate left side suprasphincteric fistula (red arrows) with a branch and abscess seen in the left ishio-rectal fossa (yellow arrows). The tract opens in the anal canal at 6 o’clock (white arrow).
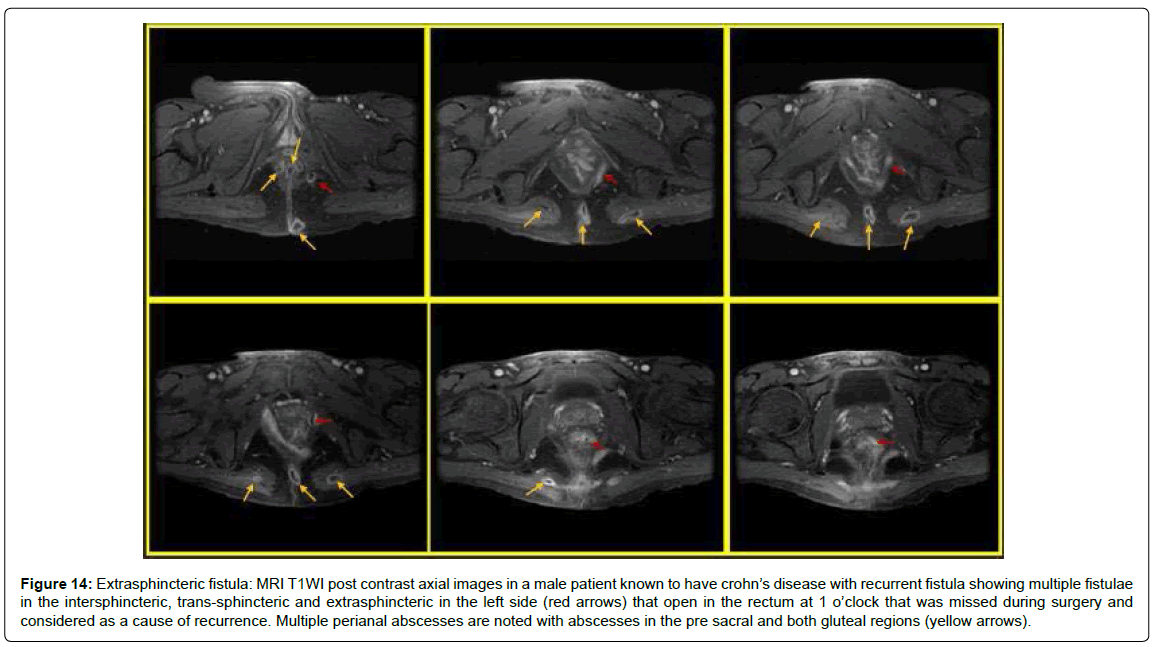
Figure 14: Extrasphincteric fistula: MRI T1WI post contrast axial images in a male patient known to have crohn’s disease with recurrent fistula showing multiple fistulae in the intersphincteric, trans-sphincteric and extrasphincteric in the left side (red arrows) that open in the rectum at 1 o’clock that was missed during surgery and considered as a cause of recurrence. Multiple perianal abscesses are noted with abscesses in the pre sacral and both gluteal regions (yellow arrows).
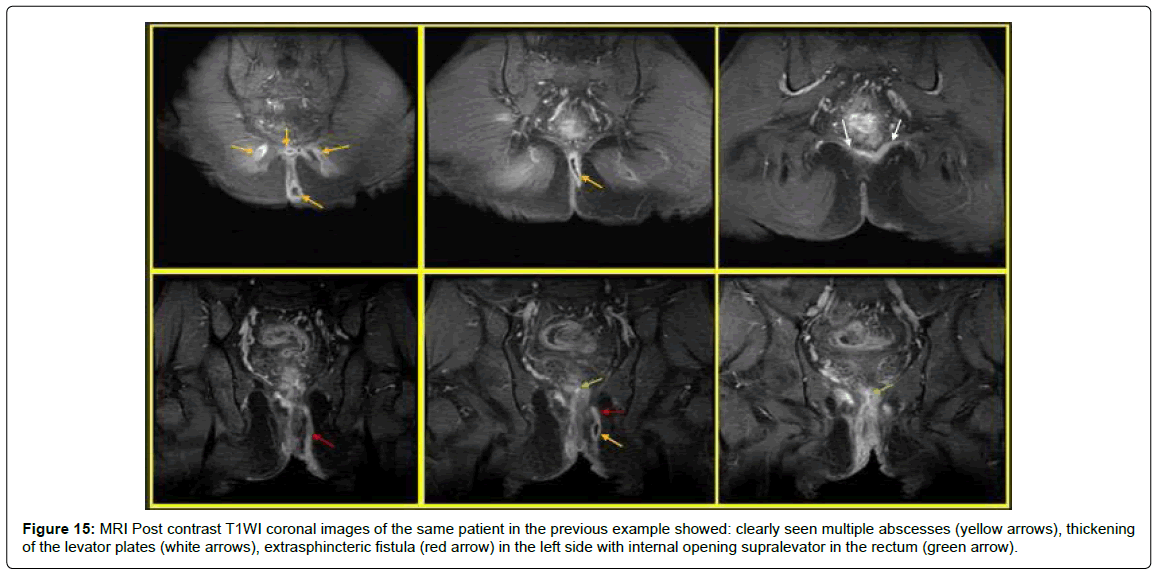
Figure 15: MRI Post contrast T1WI coronal images of the same patient in the previous example showed: clearly seen multiple abscesses (yellow arrows), thickening of the levator plates (white arrows), extrasphincteric fistula (red arrow) in the left side with internal opening supralevator in the rectum (green arrow).
The correlation of the MR imaging findings, with surgical findings demonstrated that MR imaging correctly depict primary tracks in 52 of 56 cases. MR imaging showed two false-positive findings of a primary track. Two intersphincteric fistulas were misdiagnosed at MRI, one proved to be a sinus (Figure 16) at surgery and the other one, no fistula seen. Two trans-sphincteric fistulas were missed at MRI and were seen at surgery. MR imaging depicted 20 of 20 abscess collections, five are supralevator abscess collections and 15 are intersphincteric. All 5 horseshoe fistulas detected by using MR imaging were confirmed at surgery, all secondary branches were seen properly in MRI.
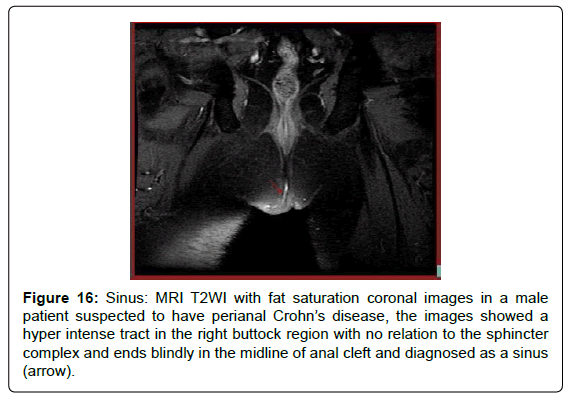
Figure 16: Sinus: MRI T2WI with fat saturation coronal images in a male patient suspected to have perianal Crohn’s disease, the images showed a hyper intense tract in the right buttock region with no relation to the sphincter complex and ends blindly in the midline of anal cleft and diagnosed as a sinus (arrow).
From the Table 2 we can see that MRI has a very high accuracy rate with high sensitivity and specificity in detection of primary tract and internal opening describing about 96% and 100% in detection of the abscess and horseshoe fistula.
| MR Imaging Finding | Sensitivity | Specificity | PPV | NPV |
|---|---|---|---|---|
| Primary track | 96(52/54) | 96(54/56) | 96(52/54) | 96(54/56) |
| Abscess | 100(20/20) | 100(20/20) | 100(20/20) | 100(20/20) |
| Horseshoe fistula | 100(5/5) | 100(5/5) | 100(5/5) | 100(5/5) |
| Internal opening | 96(52/54) | 96(54/56) | 96(52/54) | 96(54/56) |
Table 2: Accuracy of MR Imaging for the detection of a Primary track, Abscess, Horseshoe fistula and Internal opening.
Discussion
The study is a retrospective comparative between surgery and preoperative MR imaging, aiming to precisely evaluate the value of preoperative MRI examination.
Preoperative MR imaging revealed internal opening in 52 patients (96%). Two cases were misdiagnosed as a intersphincteric fistula and one proved to be a sinus and no fistula seen in the second case, it represents a prominent vessel. Another two trans-sphincteric fistula were missed in MRI and depicted at surgery, the only explanation that these cases missed is bad quality of the images as both patients are obese.
MRI can obviously detect fistulous extension with good localization in relation to surgically constant anatomical landmarks, especially in coronal sequences. The most important anatomical points for the surgeon were internal opening location which was described in clock orientation-external sphincter, course of the tract in relation to the sphincters and levatorani muscle. Preoperative characterization of the anatomical course of the fistula and all associated infection is crucial [9] and MRI helps to limit the recurrence and/or incontinence after surgery. This can be explained as it helps to avoid unnecessary wide exploratory dissection through the sphincter. While direct non imaging guided dissection may enforce the surgeon to do generous transsphincteric or supralevator dissection for detailed exploration, which is incriminated for postoperative incontinence. Also, the high possibility of missing other pathological contents, like the commonly reported side branches, horseshoe extension and abscesses, is the main causative etiology of postoperative recurrence [5].
This is also supported by many authors [2,10,11] who stated that MR imaging could depict more extensions and/or associated findings than could be gained from direct surgical exploration without preoperative imaging. So, additional information that can be obtained from preoperative MR imaging will improve the surgical results, especially in patients with complex high grade fistulas [5].
Fistulas associated with Crohn disease and recurrent fistulas, the complex fistulas, are often more difficult to evaluate during surgery because of the extensive secondary extensions and multiple perianal and perirectal abscess collections. When not identified and properly treated, these extensions and abscesses may lead to recurrences [12]. The recurrent nature of Crohn perianal disease is well known, [13] and many recurrences in patients with Crohn fistulas are simply due to overlooked abscesses and secondary extensions [14]. The largest additional value was seen in the detection of supralevator abscesses and horseshoe fistulas (Figure 4 and 5) [15].
MRI appearance
Active fistula tract appears as a hypo intense linear structure on T1-weighted imaging and hyper intense on T2-weighted imaging (best visualized with fat saturation) relative to muscle and enhances with IV contrast agent. Granulation tissue with increased vascularity is thought to account for the T2-weighted imaging hyper intensity and contrast enhancement [2]. Inactive tracts are also hypo intense on T1- weighted imaging but lack the associated T2-weighted imaging hyper intensity and contrast enhancement. Tissues surrounding the tract may also show hyper intensity on T2-weighted imaging if there is edema or inflammation. MRI can be used to assess treatment response, as suggested by Savoye-Collet et al. [16]. Loss of T2-weighted imaging hyper intense signal precedes lack of enhancement and proposed a predictable stepwise response to therapy. These stepwise changes correlated well with clinical response, suggesting that MRI follow-up may be used to guide therapy. Additionally, deep-tissue healing, as visualized by MRI, typically takes longer than the superficial healing that is apparent by clinical examination, thus offering better assessment of residual or incompletely treated disease [17]. In patients with prior surgery, it is important to note the associated findings, such as fat packing (hyper intense on T1-weighted imaging), surgical drains, particularly setons (linear low signal on T1- and T2-weighted imaging) and gas foci (focal low signal intensity on T1- and T2-weighted imaging) [3].
Conclusions
MRI is very useful in successful treatment of perianal fistula because it is like a roadmap to demonstrate accurately the anatomy of the perianal region, show the anal sphincter complex and clearly identify the relationship of fistulas to the pelvic diaphragm and ischio-rectal fossa which has important implications for surgical management and outcome. Preoperative MRI can help to recognize the unidentified infection, accurate in the detection of the secondary extension and abscess formation and markedly diminish the chance of recurrence.
High-spatial-resolution MR imaging is therefore recommended in the preoperative work-up of patients with Crohn and recurrent anal fistulas.
References
- de Miguel Criado J, del Salto LG, Rivas PF, del Hoyo LF, Velasco LG, et al. (2012) MR imaging evaluation of perianal fistulas: spectrum of imaging features. Radiographics 32: 175-194.
- Halligan S, Stoker J (2006) Imaging of fistula in ano. Radiology 239: 18-33.
- O'Malley RB, Al-Hawary MM, Kaza RK, Wasnik AP, Liu PS, et al. (2012) Rectal imaging: part 2, Perianal fistula evaluation on pelvic MRI--what the radiologist needs to know. AJR Am J Roentgenol 199: W43-53.
- Khera PS, Badawi HA, Afifi AH (2010) MRI in perianal fistulae. Indian J Radiol Imaging 20: 53-57.
- Mahmoud E, Eid M, Mansy H, Matarawy K, Wally M (2013) Preoperative MRI of perianal fistula: Is it really indispensable? Can it be deceptive?. Alexandria Journal of Medicine 49: 133-144.
- Kuster GG (1965) Relationship of anal glands to lymphatics. Dis Colon Rectum 8: 329-332.
- Parks AG, Gordon PH, Hardcastle JD (1976) A classification of fistula-in-ano. Br J Surg 63: 1-12.
- Morris J, Spencer JA, Ambrose NS (2000) MR imaging classification of perianal fistulas and its implications for patient management. Radiographics 20: 623-635.
- Halligan S, Taylor SA (2010) MRI of the anus. Med Radiol 2: 329–346.
- Jones J, Tremaine W (2005) Evaluation of perianal fistulas in patients with Crohn's disease. MedGenMed 7: 16.
- Buchanan G, Halligan S, Williams A, Cohen CR, Tarroni D, et al. (2002) Effect of MRI on clinical outcome of recurrent fistula-in-ano. Lancet 360: 1661-1662.
- Seow-Choen, Phillips RK (1991) Insights gained from the management of problematical anal fistulae at St. Mark's Hospital, 1984-88. Br J Surg 78: 539-541.
- Keighley MR, Allan RN (1986) Current status and influence of operation on perianal Crohn's disease. Int J Colorectal Dis 1: 104-107.
- Gruwez JA, Christiaens MR (1991) [Anal-perineal lesions in Crohn's disease]. Ann Gastroenterol Hepatol (Paris) 27: 251-255.
- Beets-Tan RG, Beets GL, van der Hoop AG, Kessels AG, Vliegen RF, et al. (2001) Preoperative MR imaging of anal fistulas: Does it really help the surgeon? Radiology 218: 75-84.
- Savoye-Collet C, Savoye G, Koning E, Dacher JN, Lerebours E (2011) Fistulizing perianal Crohn's disease: contrast-enhanced magnetic resonance imaging assessment at 1 year on maintenance anti-TNF-alpha therapy. Inflamm Bowel Dis 17: 1751-1758.
- Ng SC, Plamondon S, Gupta A, Burling D, Kamm MA (2009) Prospective assessment of the effect on quality of life of anti-tumour necrosis factor therapy for perineal Crohn's fistulas. Aliment Pharmacol Ther 30: 757-766.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 21680
- [From(publication date):
October-2013 - Nov 23, 2025] - Breakdown by view type
- HTML page views : 16526
- PDF downloads : 5154