Review Article Open Access
MRI of Crohn's Anoperineal Disease
| Shaunagh McDermott*, Anuradha S Shenoy-Bhangle and Michael S Gee | |
| Massachusetts General Hospital, Division of Abdominal Imaging and Interventional Radiology, 55 Fruit Street, Boston, MA 02114, USA | |
| Corresponding Author : | Shaunagh McDermott Massachusetts General Hospital Division of Abdominal Imaging and Interventional Radiology 55 Fruit Street, Boston, MA 02114, USA Tel: (857) 205-8501 Fax: (617) 726-4891 E-mail: smcdermott1@partners.org |
| Received February 01, 2012; Accepted April 02, 2012; Published April 04, 2012 | |
| Citation: McDermott S, Shenoy-Bhangle AS, Gee MS (2012) MRI of Crohn’s Anoperineal Disease. J Gastroint Dig Syst S9:001. doi: 10.4172/2161-069X.S9-001 | |
| Copyright: © 2012 McDermott S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. | |
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Anoperineal involvement is seen in up to 38% of patients with Crohn’s disease. To adopt the best treatment strategy and avoid recurrences, it is important to obtain precise radiological information of the fistulous track. Magnetic resonance imaging provides precise information on the anatomy of the anal canal, the anal sphincteric complex, and the relationship of the fistula to the pelvic floor structures. In this article we review the anatomy of the anal canal, the pathogenesis of perianal fistula, different MRI sequences for the evaluation of fistula with emphasis on their imaging findings and an MR imaging based grading system for their classification.
| Keywords |
| Crohn’s disease; Perianal fistula; Magnetic resonance imaging |
| Introduction |
| The most current population-based series [from Sweden and Minnesota] have found that anoperineal involvement is seen in 14% to 38% of patients with Crohn’s disease [1-3], with isolated perianal disease seen in only 5% [4]. The prevalence of perianal manifestations increases as the disease progresses distally, with only 15% of patients with ileocolic Crohn’s disease developing fistula but fistulae occurring in 92% of patients with Crohn’s disease involving the colon and rectum [3]. There does not appear to be a predilection for age, with between 13% and 62% of children and adolescents with Crohn’s disease experiencing perianal manifestations [2,5,6]; however, a younger age of onset increases the odds of developing perianal disease over time [7,8]. |
| Perianal Crohn’s disease is often recurrent, with 35% to 59% of patients relapsing within 2 years [9]. More than 80% of patients require surgery, and as many as 31% require a permanent stoma [1,3,10]. The presence of perianal disease is associated with a more disabling natural history [11], with increased extraintestinal manifestations [12] and greater steroid resistance [13]. Large studies have also shown an increased risk for squamous cell carcinoma and adenocarcinoma of the anus in patients with anorectal involvement [14,15]. |
| The pathogenesis of fistulae formation in Crohn’s disease is still unknown but is thought to be the end result of severe transmural bowel inflammation. One histological hallmark of fistulae caused by Crohn’s disease is dense infiltration of B lymphocytes [16]. One recent study found that Crohn’s disease fistulae were lined by mesenchymal transitional cells rather than epithelium, implicating a role for Epithelial- Mesenchymal Transition (EMT) in the cellular invasion underlying fistula formation [17]. This suggests a potential role for inhibition of the genetic pathways underlying EMT as a therapeutic strategy. |
| Over the last decade, magnetic resonance imaging (MRI) has become the reference technique in the evaluation of perianal Crohn’s disease, showing high diagnostic ability, with sensitivity and specificity, respectively, of 100% and 86% in detection of fistula’s primary tracts, and of 96% and 97% in detection of abscesses [18,19]. Moreover, surgical management may be altered in up to 40% of patients by the addition of MRI to examination under anesthesia (EUA) [19,20]. In particular, MRI has been shown to be of value to study complex perianal fistulae, being able to detect sepsis outside the anal sphincter and their relationship with the sphincter and levator ani muscle [21]. Besides an accurate morphological evaluation of the perianal space, MRI has been demonstrated to provide information about perianal disease activity, assessing inflammatory changes in fistula tracks in response to treatment with infliximab [22-26]. |
| Treatment Options for the Management of Perianal Crohn’s Disease |
| The main objective of perianal fistula treatment is to effectively close the internal opening of the fistula track while preserving sphincter integrity and continence. Treatment typically involves either or a combination of medical treatment and surgical techniques. A recently published study using evidence based practice techniques made the following recommendations for the treatment of perianal fistulae in Crohn’s disease [27]: |
| • Fistulotomy is considered for simple perianal fistula |
| • In complex perianal fistulas, antibiotics and azathioprine or 6-mercaptopurine, which are often combined with a loose seton, constitute the first-line medical treatment |
| • In cases with persistent secretions, inflimab at 5 mg/kg is given at weeks 0, 2 and 6 and subsequently every 8 weeks |
| • Adalimumab may improve fistula response in both infliximabnative patients and following infliximab treatment failure |
| • Local therapy with fibrin glue or fistula plugs is rarely effective |
| • Definitive surgical closure of perianal fistulas using an advancement flap may be attempted, but this procedure is associated with a high risk of relapse |
| • Colostomy and surgical proctectomy are the ultimate surgical treatment options for fistula |
| Anatomy of the Anal Region |
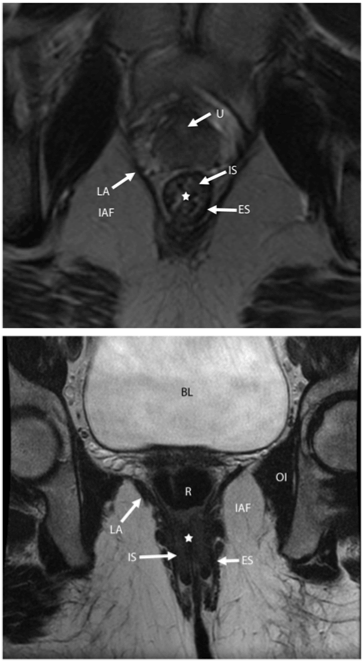
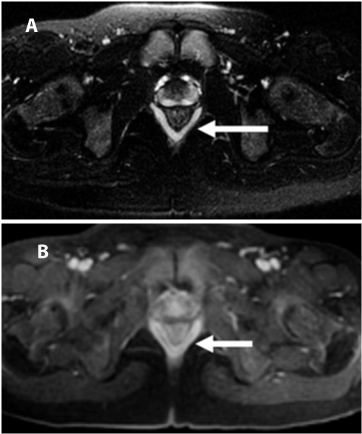
| The internal sphincter is involuntary and is composed of smooth muscle continuous with the circular smooth muscle of the rectum. It is responsible for 85% of resting tone. In most individuals, it can be divided without causing a loss of continence. The internal sphincter appears mildly hyperintense on both T1- and T2-weighted images. The external sphincter is composed of striated muscle and is continuous superiorly with the puborectalis and levator ani muscles. It contributes only 15% of resting anal tone, but its strong voluntary contractions resist defecation. A division of the external sphincter can lead to incontinence. The external sphincter appears hypointense against the background of the ischiorectal fossa fat on non-fat-suppressed T2- weighted images and appears similarly hypointense on T1-weighted and STIR images, although its conspicuity with respect to the adjacent fat is somewhat diminished with fat-suppression techniques (Figure 1). |
| Evaluation of Perianal Fistula, Before and After Treatment |
| The objectives in performing and interpreting any imaging study for perianal fistulas are simple [28]: |
| • To determine the relationship of any fistulous track to the sphincter complex. Is the sphincter involved, does the track transverse both layers of the sphincter (transsphincteric) or only the internal sphincter (intersphincteric)? Surgeons also want to know distance of the fistula from the levator ani complex in order to guide as to whether fistulotomy vs. seton placement is warranted. Setons being placed in complex fistulae or high fistulae to avoid incontinence associated with open fistulotomy. |
| • To identify any secondary fistulous tracks and the sites of any abscess cavities. The presence of abscess mandates antibiotic therapy and/or surgical drainage, and precludes use of many immunosuppressive Crohn’s medications. |
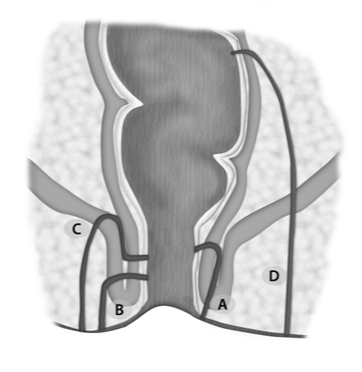
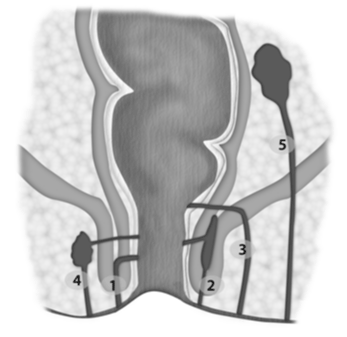
| At present, two classifications are most commonly used: a classification proposed by Parks in 1976, which was created for surgical use [29], and the St James University Hospital classification, which was developed based on a MRI examination [28,30] (Tables 1 and 2) (Figure 2 and 3). |
| A study has shown that MR imaging is better than initial surgical exploration in the prediction of patient outcome [30]. When the St James’s University Hospital classification was used, the MR imaging grading of fistulas was significantly associated with outcome: MR imaging grades 1 and 2 were associated with a satisfactory outcome (i.e. no further surgery needed), whereas grade 3-5 were associated with unsatisfactory outcome (i.e. further surgery needed). |
| More recently, MR imaging has been used to monitor the response of fistulous tracks to medical treatments. MRI demonstrates that skin healing may not be an appropriate determinant of outcome in patients with fistulous disease. Van Asche et al. [22] reported that despite healing on clinical examination, fistulous tracks persist in patients with Crohn’s disease treated with infliximab therapy. Eleven of 18 patients demonstrated clinical response evidenced by the absence of any further drainage from the fistula but 8 patients had evidence of persistent tracks and residual inflammation on MRI, raising the clinical concern about the durability of the long-term response [22]. |
| MR Imaging Techniques |
| MR imaging uses magnetic fields and radiofrequency energy to create images of soft tissues without the use of ionizing radiation. This is an important consideration, especially in young patients who, over the course of treatment and surveillance, will require multiple imaging sessions. An additional advantage is that no patient preparation is required if a body coil is used. MR imaging in the coronal and axial planes demonstrates fistulous tracks in relation to the sphincter complex, ischiorectal fossa, and levator plate. Imaging in the sagittal and oblique planes is helpful in selected cases (e.g. anovaginal or presacral disease). |
| Currently, MR examinations may be performed with either endoluminal anal or external phased array coils, or a combination of the two, with use of the phased array coil being the most common approach [31]. Endoluminal anal coils offer higher spatial resolution than body coils [32], and their use resulted in an initial increased accuracy of classification of the primary track to up to 93% [33,34]. However, examinations performed using endoanal coils alone are limited by field of view and by patient discomfort, a result of which is often suboptimal image quality due to patient motion [35]. External phased array coils represent an excellent alternative in providing high spatial resolution images with a large field of view, while avoiding the discomfort of an endoluminal coil. The larger fields of view are important for better delineation of secondary tracks and extensions [36]. |
| Intramuscular glucagon for reduction of bowel motion is not necessary for most patients, as the majority of bowel motion occurs in the upper pelvis, beyond the region of interest at the level of the anal canal. |
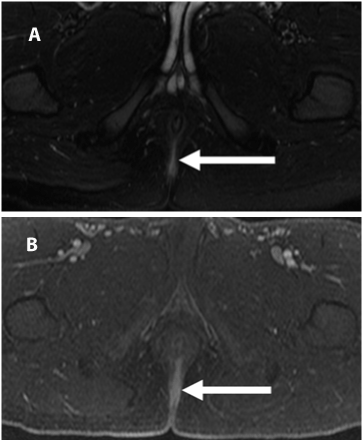
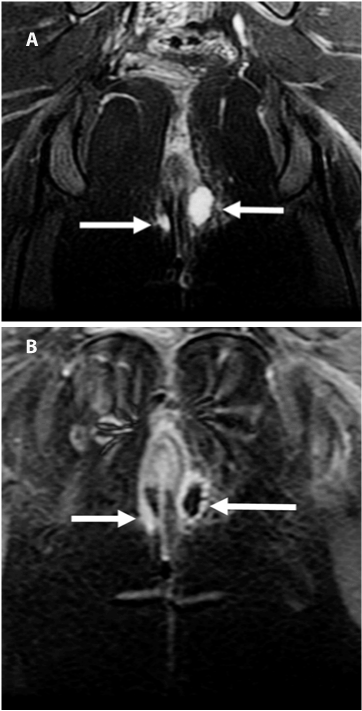
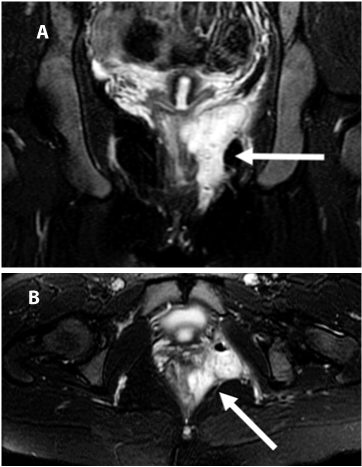
| Many different MR imaging techniques have been described for perianal fistula assessment (Figures 4-8). |
| Unenhanced T1-weighted images |
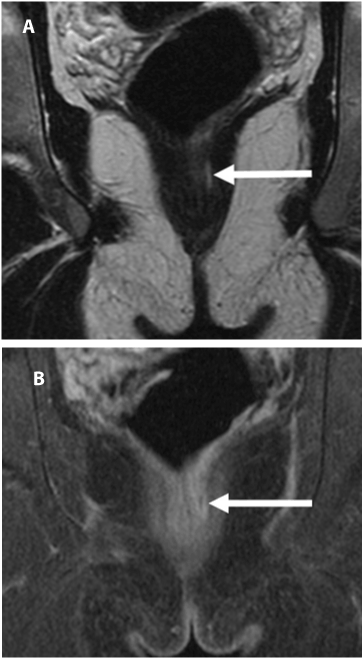
| These provide an excellent anatomical overview of the sphincter complex, levator plate, and the ischiorectal fossae. Fistulous tracks, inflammation, and abscesses, however, appear as areas of low to intermediate signal intensity and may not be distinguished from normal structures. |
| These images may be helpful in postoperative assessment. In the immediate postoperative period, hemorrhage will appear hyperintense and may thus be differentiated from a residual track. Also, fat containing ‘grafts’ may be placed to fill cavities and resection voids in restorative surgery. These hyperintense structures are readily identified on unenhanced T1-weighted sequences and can be distinguished from active disease, which appears hyperintense only after enhancement with gadolinium [28]. |
| T2-weighted and short tau inversion recovery (STIR) images |
| On these images, pathological processes including fistulas, secondary fistulous tracks, and fluid collections are clearly depicted. They appear as areas of high signal intensity in contrast with the lower signal intensity of the sphincters, muscles and fat. |
| T1-weighted dynamic contrast enhanced images (DCE-MRI) |
| With use of this technique, active fistulous tracks, secondary ramifications, and abscesses are clearly demonstrated. The tracks brilliantly enhance, as do the walls of the abscess cavities. Retained pus remains unenhanced, with resulting ring enhancement. |
| Studies have shown that DCE-MRI can help determine disease activity in perianal Crohn’s disease and might be helpful in selecting a subpopulation of patients who should be monitored more closely for development of more extensive disease [37,38]. |
| Contrast enhanced images may not be essential for all cases. They are very useful in previously operated cases, especially when there is concern about residual disease activity or abscess formation within postoperative and healing inflammatory tissue [28]. |
| Diffusion-weighted imaging (DWI) |
| Recently the use of DWI has been examined in evaluating perianal fistula [39,40]. On these images the fistulous tracks appear hyperintense, whereas the background signal is significantly suppressed. It may be a helpful adjunct to T2-weighted imaging, especially in patients with risk factors for contrast agents [39]. It was also found to be a feasible method for evaluating perianal fistula activity, as the apparent diffusion coefficient [ADC] was significantly lower in the positive inflammation activity group compared to the negative inflammation activity group [40]. |
| Studies have shown that for experienced readers, combining fatsaturated contrast enhanced T1-weighted gradient echo images with either T2-weighted or STIR images collected in both the coronal and axial planes was sufficient to make an assessment before deciding the surgical extent of the procedure [41]. |
| Limitations of MRI |
| The use of MRI requires a radiologist with additional experience in pelvic anatomy. The inter- and intra-observer agreement among expert radiologists is excellent, but outside the field, the quality of information is substandard [42]. In managing patients with fistulous tracks, the internal opening and its relationship with the dentate line influences whether one will perform a primary fistulotomy or a staged procedure. However, a limitation of MRI is that it is unable to identify the dentate line. In addition, some patients have relative or absolute contraindications to MRI including those with specific implanted devices such as pacemakers and certain cerebral aneurysm clips. Imaging will also be limited in patients who cannot receive intravenous contrast due to renal insufficiency. |
| Comparison with other Imaging Modalities |
| Fistulography was the traditional technique used to define fistula anatomy. However its reliability was unpredictable and very often it failed to define the extent of primary tracks or secondary extensions. In addition, it gave no information on the relationship of tracks to the sphincter complex or pelvic floor. Its limited value was confirmed by Kuijpers et al. [43], who reported that it was accurate in only 16% of cases. In addition, false-positive results occurred in 10% causing surgeons to search for nonexisting tracks, increasing the potential for iatrogenic injury. |
| Differentiating fistulae and resolving structures of the pelvic floor using computed tomography (CT) can be difficult [44], primarily because of the poor soft contrast of CT. One study found that CT scanning had a sensitivity of only 24% in the diagnosis of fistula-in-ano [45]. |
| Endoanal Ultrasound (EAUS) has a high level of accuracy in the characterization of perinanal fistula, with diagnostic accuracy ranging from 63-94% for classification of the primary track and with accuracy of prediction of site of the internal opening as high as 93% [46]. Several studies have found EAUS to be inferior to pelvic MRI in the diagnosis of perianal disease [47-49]. With the use of threedimensional reconstruction of two-dimensional images, however, some investigators have shown that the results of EAUS are comparable with MRI, with excellent patient tolerance [34,50]. The use of hydrogen peroxide injection in the fistula track during EAUS substantially improves accuracy [51]. The diagnostic accuracy of EAUS can also be improved to values comparable with those of MRI by using a computerassisted evaluation of the ultrasound images [52]. |
| The question of which modality is best for discriminating simple and complex fistulas was specifically addressed using established evidence based practice principals by Sahni et al. [21]. They found that MRI is the superior technique with sensitivity and specificity of 97% and 96% compared with 92% and 85% for anal ultrasound and 75% and 64% for clinical examination. |
| Conclusion |
| MRI has an important role in the management of Crohn’s Perianal disease. Preoperative MRI can dictate the surgical procedure and is an important determinate of outcome. It also plays a critical role in assessing the response of fistulous tracks to the milieu of new medical therapies. |
| References |
References
- Hellers G, Bergstrand O, Ewerth S, Holmström B (1980) Occurrence and outcome after primary treatment of anal fistulae in Crohn's disease. Gut 21: 525-527.
- Sandborn WJ, Fazio VW, Feagan BG, Hanauer SB, American Gastroenterological Association Clinical Practice Committee (2003) AGA technical review on perianal Crohn's disease. Gastroenterology 125: 1508-1530.
- Schwartz DA, Loftus EV, Tremaine WJ, Panaccione R, Harmsen WS, et al. (2002) The natural history of fistulizing Crohn's disease in Olmsted County, Minnesota. Gastroenterology 122: 875-880.
- Lockhart-Mummery HE (1975) Symposium. Crohn's disease: anal lesions. Dis Colon Rectum 18: 200-202.
- Palder SB, Shandling B, Bilik R, Griffiths AM, Sherman P (1991) Perianal complications of pediatric Crohn's disease. J Pediatr Surg 26: 513-515.
- Markowitz J, Daum F, Aiges H, Kahn E, Silverberg M, et al. (1984) Perianal disease in children and adolescents with Crohn's disease. Gastroenterology 86: 829-833.
- Cosnes J, Cattan S, Blain A, Beaugerie L, Carbonnel F, et al. (2002) Long-term evolution of disease behavior of Crohn's disease. Inflamm Bowel Dis 8: 244-250.
- Roberts PL, Schoetz DJ, Pricolo R, Veidenheimer MC (1990) Clinical course of Crohn's disease in older patients. A retrospective study. Dis Colon Rectum 33: 458-462.
- Makowiec F, Jehle EC, Starlinger M (1995) Clinical course of perianal fistulas in Crohn's disease. Gut 37: 696-701.
- Mueller MH, Geis M, Glatzle J, Kasparek M, Meile T, et al. (2007) Risk of fecal diversion in complicated perianal Crohn's disease. J Gastrointest Surg 11: 529-537.
- Beaugerie L, Seksik P, Nion-Larmurier I, Gendre JP, Cosnes J (2006) Predictors of Crohn's disease. Gastroenterology 130: 650-656.
- Rankin GB, Watts HD, Melnyk CS, Kelley ML. (1979) National Cooperative Crohn's Disease Study: extraintestinal manifestations and perianal complications. Gastroenterology 77: 914-920.
- Gelbmann CM, Rogler G, Gross V, Gierend M, Bregenzer N, et al. (2002) Prior bowel resections, perianal disease, and a high initial Crohn's disease activity index are associated with corticosteroid resistance in active Crohn's disease. Am J Gastroenterol 97: 1438-1445.
- Sjödahl RI, Myrelid P, Söderholm JD (2003) Anal and rectal cancer in Crohn's disease. Colorectal Dis 5: 490-495.
- Ky A, Sohn N, Weinstein MA, Korelitz BI (1998) Carcinoma arising in anorectal fistulas of Crohn's disease. Dis Colon Rectum 41: 992-996.
- Bataille F, Klebl F, Rümmele P, Schroeder J, Farkas S, et al. (2004) Morphological characterisation of Crohn's disease fistulae. Gut 53: 1314-1321.
- Bataille F, Rohrmeier C, Bates R, Weber A, Rieder F, et al. (2008) Evidence for a role of epithelial mesenchymal transition during pathogenesis of fistulae in Crohn's disease. Inflamm Bowel Dis 14: 1514-1527.
- Caprilli R, Gassull MA, Escher JC, Moser G, Munkholm P, et al. (2006) European evidence based consensus on the diagnosis and management of Crohn's disease: special situations. Gut 55: i36-i58.
- Beets-Tan RG, Beets GL, van der Hoop AG, Kessels AG, Vliegen RF, et al. (2001) Preoperative MR imaging of anal fistulas: Does it really help the surgeon? Radiology 218: 75-84.
- Buchanan GN, Halligan S, Williams AB, Cohen CR, Tarroni D, et al. (2003) Magnetic resonance imaging for primary fistula in ano. Br J Surg 90: 877-881.
- Sahni VA, Ahmad R, Burling D (2008) Which method is best for imaging of perianal fistula? Abdom Imaging 33: 26-30.
- Van Assche G, Vanbeckevoort D, Bielen D, Coremans G, Aerden I, et al. (2003) Magnetic resonance imaging of the effects of infliximab on perianal fistulizing Crohn's disease. Am J Gastroenterol 98: 332-339.
- Bell SJ, Halligan S, Windsor AC, Williams AB, Wiesel P, et al. (2003) Response of fistulating Crohn's disease to infliximab treatment assessed by magnetic resonance imaging. Aliment Pharmacol Ther 17: 387-393.
- Savoye-Collet C, Savoye G, Koning E, Dacher JN, Lerebours E (2011) Fistulizing perianal Crohn's disease: contrast-enhanced magnetic resonance imaging assessment at 1 year on maintenance anti-TNF-alpha therapy. Inflamm Bowel Dis 17: 1751-1758.
- Ng SC, Plamondon S, Gupta A, Burling D, Swatton A, et al. (2009) Prospective evaluation of anti-tumor necrosis factor therapy guided by magnetic resonance imaging for Crohn's perineal fistulas. Am J Gastroenterol 104: 2973-2986.
- Horsthuis K, Ziech ML, Bipat S, Spijkerboer AM, de Bruine-Dobben AC, et al. (2011) Evaluation of an MRI-based score of disease activity in perianal fistulizing Crohn's disease. Clin Imaging 35: 360-365.
- Hvas CL, Dahlerup JF, Jacobsen BA, Ljungmann K, Qvist N, et al. (2011) Diagnosis and treatment of fistulising Crohn's disease. Dan Med Bull 58: C4338.
- Morris J, Spencer JA, Ambrose NS (2000) MR imaging classification of perianal fistulas and its implications for patient management. Radiographics 20: 623-635.
- Parks AG, Gordon PH, Hardcastle JD (1976) A classification of fistula-in-ano. Br J Surg 63: 1-12.
- Spencer JA, Chapple K, Wilson D, Ward J, Windsor AC, et al. (1998) Outcome after surgery for perianal fistula: predictive value of MR imaging. AJR Am J Roentgenol 171: 403-406.
- Halligan S, Stoker J (2006) Imaging of fistula in ano. Radiology 239: 18-33.
- Stoker J, Hussain SM, van Kempen D, Elevelt AJ, Laméris JS (1996) Endoanal coil in MR imaging of anal fistulas. AJR Am J Roentgenol 166: 360-362.
- Hussain SM, Stoker J, Schouten WR, Hop WC, Laméris JS (1996) Fistula in ano: endoanal sonography versus endoanal MR imaging in classification. Radiology 200: 475-481.
- West RL, Dwarkasing S, Felt-Bersma RJ, Schouten WR, Hop WC, et al. (2004) Hydrogen peroxide-enhanced three-dimensional endoanal ultrasonography and endoanal magnetic resonance imaging in evaluating perianal fistulas: agreement and patient preference. Eur J Gastroenterol Hepatol 16: 1319-1324.
- Halligan S, Bartram CI (1998) MR imaging of fistula in ano: are endoanal coils the gold standard? AJR Am J Roentgenol 171: 407-412.
- deSouza NM, Gilderdale DJ, Coutts GA, Puni R, Steiner RE (1998) MRI of fistula-in-ano: a comparison of endoanal coil with external phased array coil techniques. J Comput Assist Tomogr 22: 357-363.
- Horsthuis K, Lavini C, Bipat S, Stokkers PC, Stoker J (2009) Perianal Crohn disease: evaluation of dynamic contrast-enhanced MR imaging as an indicator of disease activity. Radiology 251: 380-387.
- Villa C, Pompili G, Franceschelli G, Munari A, Radaelli G, et al. (2012) Role of magnetic resonance imaging in evaluation of the activity of perianal Crohn's disease. Eur J Radiol 81: 616-622.
- Hori M, Oto A, Orrin S, Suzuki K, Baron RL (2009) Diffusion-weighted MRI: a new tool for the diagnosis of fistula in ano. J Magn Reson Imaging 30: 1021-1026.
- Yoshizako T, Wada A, Takahara T, Kwee TC, Nakamura M, et al. (2011) Diffusion-weighted MRI for evaluating perianal fistula activity: Feasibility study. Eur J Radiol.
- Yildirim N, Gökalp G, Öztürk E, Zorluoglu A, Yilmazlar T, et al. (2012) Ideal combination of MRI sequences for perianal fistula classification and the evaluation of additional findings for readers with varying levels of experience. Diagn Interv Radiol 18: 11-19.
- Buchanan GN, Halligan S, Taylor S, Williams A, Cohen R, et al. (2004) MRI of fistula in ano: inter- and intraobserver agreement and effects of directed education. AJR Am J Roentgenol 183: 135-140.
- Kuijpers HC, Schulpen T (1985) Fistulography for fistula-in-ano. Is it useful? Dis Colon Rectum 28: 103-104.
- Yousem DM, Fishman EK, Jones B (1988) Crohn disease: perirectal and perianal findings at CT. Radiology 167: 331-334.
- Schratter-Sehn AU, Lochs H, Vogelsang H, Schurawitzki H, Herold C, et al. (1993) Endoscopic ultrasonography versus computed tomography in the differential diagnosis of perianorectal complications in Crohn's disease. Endoscopy 25: 582-586.
- Sun MR, Smith MP, Kane RA (2008) Current techniques in imaging of fistula in ano: three-dimensional endoanal ultrasound and magnetic resonance imaging. Semin Ultrasound CT MR 29: 454-471.
- Schwartz DA, Wiersema MJ, Dudiak KM, Fletcher JG, Clain JE, et al. (2001) A comparison of endoscopic ultrasound, magnetic resonance imaging, and exam under anesthesia for evaluation of Crohn's perianal fistulas. Gastroenterology 121: 1064-1072.
- Buchanan GN, Halligan S, Bartram CI, Williams AB, Tarroni D, et al. (2004) Clinical examination, endosonography, and MR imaging in preoperative assessment of fistula in ano: comparison with outcome-based reference standard. Radiology 233: 674-681.
- Lunniss PJ, Barker PG, Sultan AH, Armstrong P, Reznek RH, et al. (1994) Magnetic resonance imaging of fistula-in-ano. Dis Colon Rectum 37: 708-718.
- West RL, Zimmerman DD, Dwarkasing S, Hussain SM, Hop WC, et al. (2003) Prospective comparison of hydrogen peroxide-enhanced three-dimensional endoanal ultrasonography and endoanal magnetic resonance imaging of perianal fistulas. Dis Colon Rectum 46: 1407-1415.
- Poen AC, Felt-Bersma RJ, Eijsbouts QA, Cuesta MA, Meuwissen SG (1998) Hydrogen peroxide-enhanced transanal ultrasound in the assessment of fistula-in-ano. Dis Colon Rectum 41: 1147-1152.
- Caprioli F, Losco A, Viganò C, Conte D, Biondetti P, et al. (2006) Computer-assisted evaluation of perianal fistula activity by means of anal ultrasound in patients with Crohn's disease. Am J Gastroenterol 101: 1551-1558.
Tables and Figures at a glance
| Table 1 | Table 2 |
Figures at a glance
 |
 |
 |
 |
| Figure 1 | Figure 2 | Figure 3 | Figure 4 |
 |
 |
 |
 |
| Figure 5 | Figure 6 | Figure 7 | Figure 8 |
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 13754
- [From(publication date):
specialissue-2013 - Nov 23, 2025] - Breakdown by view type
- HTML page views : 9047
- PDF downloads : 4707
