Review Article Open Access
New Endoscopic Diagnosis and Treatment Options for Early Esophageal Cancer
Quan-Lin Li, Yun-Shi Zhong, Wei-Feng Chen, Meng-Jiang He, Ping-Hong Zhou and Li-Qing Yao*
Endoscopy Center and Endoscopy Research Institute, Zhongshan Hospital, Fudan University, 180 Fenglin Road, Shanghai 200032, People’s Republic of China
- *Corresponding Author:
- Li-Qing Yao
Endoscopy Center and Endoscopy Research Institute
Zhongshan Hospital, Fudan University
180 Fenglin Road, Shanghai 200032
People’s Republic of China
E-mail: yao.liqing@zs-hospital.sh.cn
Received date: September 29, 2011; Accepted date: April 05, 2012; Published date: April 07, 2012
Citation: Li QL, Zhong YS, Chen WF, He MJ, Zhou PH, et al. (2012) New Endoscopic Diagnosis and Treatment Options for Early Esophageal Cancer. J Gastroint Dig Syst 2:108. doi: 10.4172/2161-069X.1000108
Copyright: © 2012 Li QL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Recent advances in endoscopic technology have increased detection rates of early esophageal cancer. Novel imaging techniques, such as narrow band imaging, autofluorescence imaging, confocal laser endomicroscopy and optical coherence tomography, have been recently introduced that may be taking us closer to the “optical biopsy” for dignosis of esophageal lesions. Along with the increased endoscopic detection rates, more and more early esophageal lesions have been also treated by endoscopic options. There are now long-term randomized controlled data concerning the effectiveness of ablative approaches (photodynamic therapy, cryotherapy and radiofrequency ablation); however, the ablation does not provide a tissue specimen for histopathological assessment. Unlike ablative techniques, endoscopic resection permits histopathological assessment, similar to surgery. As a new resection technique, endoscopic submucosal dissection results in complete eradication of all diseases and may translate to a lower recurrence rate than conventional endoscopic mucosal resection procedure.
Keywords
Early esophageal cancer; Optical biopsy; Endoscopic therapy; Endoscopic submucosal dissection
Abbreviations
HGIN: High-Grade Intraepithelial Neoplasia; HRE: High-Resolution Endoscopy; NBI: Narrow Band Imaging; EAC: Esophageal Adenocarcinoma; BE: Barrett’s Esophagus; SCC: Squamous Cell Cancer; AFI: Autofluorescence Imaging; CLE: Confocal Laser Endomicroscopy; OCT: Optical Coherence Tomography; EUS: Endoscopic Ultrasound; PDT: Photodynamic Therapy; RFA: Radiofrequency Ablation; ER: Endoscopic Resection; EMR: Endoscopic Mucosal Resection; ESD: Endoscopic Submucosal Dissection
Introduction
Early esophageal cancer is defined as tumor limited to the mucosa or submucosa, and without lymphatic spread or distant metastasis. Intraepithelial neoplasia, including low-grade and high-grade intraepithelial neoplasia (HGIN), is defined as the precancerous change of esophageal cancer. Recent advances in endoscopic technology have increased detection rates of early esophageal cancer. In 31% of patients the esophageal cancer is detected early in situ. Along with endoscopic detection, more and more early esophageal lesions have been also treated by endoscopic options. Compared with conventional esophagectomy, endoscopic therapies for intraepithelial neoplasia and early esophageal cancer are viable alternatives with significantly lower morbidities. Endoscopic diagnosis and treatment for early esophageal cancer has represented the trend of future. Thus, this review summarizes recent progress on endoscopic diagnosis and treatment options for early esophageal cancer.
Advances in Endoscopic Diagnosis
The quality of the endoscopic image is an important factor in the detection of small esophageal lesions, which may harbor neoplasia or early esophageal cancer. Novel imaging techniques have been recently introduced that may be taking us closer to the “optical biopsy.” The capabilities of the “perfect” imaging modality would include the following characteristics: (1) detect dysplastic changes; (2) have microscopic resolution; (3) be able to obtain real-time images; (4) differentiate dysplasia from inflammation-related changes; (5) localize dysplasia for targeted biopsies; (6) be inexpensive [1]. Unfortunately, no one imaging modality exhibits all of these characteristics.
High-Resolution and Magnified Endoscopy
The endoscopic detection of abnormal mucosa is paramount. Conventional white light endoscopic images less reliably detect mucosal histology. Both high-resolution endoscopy (HRE) and magnification endoscopy are available and have significantly increased the sensitivity and specificity in the differentiation of mucosal lesions [2]. Highresolution endoscopes are now capable of discriminating objects that are as small as 0.01 mm. Magnified endoscopy can produce an increase in image size of up to 105-fold. These advances allow the possibility of detecting changes in the mucosa or potentially characterizing dysplasia and facilitating directed biopsies [3].
Narrow Band Imaging (NBI)
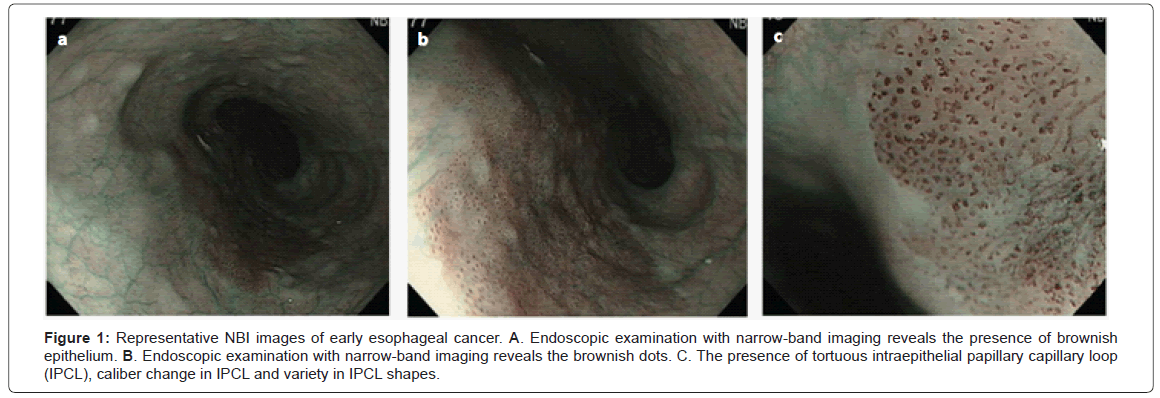
NBI uses an optical filter to select blue (440-460 nm) and green (540-560 nm) wavelength light. This technology improves the visibility of superficial capillaries and mucosal structures by optimizing the absorbance and scattering characteristics of light [4]. NBI allows clear visualization of the mucosa pit patterns and capillary patterns, and facilitates targeted biopsies. The authors of a recent meta-analysis reported the pooled sensitivity and specificity for the discrimination of HGIN or esophageal adenocarcinoma (EAC) in a field of Barrett’s esophagus (BE) as 96% and 94%, respectively. Compared with staining chromoendoscopy (methylene blue, indigo carmine, etc), which has been questioned to be no better than random biopsies [5], NBI offers advantages in detecting subtle mucosal lesions due to HGIN / EAC and in enabling directed biopsies to areas of concern. This modality has been proven to aid in the diagnostic precision for HGIN or EAC compared with white light endoscopy alone [6]. Recently, the authors from Asia– Pacific countries also demonstrated that NBI is useful for detection and characterization of superficial esophageal squamous cell cancer (SCC), and could replace chromoendoscopy in routine examination because it is easy to use and adds much information to conventional white light images [7]. Representative NBI images of early esophageal cancer were shown in Figure 1.
Figure 1: Representative NBI images of early esophageal cancer. A. Endoscopic examination with narrow-band imaging reveals the presence of brownish epithelium. B. Endoscopic examination with narrow-band imaging reveals the brownish dots. C. The presence of tortuous intraepithelial papillary capillary loop (IPCL), caliber change in IPCL and variety in IPCL shapes.
Autofluorescence Imaging (AFI)
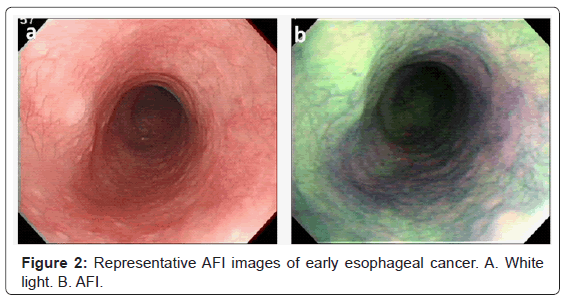
AFI is a technology that exploits certain endogenous substances in the tissue (fluorophores) which, when excited by light, emit light of a longer wavelength than the exciting light [8]. AFI produces images in which normal esophageal squamous and nondysplastic Barrett’s esophagus appear green, areas of HGIN/ EAC appear bluish- purple, and squamous islands amidst intestinal metaplasia appear pinkish. Advantages to AFI include its potential as a ‘red flag’ technique and the fact that it requires no additional contrast agent [9,10]. The main limitation is the high false positive rate [9,11]. Thus, other endoscopic options are needed to increase the accuracy of detecting suspicious lesions. Autofluorescence intensity and abnormal white light appearance of areas identified by AFI correlated with true positives [12]. A proof-ofprinciple study confirmed the feasibility of combining AFI and NBI for improving the accuracy of the endoscopic detection of HGIN in BE [9]. A trimodal prototype system with AFI and NBI integrated into a HRE endoscope was more effective than standard endoscopy in indetifying early-stage neoplasia in BE. AFI detected more lesions not seen with HRE, and NBI decreased the false-positive rate [13,14]. However, the use of AFI as part of a trimodal system (HRE, AFI, and NBI) that can identify occult neoplasia and assist in endoscopic mucosal resection warrants further investigation [15]. Representative AFI images of early esophageal cancer were shown in Figure 2.
Confocal Laser Endomicroscopy (CLE)
NBI, AFI, and chromoendoscopy allow the endoscopist to target suspicious areas to obtain biopsy and resection specimens for histological analysis. CLE is a recently released endoscopic imaging modality that is able to generate high-resolution microscopic-like images of the mucosal layer. After the patient is injected with an intravenous contrast agent, a probe is introduced through the endoscopic working channel, and a low-power laser is trained on to the mucosa. It is the detection of the fluorescent light back from the tissue that provides an “optical biopsy” for histologic examination of the superficial mucosal layer in question [16]. CLE has the potential to distinguish different types of epithelial cells and differentiate both dysplasia and neoplasia in BE or EAC [17-19]. Compared with standard four-quadrant biopsy for evaluation of neoplasia in BE, probe-based CLE could be regarded as non-inferior to endoscopic biopsy in excluding neoplasia of Barrett’s esophagus mucosa [20]. In a recent prospective randomized controlled trial, CLE with targeted biopsy significantly improved the diagnostic yield for endoscopically inapparent BE neoplasia compared to a standard endoscopy with a random-biopsy protocol. CLE with targeted biopsy also greatly reduced the number of biopsies needed per patient and allows some patients without neoplasia to completely forgo mucosal biopsy [21]. In a pilot study of patients with superficial esophageal SCC, CLE could also be used to distinguish cancerous from normal epithelium, which gives it potential value for early detection of esophageal carcinoma. The difficulty in obtaining good images in the esophagus by CLE was a latent problem [22].
It is important to remember that this approach is examiner dependent because the interpretation of the “optical biopsy” is performed in real time in vivo. Endoscopists who perform CLE must have some familiarity with tissue architecture of the gastrointestinal tract in order to interpret images [23]. Due to its low positive predictive values and sensitivity, CLE may currently not replace standard biopsy techniques for the diagnosis of esophageal lesions. Further technical development of CLE and a better understanding of its role in relation to other imaging technologies are necessary [20].
Optical Coherence Tomography (OCT)
OCT uses near-infrared light to produce high-resolution (10 μm axial resolution) cross-sectional images of gastrointestinal mucosa with depth of 1-2 mm. OCT is also performed by placing a probe through the working channel of an endoscope and is a noninvasive technique able to capture micrometer resolution of biological tissues with 3-dimensional image output. OCT processes coherent back scattered light providing real-time in-vivo microscopic resolution of subsurface tissue architectural morphology [24]. It has been investigated for detecting dysplasia in BE with a 78% accuracy in a prospective, double-blinded study [25]. A preliminary descriptive clinical study demonstrated the feasibility of carrying out ultrahigh resolution OCT imaging in conjunction with standard endoscopy for in vivo real-time imaging of Barrett’s esophagus, dysplasia, and esophageal adenocarcinoma [26]. In addition, ultrahigh resolution OCT imaging is capable of identifying subsquamous Barrett’s epithelium in patients who have undergone ablative therapy, which is not visible on standard endoscopic examination [27]. In superficial esophageal SCC, OCT might also be useful for the preoperative staging with a high degree of accuracy [28].
Problems identified were the variability in endoscopists’ accuracy rates, difficulty in real-time interpretation, and the need for refined criteria of dysplasia by OCT imaging. OCT has the potential to diagnose dysplasia. This techniques, however, is still in a state of development and is not currently commercially available.
Spectroscopy
Spectroscopic modalities can analyze the interactions between light and tissue to provide information about the nanoscale architecture of the esophageal epithelium. These technologies are being investigated in preliminary studies to differentiate between normal and abnormal tissue [1], and some have found their way to the clinical arena. Recently, some researchers developed a system called endoscopic polarized scanning spectroscopy, which offered great promise for the early detection of dysplasia in Barrett’s esophagus. They concluded that if the technique were to be used to guide biopsy routinely, unnecessary biopsies would be avoided and focal dysplastic spots would be biopsied that otherwise would be missed [29].
Spectroscopic modalities include light scattering, reflectance, and Raman-based technology. Light scattering spectroscopy uses the scattered light across a full spectrum and can elicit details about characteristics of cell nuclei. Light scattering spectroscopy has been demonstrated to quantify differences between dysplastic and nondysplastic tissue in Barrett’s esophagus [30,31], and thus can serve as a guide for biopsy [32,33]. Reflectance spectroscopy is used to measure the color and intensity of reflected light. Raman scattering or Raman spectroscopy is an inelastic scattering technique which provides a biochemical fingerprint of a tissue sample under interrogation. It has been demonstrated that Raman spectroscopy can be used to identify and classify the subtle pre-malignant biochemical changes related to the carcinogenesis process in the oesophagus [34-36]. Raman spectroscopy may have promising potential for the real-time, in vivo diagnosis and detection of esophageal cancer during clinical endoscopic examination [37,38].
Endocytoscopy
Endocytoscopy, adapted for clinical use in 2003, tries to approach in-vivo microscopy. This technology is an ultra-high-power magnifying endoscope that enables magnifications of up to 450 or 1125-fold, thereby allowing the identification of single cells and nuclei in a limited sampling area less than half a millimeter in diameter. In a pilot trail in patients with various types of esophageal lesions, the endocytoscopic diagnoses were compared with the histopathological diagnoses. The overall accuracy of endocytoscopy for differentiating between nonmalignant tissue and malignant tissue was 82% [39]. Some studies have demonstrated that this diagnostic tool makes it possible to omit histological examination of biopsy samples in approximately 84% of esophageal SCC, as evidence for both an increase of cell density and nuclear abnormalities is considered to be convincing proof that a lesion is malignant [40-42]. It has also been investigated the accuracy in correlation with histology in BE in a study [43]. With 450-fold magnification, only 23% of images were sufficiently interpretable to identify characteristics of neoplasia, whereas for 1125-fold magnification, it was 41%.
However, when not supported by macroscopic evidence, endoscopic histology using endocytoscopy lacked sufficient image quality to be of use in identifying neoplastic areas [43]. Moreover, it is also not surprising that the quality of the majority of images was poor, because of the difficulty of maintaining a moving organ in focus using an endoscope. In addition, endoscopists are not trained in assessment of cytology, and a pathologist was needed. Appropriate preconditioning to constantly obtain sufficient image quality and universal criteria for endocytoscopic diagnosis of various diseases are essential before clinical application [42].
Endoscopic Ultrasound (EUS)
The precise differentiation of esophageal wall layers, direct imaging of the surrounding organs and tissues, and tissue sampling with fine needle aspiration has allowed EUS to play a pivotal role in the staging of patients with esophageal cancer. EUS accuracy in esophageal cancer staging has been evaluated in multiple studies; it was found to be the most accurate imaging modality in the assessment of tumor invasion and locoregional lymph node status [44].
The recent increase in the incidence of superficial esophageal cancer and promising developments in potentially curative endoscopic therapies have placed EUS in a central position with regard to decision making. EUS has emerged as a requisite in the staging of patients with early esophageal cancer. In a meta-analysis, the pooled sensitivity and specificity to diagnose T1 stage cancer was 81.6% and 99.4%, respectively, which is much lower than for more advanced T stages [45]. The largest study prospectively evaluating staging in early esophageal cancer using high-frequency ultrasound probes revealed an overall accuracy of 80%. However, the sensitivity for submucosal invasion fell to 48% [46]. The location of tumor with submucosal invasion and the depth of submucosal invasion were found to play an important role in staging.
However with current technology, EUS – even with high-frequency probes has limited accuracy in differentiating early tumors that are confined to the mucosa from those that invade the submucosa, and it is used principally to screen for lymphatic metastases in the setting of early-stage disease [44]. In addition, the accuracy of EUS is operatordependent. Operator’s experience and machine-dependent factors may play an important role in the accurate staging of esophageal carcinoma [47]. Improvement in technology will probably decrease instrumentdependent factors causing artifacts.
Advances in Endoscopic Therapy
Endoscopic therapy has become a viable alternative for patients with neoplasia or early esophageal cacer due to the low rates of lymphatic or hematogenous dissemination, corroborated with the adverse effects of esophagectomy [48]. These treatments used either alone or in combination with other techniques, can be grouped into two categories: (1) ablative and (2) non-ablative. Ablative approaches, which use different energy sources to obliterate the lesions, include photodynamic therapy, radiofrequency ablation, argon plasma coagulation, and cryotherapy. Non-ablative endoscopic approaches primarily comprise endoscopic resection and variations of this technique.
Photodynamic Therapy (PDT)
PDT has been the most investigated of all the ablative techniques developed for dysplasia and early esophageal cacer treatment. A photosensitizing agent selectively absorbed by fast-growing cells, such as cancer cells, is introduced intravenously or orally. Activation of the photosensitizer is achieved by endoscopically applied laser directly to the malignant tumor. This results in the formation of free oxygen radicals in the tumor tissue leading to ischemic necrosis of the tumor cells. A prospective randomized study showed a significant difference for the study group treated with PDT and omeprazole comparative with the group treated with omeprazole only regarding the ablation of high grade dysplasia and the recurrence of neoplasia [49]. After PDT with Photofrin and red light, the adverse effects were minimal and the median survival was 60.5 months. A system review also showed PDT is effective for the ablation of dysplasia in Barrett’s esophagus, although the frequency of adverse events is quite high [50]. The efficacy of PDT for superficial esophageal SCC has been evaluated in multiple studies [51-53]. PDT eradicated early esophageal SCC (Tis and T1a) of the esophagus efficiently [51,52]. A long-term follow-up study revealed that PDT was a potentially curative treatment for large superficial esophageal SCC. Complete remission was achieved in 87% patients, and after a median follow-up period of 64 months after PDT, the overall 5-year survival rate was 76% [53].
However, its use has been limited by serious side effects including prolonged cutaneous photosensitivity, stricture formation and recurrence. A long-term follow-up study demonstrated that only up to one-third of patients could be successfully treated with PDT without having a recurrence [54]. In addition, almost one-half developed very symptomatic and refractory esophageal strictures. Recurrence of dysplasia/neoplasia after PDT ablation was associated with advanced age, smoking, and residual BE [55]. The incidence of severe stricture was related to the light dose. Decreasing the light dose below 115 J/cm appeared to result in a reduced incidence rate of severe stricture but higher relative frequencies of residual HGIN/T1 in BE [56]. Moreover, perhaps the biggest drawback of PDT ablation therapies for BE is the well-recognized risk that the regenerating squamous epithelium could cover residual underlying Barrett’s esophagus mucosa, giving rise to the problem of ‘buried glands’. Buried glands have been reported in up to 24% of patients following 5-aminolaevulinic acid PDT [57]. There is concern that such glands are difficult to access at endoscopic surveillance, but might still be at risk of malignant progression. The recent development of radiofrequency ablation (RFA) might have gone some way towards addressing the problem of ‘buried glands’.
Radiofrequency Ablation (RFA)
RFA delivers the direct application of thermal energy to the mucosa of the esophagus. This is accomplished with either an ablation catheter for circumferential treatment or an endoscope mounted device for more focal ablation. The energy delivered provides uniform treatment to a depth of approximately 500 μm. Therefore, the depth of treatment does not enter into the submucosal layer, and the risk of stenosis is reduced.
The authors of initial pilot studies reported complete eradication of HGIN in up to 90% of patients [58]. These encouraging results led to a multicenter, sham-controlled trial in patients with dysplastic BE [59-61]. In this trial, 127 highly selected patients with low-grade and HGIN were randomized to treatment with RFA or a sham endoscopic ablation. Primary outcomes at 12 months showed that in those patients with HGIN, 81% had complete eradication of dysplasia with ablation compared with 19% in the control arm (P<0.001). In addition, shamcontrolled trial of RFA was associated with improvement in diseasespecific health-related quality of life [60]. During a mean time of 3.05 years follow-up, dysplasia remained eradicated in >85% of patients and intestinal metaplasia in >75%, without maintenance RFA. Serious adverse events occurred in 4 of 119 subjects (3.4%); the rate of stricture was 7.6%. There was no cancer-related morbidity or mortality [61]. So in subjects with dysplastic BE, RFA therapy has an acceptable safety profile, is durable, and is associated with a low rate of disease progression, for up to 3 years. Besides, some studies also suggested that RFA for squamous HGIN and early esophageal SCC was feasible and effective, speculating that RFA might also be suited for early squamous neoplasia of the esophagus [62,63].
An inherent weakness of RFA ablation, as with all other nonresectional endoscopic ablation techniques, is that it does not provide a tissue specimen for histopathological assessment. This means that if RFA is used for the treatment of intramucosal cancer, there is no way to assess the depth of tumor invasion or to confirm that the tumor treated is actually confined to the mucosa,
Cryotherapy
Liquid nitrogen or freezing carbon dioxide gas is sprayed directly onto the tumour via an open-tipped catheter. The targeted tissue is “frozen”, and then allowed to thaw. Repeating the “freeze and thaw” cycle subsequently destroys the lesion. The authors of a pilot study demonstrated complete reversal of BE with dysplasia in 82% of patients treated with cryotherapy [64]. In a recent prospective open-label study, 31 patients with either HGIN (n = 26) or intramucosal carcinoma (n = 5) who were nonoperative candidates were treated with cryotherapy [65]. At a median follow-up of 12 months, 90% of patients demonstrated at least a partial response. A complete response rate was seen in 68% of those with HGIN and 80% of those with carcinoma. The authors of another slightly larger retrospective cohort study demonstrated that those patients completing all planned treatments had 97% eradication of HGIN [66]. The most common complaint was chest pain, followed by dysphagia, odynophagia, and sore throat [67]. Although endoscopic spray cryotherapy may be a promising ablative modality for treatment of BE and early esophageal cancer, limitations of current studies include small sample sizes and short durations of follow up, and further studies are needed to validate the promising early results.
Endoscopic Resection (ER)
Endoscopic resection (ER) is the general term for all of the different resection techniques used to treat neoplastic and uncertain lesions in the gastrointestinal tract. Endoscopic resection results in complete eradication of all disease with a possible restoration of the molecular baseline within the neosquamous epithelium. This may translate to a lower recurrence rate. In contrast to ablative treatment methods such as PDT and RFA, ER allows histological assessment of the resected specimen in order to assess the depth of infiltration of the tumour and freedom from neoplasia at the lateral and (more importantly) basal margins, imitating the surgical situation. These significant advantages of ER are the main reason why ER should be preferred to ablative treatment methods, even RFA, whenever possible, especially bearing in mind the low accuracy of EUS regarding local tumour staging [68].
The conventional ER procedure is endoscopic mucosal resection (EMR). Several EMR techniques of endoscopic resection have been described, including with or without a suction device [68]. The different techniques have shown very similar results in terms of the specimen resected, and the success of the procedure. Most studies report complete resection rates of about 80–90%, but with strict patient selection, R0 resection can be achieved in 98–100% of patients [69].
The major drawback of EMR technique appears to be that only small lesions with a diameter of less than 20 mm can be resected en bloc with tumour-free lateral margins. Ulcerated lesions often have fibrosis attaching the submucosa to the lamina muscularis propria, resulting in failure of the lesion to lift. In these cases, EMR is not advisable, or should only be performed with caution. Larger lesions can usually be resected completely using the piecemeal technique, but this method appears to be associated with a higher recurrence rate because of small neoplastic residues resulting from insufficient overlapping of the resection areas. At a median follow-up of 61 months, Pech et al. [70] found metachronous tumors in 21% of 231 patients, while the Mayo Clinic reported recurrences in 12% of 132 patients over a similar time-frame [71]. Similarly a follow-up study in Japan found metachronous lesions in up to 15% of patients undergoing EMR for squamous cell cancer [72]. In addition, en bloc resection allows more accurate histological evaluation of the neoplastic lesion, especially of the lateral and basal margins. A new resection technique, endoscopic submucosal dissection (ESD), was therefore developed.
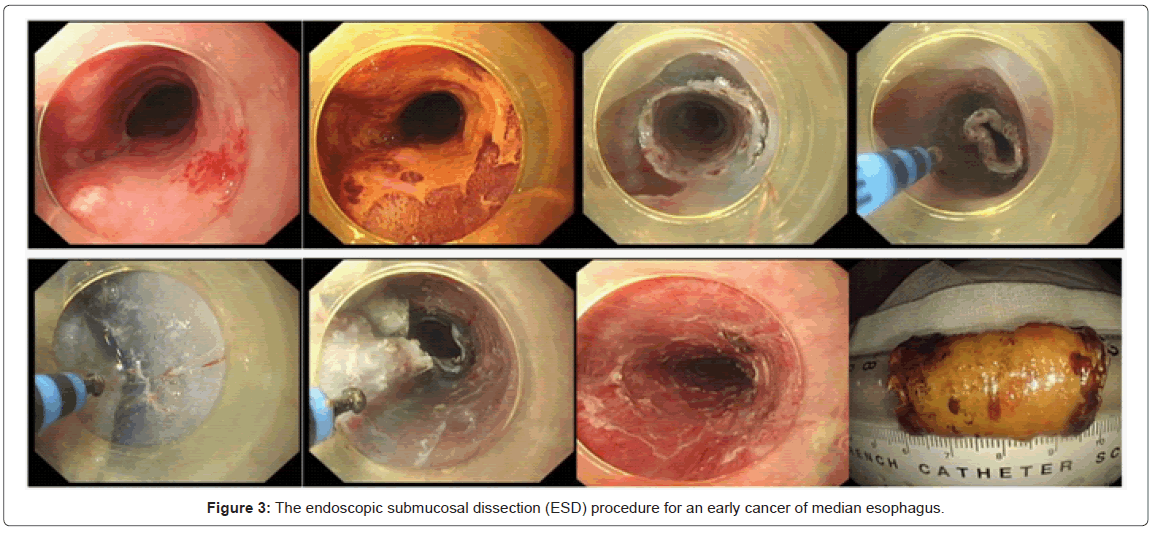
The ESD procedure is a method using an electronic knife to dissect the submucosal layer underneath the carcinoma in order to obtain a large resection specimen with the neoplasm resected en bloc. It is a three step process in which the tumour is first marked using electrocautery, then raised by injecting a saline solution below it, and finally excised using a electrocautery knife [73]. The endoscopic submucosal dissection (ESD) procedure for an early cancer of median esophagus was shown in Figure 3.
Recently, a retrospective study from Japan assessed the long-term outcomes of ESD for esophageal squamous cell neoplasms, and revealed that ESD is a potentially curative treatment for superficial esophageal squamous cell neoplasms [74]. In this study, 107 superficial esophageal squamous cell neoplasms in 84 patients were treated by ESD. The rates of en bloc resection and complete resection were 100% and 88%, respectively. One patient had local recurrence 6 months after ESD. In 2 patients with intramucosal invasive carcinomas in the muscularis mucosa, distant metastases were observed 9 and 18 months after ESD. During the the median observation of 632 days, 3 patients died of esophageal carcinoma. Another study from Western also confirmed that that ESD is a potentially curative treatment for superficial esophageal squamous cell neoplasia [75]. Besides, studies also showed that ESD can be adequately adopted as an effective treatment for superficial adenocarcinomas at the esophagogastric junction, with no local or distant recurrences observed in any patient achieving curative resection during long-term follow-up [76,77].
Some studies comparing ESD and conventional EMR in early esophageal cancer have been published. A retrospective cohort study from Japan involved 300 patients with esophageal SCC who underwent either EMR (n = 184) or ESD (n = 116). En bloc resection and the local recurrence rate were significantly better in the ESD group (P = 0.0009 and 0.065, respectively). During the median observation of 65 months, ESD gave fewer local recurrence compared with EMR [78]. Another report also demonstrated that larger superficial esophageal tumors (>15 mm) should be treated with ESD to reduce local recurrence [79].
Some reports demonstrate that ESD may be associated with a higher risk of strictures and an esophageal perforation rate compared with EMR [80], however, in comparative medical center, these complication rates did not differ significantly [77-79]. Most complications can be readily dealt with by endoscopic dilatation or clips. Only few complications need to be dealt with by surgery.
Summary
The rapid development of endoscopic technology has improved current endoscopic diagnostic and therapic capabilities for early esophageal cancer. Compared with chromoendoscopy, using Highresolution and magnified endoscopy, NBI, AFI or CLE appears to be ease of use and the possibility of targeted biopsies as subtle mucosal lesions are detected. OCT, Spectroscopy and Endocytoscopy remain experimental. EUS has emerged as a requisite in the staging of patients with esophageal lesions. In terms of endoscopic treatment, there are now long-term randomized controlled data concerning the effectiveness of PDT and cryotherapy; however, the ablation may be not complete, and complications such as strictures and recurrence remain problematic. Endoscopic radiofrequency ablation appears a durable safety profile, which is associated with a low rate of disease progression compared with other ablative techniques. Unlike ablative techniques, endoscopic resection permits histopathological assessment, similar to surgery. These significant advantages of ER are the main reason why ER should be preferred to ablative treatment methods. As a new resection technique, ESD results in complete eradication of all disease and may translate to a lower recurrence rate than conventional EMR procedure.
Financial Support
This study was supported by grants from the Major Project of Shanghai Municipal Science and Technology Committee (10411955900), Medical Leading Project of Shanghai Municipal Science and Technology Committee (11411950502), and Special Project of Public Welfare Fund of Ministry of Health (200902002-3).
References
- Shahid MW, Wallace MB (2010) Endoscopic imaging for the detection of esophageal dysplasia and carcinoma. Gastrointest Endosc Clin N Am 20: 11-24.
- Reddymasu SC, Sharma P (2008) Advances in endoscopic imaging of the esophagus. Gastroenterol Clin North Am 37: 763-774.
- Stevens PD, Lightdale CJ, Green PH, Siegel LM, Garcia-Carrasquillo RJ, et al. (1994) Combined magnification endoscopy with chromoendoscopy for the evaluation of Barrett's esophagus. Gastrointest Endosc 40: 747-749.
- Gono K, Obi T, Yamaguchi M, Ohyama N, Machida H, et al. (2004) Appearance of enhanced tissue features in narrow-band endoscopic imaging. J Biomed Opt 9: 568-577.
- Ngamruengphong S, Sharma VK, Das A (2009) Diagnostic yield of methylene blue chromoendoscopy for detecting specialized intestinal metaplasia and dysplasia in Barrett's esophagus: a meta-analysis. Gastrointest Endosc 69: 1021-1028.
- Wolfsen HC, Crook JE, Krishna M, Achem SR, Devault KR, et al. (2008) Prospective, controlled tandem endoscopy study of narrow band imaging for dysplasia detection in Barrett's Esophagus. Gastroenterology 135: 24-31.
- Uedo N, Fujishiro M, Goda K, Hirasawa D, Kawahara Y, et al. (2011) Role of narrow band imaging for diagnosis of early-stage esophagogastric cancer: current consensus of experienced endoscopists in Asia-Pacific region. Dig Endosc 23: 58-71.
- Falk GW (2009) Autofluorescence endoscopy. Gastrointest Endosc Clin N Am 19: 209-220.
- Kara MA, Peters FP, Fockens P, ten Kate FJ, Bergman JJ. Endoscopic video-autofluorescence imaging followed by narrow band imaging for detecting early neoplasia in Barrett's esophagus. Gastrointest Endosc 2006; 64: 176-185.
- Suzuki H, Saito Y, Ikehara H, Oda I (2009) Evaluation of visualization of squamous cell carcinoma of esophagus and pharynx using an autofluorescence imaging videoendoscope system. J Gastroenterol Hepatol 24: 1834-1839.
- Kara MA, Peters FP, Ten Kate FJ, Van Deventer SJ, Fockens P, et al. (2005) Endoscopic video autofluorescence imaging may improve the detection of early neoplasia in patients with Barrett's esophagus. Gastrointest Endosc 61: 679-685.
- Curvers WL, Singh R, Wallace MB, Song LM, Ragunath K, et al. (2009) Identification of predictive factors for early neoplasia in Barrett’s esophagus after autofluorescence imaging: a stepwise multicenter structured assessment. Gastrointest Endosc 70: 9-17.
- Curvers WL, Singh R, Song LM, Wolfsen HC, Ragunath K, et al. (2008) Endoscopic tri-modal imaging for detection of early neoplasia in Barrett’s oesophagus: a multicentre feasibility study using high-resolution endoscopy, autofluorescence imaging and narrow band imaging incorporated in one endoscopy system. Gut 57:167-172.
- Curvers WL, Herrero LA, Wallace MB, Wong Kee Song LM, Ragunath K, et al. (2010) Endoscopic tri-modal imaging is more effective than standard endoscopy in identifying early-stage neoplasia in Barrett's esophagus. Gastroenterology 139: 1106-1114.
- Thomas T, Singh R, Ragunath K (2009) Trimodal imaging-assisted endoscopic mucosal resection of early Barrett's neoplasia. Surg Endosc 23: 1609-1613.
- Wang TD, Van Dam J (2004) Optical biopsy: a new frontier in endoscopic detection and diagnosis. Clin Gastroenterol Hepatol 2: 744-753.
- Leung KK, Maru D, Abraham S, Hofstetter WL, Mehran R, et al. (2009) Optical EMR: confocal endomicroscopy-targeted EMR of focal high-grade dysplasia in Barrett's esophagus. Gastrointest Endosc 69: 170-172.
- Pohl H, Rösch T, Vieth M, Koch M, Becker V, et al. (2008) Miniprobe confocal laser microscopy for the detection of invisible neoplasia in patients with Barrett's oesophagus. Gut 57: 1648-1653.
- Wallace MB, Sharma P, Lightdale C, Wolfsen H, Coron E, et al. (2010) Preliminary accuracy and interobserver agreement for the detection of intraepithelial neoplasia in Barrett's esophagus with probe-based confocal laser endomicroscopy. Gastrointest Endosc 72: 19-24.
- Bajbouj M, Vieth M, Rösch T, Miehlke S, Becker V, et al. (2010) Probe-based confocal laser endomicroscopy compared with standard four-quadrant biopsy for evaluation of neoplasia in Barrett's esophagus. Endoscopy 42: 435-440.
- Dunbar KB, Okolo P 3rd, Montgomery E, Canto MI (2009) Confocal laser endomicroscopy in Barrett's esophagus and endoscopically inapparent Barrett's neoplasia: a prospective, randomized, double-blind, controlled, crossover trial. Gastrointest Endosc 70: 645-654.
- Liu H, Li YQ, Yu T, Zhao YA, Zhang JP, et al. (2009) Confocal laser endomicroscopy for superficial esophageal squamous cell carcinoma. Endoscopy 41: 99-106.
- Nguyen NQ, Leong RW (2008) Current application of confocal endomicroscopy in gastrointestinal disorders. J Gastroenterol Hepatol 23: 1483-1491.
- Evans JA, Nishioka NS (2005) The use of optical coherence tomography in screening and surveillance of Barrett's esophagus. Clin Gastroenterol Hepatol 3: S8-11.
- Isenberg G, Sivak MV Jr, Chak A, Wong RC, Willis JE, et al. (2005) Accuracy of endoscopic optical coherence tomography in the detection of dysplasia in Barrett's esophagus: a prospective, double-blinded study. Gastrointest Endosc 62: 825-831.
- Chen Y, Aguirre AD, Hsiung PL, Desai S, Herz PR, et al. (2007) Ultrahigh resolution optical coherence tomography of Barrett's esophagus: preliminary descriptive clinical study correlating images with histology. Endoscopy 39: 599-605.
- Cobb MJ, Hwang JH, Upton MP, Chen Y, Oelschlager BK, et al. (2010) Imaging of subsquamous Barrett's epithelium with ultrahigh-resolution optical coherence tomography: a histologic correlation study. Gastrointest Endosc 71: 223-230.
- Hatta W, Uno K, Koike T, Yokosawa S, Iijima K, et al. (2010) Optical coherence tomography for the staging of tumor infiltration in superficial esophageal squamous cell carcinoma. Gastrointest Endosc 71: 899-906.
- Qiu L, Pleskow DK, Chuttani R, Vitkin E, Leyden J, et al. (2010) Multispectral scanning during endoscopy guides biopsy of dysplasia in Barrett's esophagus. Nat Med 16: 603-606.
- Wallace MB, Perelman LT, Backman V, Crawford JM, Fitzmaurice M, et al. (2000) Endoscopic detection of dysplasia in patients with Barrett's esophagus using light-scattering spectroscopy. Gastroenterology 119: 677-682.
- Georgakoudi I, Jacobson BC, Van Dam J, Backman V, Wallace MB, et al. (2001) Fluorescence, reflectance, and light-scattering spectroscopy for evaluating dysplasia in patients with Barrett's esophagus. Gastroenterology 120: 1620-1629.
- Qiu L, Chuttani R, Zhang S, Feng J, Itani S, et al. (2009) Diagnostic imaging of esophageal epithelium with clinical endoscopic polarized scanning spectroscopy instrument. Conf Proc IEEE Eng Med Biol Soc 2009: 1997-2000.
- Lovat LB, Johnson K, Mackenzie GD, Clark BR, Novelli MR, et al. (2006) Elastic scattering spectroscopy accurately detects high grade dysplasia and cancer in Barrett's oesophagus. Gut 55: 1078-1083.
- Kendall C, Stone N, Shepherd N, Geboes K, Warren B, et al. (2003) Raman spectroscopy, a potential tool for the objective identification and classification of neoplasia in Barrett's oesophagus. J Pathol 200: 602-609.
- Hutchings J, Kendall C, Shepherd N, Barr H, Stone N (2010) Evaluation of linear discriminant analysis for automated Raman histological mapping of esophageal high-grade dysplasia. J Biomed Opt 15: 066015.
- Kendall C, Day J, Hutchings J, Smith B, Shepherd N, et al. (2010) Evaluation of Raman probe for oesophageal cancer diagnostics. Analyst 135: 3038-3041.
- Bergholt MS, Zheng W, Lin K, Ho KY, Teh M, et al. (2011) In vivo diagnosis of esophageal cancer using image-guided Raman endoscopy and biomolecular modeling. Technol Cancer Res Treat 10: 103-112.
- Bergholt MS, Zheng W, Lin K, Ho KY, Teh M, et al. (2011) Characterizing variability in in vivo Raman spectra of different anatomical locations in the upper gastrointestinal tract toward cancer detection. J Biomed Opt 16: 037003.
- Inoue H, Sasajima K, Kaga M, Sugaya S, Sato Y, et al. (2006) Endoscopic in vivo evaluation of tissue atypia in the esophagus using a newly designed integrated endocytoscope: a pilot trial. Endoscopy 38: 891-895.
- Kumagai Y, Kawada K, Yamazaki S, Iida M, Momma K, et al. (2009) Endocytoscopic observation for esophageal squamous cell carcinoma: can biopsy histology be omitted? Dis Esophagus 22: 505-512.
- Kumagai Y, Kawada K, Yamazaki S, Iida M, Ochiai T, et al. (2010) Endocytoscopic observation of esophageal squamous cell carcinoma. Dig Endosc 22: 10-16.
- Fujishiro M, Takubo K, Sato Y, Kaise M, Niwa Y, et al. (2007) Potential and present limitation of endocytoscopy in the diagnosis of esophageal squamous-cell carcinoma: a multicenter ex vivo pilot study. Gastrointest Endosc 66: 551-555.
- Pohl H, Koch M, Khalifa A, Papanikolaou IS, Scheiner K, et al. (2007) Evaluation of endocytoscopy in the surveillance of patients with Barrett's esophagus. Endoscopy 39: 492-496.
- Attila T, Faigel DO (2009) Role of endoscopic ultrasound in superficial esophageal cancer. Dis Esophagus 22: 104-112.
- Puli SR, Reddy JB, Bechtold ML, Antillon D, Ibdah JA, et al. (2008) Staging accuracy of esophageal cancer by endoscopic ultrasound: A meta-analysis and systematic review. World J Gastroenterol14:1479-1490.
- May A, Günter E, Roth F, Gossner L, Stolte M, et al. (2004) Accuracy of staging in early oesophageal cancer using high resolution endoscopy and high resolution endosonography: a comparative, prospective, and blinded trial. Gut 53: 634-640.
- Catalano MF, Sivak MV Jr, Bedford RA, Falk GW, van Stolk R, et al. (1995) Observer variation and reproducibility of endoscopic ultrasonography. Gastrointest Endosc 41: 115-120.
- McCann P, Stafinski T, Wong C, Menon D (2011) The safety and effectiveness of endoscopic and non-endoscopic approaches to the management of early esophageal cancer: a systematic review. Cancer Treat Rev 37: 11-62.
- Overholt BF, Wang KK, Burdick JS, Lightdale CJ, Kimmey M, et al. (2007) Five-year efficacy and safety of photodynamic therapy with Photofrin in Barrett's high-grade dysplasia. Gastrointest Endosc 66: 460-468.
- Sánchez A, Reza M, Blasco JA, Callejo D (2010) Effectiveness, safety, and cost-effectiveness of photodynamic therapy in Barrett's esophagus: a systematic review. Dis Esophagus 23: 633-640.
- Savary JF, Grosjean P, Monnier P, Fontolliet C, Wagnieres G, et al. (1998) Photodynamic therapy of early squamous cell carcinomas of the esophagus: a review of 31 cases. Endoscopy 30: 258-265.
- Eickhoff A, Jakobs R, Weickert U, Hartmann D, Schilling D, et al. (2006) Long-Segment early squamous cell carcinoma of the proximal esophagus: curative treatment and long-term follow-up after 5-aminolevulinic acid (5-ALA)-photodynamic therapy. Endoscopy 38: 641-643.
- Tanaka T, Matono S, Nagano T, Murata K, Sueyoshi S, et al. (2011) Photodynamic therapy for large superficial squamous cell carcinoma of the esophagus. Gastrointest Endosc 73: 1-6.
- Keeley SB, Pennathur A, Gooding W, Landreneau RJ, Christie NA, et al. (2007) Photodynamic therapy with curative intent for Barrett's esophagus with high grade dysplasia and superficial esophageal cancer. Ann Surg Oncol 14: 2406-2410.
- Badreddine RJ, Prasad GA, Wang KK, Song LM, Buttar NS, et al. (2010) Prevalence and predictors of recurrent neoplasia after ablation of Barrett's esophagus. Gastrointest Endosc 71: 697-703.
- Panjehpour M, Overholt BF, Phan MN, Haydek JM (2005) Optimization of light dosimetry for photodynamic therapy of Barrett's esophagus: efficacy vs. incidence of stricture after treatment. Gastrointest Endosc 61: 13-18.
- Kelty CJ, Ackroyd R, Brown NJ, Stephenson TJ, Stoddard CJ, et al. (2004) Endoscopic ablation of Barrett's oesophagus: a randomized-controlled trial of photodynamic therapy vs. argon plasma coagulation. Aliment Pharmacol Ther 20: 1289-1296.
- Ganz RA, Overholt BF, Sharma VK, Fleischer DE, Shaheen NJ, et al. (2008) Circumferential ablation of Barrett's esophagus that contains high-grade dysplasia: a U.S. Multicenter Registry. Gastrointest Endosc 68: 35-40.
- Shaheen NJ, Sharma P, Overholt BF, Wolfsen HC, Sampliner RE, et al. (2009) Radiofrequency ablation in Barrett's esophagus with dysplasia. N Engl J Med 360: 2277-2288.
- Shaheen NJ, Peery AF, Hawes RH, Rothstein RI, Spechler SJ, et al. (2010) Quality of life following radiofrequency ablation of dysplastic Barrett's esophagus. Endoscopy 42: 790-799.
- Shaheen NJ, Overholt BF, Sampliner RE, Wolfsen HC, Wang KK, et al. (2011) Durability of Radiofrequency Ablation in Barrett's Esophagus with Dysplasia. Gastroenterology 141: 460-468.
- Zhang YM, Bergman JJ, Weusten B, Dawsey SM, Fleischer DE, et al. (2010) Radiofrequency ablation for early esophageal squamous cell neoplasia. Endoscopy 42: 327-333.
- van Vilsteren FG, Alvarez Herrero L, Pouw RE, ten Kate FJ, Visser M, et al. (2011) Radiofrequency ablation for the endoscopic eradication of esophageal squamous high grade intraepithelial neoplasia and mucosal squamous cell carcinoma. Endoscopy 43: 282-290.
- Johnston MH, Eastone JA, Horwhat JD, Cartledge J, Mathews JS, et al. (2005) Cryoablation of Barrett's esophagus: a pilot study. Gastrointest Endosc 62: 842-848.
- Dumot JA, Vargo JJ 2nd, Falk GW, Frey L, Lopez R, et al. (2009) An open-label, prospective trial of cryospray ablation for Barrett's esophagus high-grade dysplasia and early esophageal cancer in high-risk patients. Gastrointest Endosc 70: 635-644.
- Shaheen NJ, Greenwald BD, Peery AF, Dumot JA, Nishioka NS, et al. (2010) Safety and efficacy of endoscopic spray cryotherapy for Barrett's esophagus with high-grade dysplasia. Gastrointest Endosc 71: 680-685.
- Greenwald BD, Dumot JA, Horwhat JD, Lightdale CJ, Abrams JA (2010) Safety, tolerability, and efficacy of endoscopic low-pressure liquid nitrogen spray cryotherapy in the esophagus. Dis Esophagus 23: 13-19.
- Pech O, May A, Rabenstein T, Ell C (2007) Endoscopic resection of early oesophageal cancer. Gut 56: 1625-1634.
- Gan S, Watson DI (2010) New endoscopic and surgical treatment options for early esophageal adenocarcinoma. J Gastroenterol Hepatol 25: 1478-1484.
- Pech O, Behrens A, May A, Nachbar L, Gossner L, et al. (2008) Long-term results and risk factor analysis for recurrence after curative endoscopic therapy in 349 patients with high-grade intraepithelial neoplasia and mucosal adenocarcinoma in Barrett's oesophagus. Gut 57: 1200-1206.
- Prasad GA, Wu TT, Wigle DA, Buttar NS, Wongkeesong LM, et al. (2009) Endoscopic and surgical treatment of mucosal (T1a) esophageal adenocarcinoma in Barrett's esophagus. Gastroenterology 137: 815-823.
- Shimizu Y, Tukagoshi H, Fujita M, Hosokawa M, Kato M, et al. (2001) Metachronous squamous cell carcinoma of the esophagus arising after endoscopic mucosal resection. Gastrointest Endosc 54: 190-194.
- ASGE Technology Committee, Kantsevoy SV, Adler DG, Conway JD, Diehl DL, et al. (2008) Endoscopic mucosal resection and endoscopic submucosal dissection. Gastrointest Endosc 68: 11-18.
- Ono S, Fujishiro M, Niimi K, Goto O, Kodashima S, et al. (2009) Long-term outcomes of endoscopic submucosal dissection for superficial esophageal squamous cell neoplasms. Gastrointest Endosc 70: 860-866.
- Repici A, Hassan C, Carlino A, Pagano N, Zullo A, et al. (2010) Endoscopic submucosal dissection in patients with early esophageal squamous cell carcinoma: results from a prospective Western series. Gastrointest Endosc 71: 715-721.
- Yoshinaga S, Gotoda T, Kusano C, Oda I, Nakamura K, et al. (2008) Clinical impact of endoscopic submucosal dissection for superficial adenocarcinoma located at the esophagogastric junction. Gastrointest Endosc 67: 202-209.
- Teoh AY, Chiu PW, Yu Ngo DK, Wong SK, Lau JY, et al. (2010) Outcomes of endoscopic submucosal dissection versus endoscopic mucosal resection in management of superficial squamous esophageal neoplasms outside Japan. J Clin Gastroenterol 44: e190-194.
- Takahashi H, Arimura Y, Masao H, Okahara S, Tanuma T, et al. (2010) Endoscopic submucosal dissection is superior to conventional endoscopic resection as a curative treatment for early squamous cell carcinoma of the esophagus (with video). Gastrointest Endosc 72: 255-264.
- Urabe Y, Hiyama T, Tanaka S, Yoshihara M, Arihiro K, et al. (2011) Advantages of endoscopic submucosal dissection versus endoscopic oblique aspiration mucosectomy for superficial esophageal tumors. J Gastroenterol Hepatol 26: 275-280.
- Mizuta H, Nishimori I, Kuratani Y, Higashidani Y, Kohsaki T, et al. (2009) Predictive factors for esophageal stenosis after endoscopic submucosal dissection for superficial esophageal cancer. Dis Esophagus 22: 626-631.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 19890
- [From(publication date):
June-2012 - Oct 31, 2025] - Breakdown by view type
- HTML page views : 14953
- PDF downloads : 4937