Review Article Open Access
Novel Atmospheric Biomolecule Ionization Technologies
Nelson G Chen1 , Jung-Lee Lin2 , Chen-Yi Wu2 , Kent J Gillig2 , Abdil Ozdemir3 * and Chung-Hsuan Chen2 *1Department of Electric and Computer Engineering, National Chiao Tung University, Taipei, Taiwan
2Genomics Research Center, Academia Sinica, Taipei, Taiwan
3Department of Chemistry, Faculty of Arts and Sciences, Sakarya University, Sakarya, Turkey
- *Corresponding Author:
- Chung-Hsuan Chen
Distinguished Research Fellow and Director
The Genomics Research Center, Academia Sinica
128 Academia Road, Sec. 2, Nankang, Taipei 115, Taiwan
Tel: +886-2-2787-1200
Fax: +886-2-2789-9923
E-mail: winschen@gate.sinica.edu.tw
Abdil Ozdemir
Department of Chemistry, Faculty of Arts and Sciences
Sakarya University, 54187 Esentepe, Sakarya, Turkey
E-mail: Abdil@yahoo.com
Received date: September 26, 2013; Accepted date: October 24, 2013; Published date: October 28, 2013
Citation: Chen NG, Lin JL, Wu CY, Gillig KJ, Ozdemir A, et al. (2013) Novel Atmospheric Biomolecule Ionization Technologies. J Anal Bioanal Tech S2:003. doi: 10.4172/2155-9872.S2-003
Copyright: © 2013 Chen NG, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
In general, atmospheric ionization is defined as an ionization process outside of the mass spectrometer vacuum chamber. During the past two decades, several novel atmospheric ionization methods were developed. Nevertheless, they can be divided into two major categories. One is direct analysis at real time (DART) and the other is modifications on electrospray ionization (ESI). In addition, some methods are used to analyze samples directly without any pretreatments. Those methods are often called as ambient ionization methods. Many atmospheric ionization methods are also considered as ambient ionizations. DART and desorption electrospray ionization are examples. Nevertheless, most atmospheric ionization methods involve the need of high voltage outside of the mass spectrometer vacuum chamber. In this review, we will only briefly introduce most of the existing methods which need high voltages. Most effort will be placed on the newly developed novel methods which do not need high voltages. They include Ultrasound Ionization (UI), Triboelectric Spray Ionization (TeSI) and Kelvin spray ionization (KeSI). These ionization methods don’t need to have any external high voltage power supply for ionization. They have the advantages of very soft ionization to keep bio molecular ions more similar to the structures in solution phase. Some related mechanisms will also be discussed.
Keywords
Atmospheric ionization; Ultrasound ionization; Triboelectric spray ionization; Kelvin spray ionization; Ambient ionization; Biomolecule ionization; Biological mass spectrometer
Introduction of Atmospheric Ionization Methods
Mass spectrometry has been regarded as one of the most valuable analytical instruments since any compound has its intrinsic mass value. However, mass spectrometry can only be used to measure the mass-tocharge ratio (m/z) of a charged particle. Therefore, ionization process is usually needed unless the particle to be measured has an intrinsic charge. Nevertheless, the ionization efficiency is usually significantly lower than 1%. For some molecules, ionization efficiency is practically zero such as ultra-large polysaccharide (MW>1,000,000 Da). Therefore, the development of efficient ionization has always been playing a key role in mass spectrometry. In the early time, most ionization processes were pursued inside of the vacuum chamber which contains the mass spectrometer. Nevertheless, it is inconvenient if a mass spectrometer is to be used to monitor samples at in situ or real-time basis. Thus, atmospheric ionization is preferred with the advantage of little or no complex sample preparations required. Major barriers encountered for atmospheric ionization are (1) ions can be quenched due to the collision if ionization is pursued at one atmosphere (2) low transmission efficiency for ions in interest to “safely” reach to the mass analyzer which usually is under vacuum and (3) high pumping requirement to keep the pressure inside of mass spectrometer vacuum chamber stay low enough for normal operation. During the past three decades, many of the above barriers have been overcome.
Direct analysis at real time (DART) [1,2] is one of the major ambient ionization processes. DART technology was developed and patented in 2005. Ionization source of DART usually involves a discharge source to produce metastable states of rare gas atoms. Ionization process is often achieved by Penning ionization process. Most analytes analyzed by DART are somewhat small molecules (MW<2000 Da). DART has been extensively used for the analysis for drug [3], forensic [4], chemical [5] and food industries [6]. Since DART is not capable of doing molecules with a large mass, DART is somewhat inconvenient on the application of biomolecular sample analysis such as proteomics and glycomics. An extensive review on DART has been given by Chernetsova et al. [7].
In 1988, Tanaka et al. obtained large biomolecular mass spectra by using nano metal particles assisted laser desorption and aroused lots interest to pursue different methods for ionization of biomolecules [8]. Then, Hillenkamp and his co-workers developed matrix-assisted laser desorption/ionization (MALDI) mass spectrometry (MS) which can rapidly measure the molecular weights of different proteins with a time-of-flight (TOF) mass spectrometer [9]. At about the same time, Fenn et al. developed Electrospray Ionization (ESI) mass spectrometry which also can give soft ionization of proteins [10]. MALDI and ESI became the two major ionization tools for protein analysis. After the discovery of MALDI, AP-MALDI (atmosphere pressure MALDI) was developed and can be considered as one of the atmospheric ionization methods derived from MALDI [11]. Takáts et al. developed desorption electrospray ionization (DESI) which was broadly used for applications in different fields [12]. Many different atmospheric and/or ambient ionization technologies were developed based on the modifications of DESI or APCI. They include gas chromatography electrospray ionization (GC-ESI) [13], fused-droplet electrospray ionization (FDESI) [14], electrospray laser desorption ionization (ELDI) [15], laser ablation electrospray ionization (LA-ESI) [16], laser induced acoustic desorption electrospray ionization (LIAD-ESI) [17], MALDIESI [18], plasma-assisted desorption ionization (PADI) [19], Electrode assisted desorption electrospray ionization (EADESI) [20] and etc. There are many other methods with slightly different approaches. We will only give very brief introductions on the methods mentioned in the above and have no intention to cover all different atmospheric and/or ambient ionization methods in this review. More detailed review on ambient ionization has been given by Shiea and his co-workers [21].
Various Atmospheric Ionization Methods
GC-ESI and LC-ESI
GC/MS has been available for small molecule analysis in a complex sample for an extensive period of time. Neutral molecules separated from GC are introduced into a mass spectrometer for ionization in the vacuum chamber. Typical methods include electron impact ionization, chemical ionization, discharge ionization and etc. For GC-ESI [13], ionization is pursued by ESI with the ionization at one atmosphere environment. Analytes from GC exit entering into ESI plume and react with charged solvent to produce ions. GC-ESI has been extensively used for fatty acid analysis. When GC is replaced by LC, it is LC-ESI which has been extensively used for proteomic analysis lately.
AP-MALDI
AP-MALDI has several advantages compared with vacuum MALDI. The major advantage is the flexibility in handling samples since the ionization is pursued in one atmosphere. For AP-MALDI, ionization is outside of the vacuum chamber so that the ionization and desorption processes are totally decoupled from the mass analyzer. The disadvantage is somewhat low transmission efficiency to lead to low detection sensitivity during the early stage of development. Nevertheless, detection limit reaching femto mole region has been reported [11]. AP-MALDI has been used in mass spectrometry (MS) in a variety of applications including analysis of DNA, RNA, lipid and proteomic samples [22].
Desorption electrospray ionization (DESI)
DESI [12] is a combination of ESI and desorption/ionization processes. Desorption/ionization takes place by a charged droplet impacting onto the sample on surface [23]. Charged droplets are attracted to the surface of the sample holder with a high voltage. After the completion of ionization, the ions travel through air into the atmospheric pressure interface which is closely coupled to the mass spectrometer. DESI is a technique that allows for ambient ionization of a trace sample at atmospheric pressure, with little sample preparation.
DESI was developed by Cooks and his co-works and is by far the most broadly used ambient ionization technique. Its applications include organic and bio molecule detections for the application in food and drug industry. It has also been used for forensic application and explosive detection [24]. DESI was also used for molecular imaging although the space resolution is still somewhat worse than the imaging by MALDI [25]. A commercial unit of DESI without the mass analyzer has also been marketed. (http://www.Prosolia.com).
Electrospray laser desorption ionization (ELDI) [15], Laser ablation electrospray ionization (LA-ESI) [16], LIAD-ESI [17] and MALDIESI [18]
There are several different desorption methods which are used to desorb analytes which are subsequently placed into ESI plume for ionization. ELDI, LA-ESI, LIAD-ESI and MALDIESI are among them. For ELDI, a laser beam is used to desorb analytes on a sample plate [15]. Desorbed analyte molecules are post-ionized by the typical ESI process. Intact proteins can be detected without the need of matrix. Since matrix is not needed, ELDI can be used to detect small molecules as well as biomolecules. ELDI can also be used for molecular imaging [21]. Due to the capability of tight focusing, ELDI often can have better space resolution than that from DESI for molecular imaging.
Similar to ELDI, LA-ESI [16] was developed with an infrared laser to desorb samples mostly through a heating process. The desorbed samples again are ionized in ESI plume. Since a higher laser power is often used for LA-ESI, larger biomolecules are more difficult to be detected. For LIAD-ESI, the sample is usually deposited on a metal foil; the rear side of the foil is then irradiated with a laser. The resulting shockwave causes the sample to be desorbed. The desorbed species then enter into ESI plume to react with charged solvent to form analyte ions.
MALDIESI [18] was also developed for detecting biomolecules. MALDI was typically used to desorb biomolecular samples mixed with matrix; the desorbed analytes were then ionized with ESI. Mass spectra obtained by MALDIESI are typically more similar to the spectra from ESI instead of spectra from MALDI. Since MALDI can be used to detect biomolecules, why bother to combine with ESI? The advantage is large biomoecular ions can be produced with multiple charges so that ions with smaller m/z can be produced. Ions with small m/z have better detection sensitivity than that with high mass-to-charge ratio. For most commercial ion trapping mass analyzers such as ion trap, ion cyclotron resonance (ICR) and orbitrap, the detection range for m/z is usually less than 10,000 so that large biomolecular ions produced by MALDI cannot be detected. MALDIESI also provides the capability to do molecular imaging with good space resolution.
Plasma-assisted desorption ionization (PADI) [19]
Discharge has been used for ionization for an extensive period of time. One of the disadvantages for plasma ionization is the easy fragmentation. In order to achieve soft ionization, low temperature plasma is preferred. Plasmaassisted desorption/ionization (PADI) usually takes the approach of RF to produce low temperature plasma from rare gas at ambient condition [19]. The ionization process can be due to low-energy electron ionization, ion-moelcule interaction and/or Penning ionization. PADI can be used for thermally unstable samples.
Electrode assisted desorption electrospray ionization (EADESI) [20]
EADESI is based on the ionization of molecules on different surfaces by highly charged droplets produced on a sharp-edged high voltage tip and ions produced are introduced into the mass spectrometer through a capillary. EADESI can be applied to various samples including amino acids, peptides, proteins, drugs and human fluids such as urine and blood. EADESI can be used for routine analyses in different fields such as forensic, environmental and material sciences. EADESI interface can be fit to a conventional ion trap mass spectrometer. It can be used for various types of samples with a broad mass range. EADESI can also provide real-time analysis which is very valuable for biomedical applications. Due to the sharp electrode to guide the discharge, the space resolution from EADESI can be better than that from droplet induced desorption.
ESI with various substrates
In addition to DESI, MALDIESI and LIAD-ESI, several other different ESI methods with different materials to hold samples have also been successfully developed. They include paper [26] and wood [27]. These results indicate broad range of materials can be used for holding biological samples such as urine and saliva without the need of chemical processing. The sensitivities are comparable to that from DESI. Chemical contents in a piece of leaf have also been analyzed directly [28]. It is clear that leaf can also be used as substrate for holding biological samples for analysis if needed.
Extractive electrospray ionization (EESI)
EESI was developed for direct analysis of urine, polluted water, milk and etc. EESI uses two sprayers [29]. One is for sample nebulization and the other is for conventional ESI solvent. In principle, samples can be analyzed with each microdroplet.
Matrix assisted inlet ionization (MAII) and Solvent Assisted inlet ionization (SAII) [30,31]
MALDI and ESI are still the most common methods for the analysis of biological molecules. AP-MALDI and ESI can be pursued with atmospheric ionization. Nevertheless, high voltages are needed for both MALDI and ESI. Recently, Trimpin and McEwen found that 3-nitrobenzonitrile (3-NBN) can bring biomolecule analytes into gas-phase in ionic states by just placing samples in vacuum [30,32]. This method is called matrix assisted ionization vacuum (MAIV). If the sample is placed closed to the inlet of AP-MALDI, bio molecular ions can also be observed. It is called as matrix assisted inlet ionization (MAII) [33]. Inlet ionization is also applicable to solution samples. Mass spectra of biomolecules similar to MAII are obtained without voltage by introducing the solution into the inlet orifice of a heated transfer tube of a mass spectrometer. This process is called solvent assisted inlet ionization (SAII) [31]. The detection sensitivity for small proteins by SAII and MAII can reach to femto mole region.
Sonic spray ionization (SSI)
The most well-known high-voltage (HV) free ambient ionization methods is sonic spray ionization (SSI) which was first developed by Hirabayashi and his co-workers [34,35]. In SSI, a solution from a capillary is sprayed with a sonic gas flow. Ions are produced more efficiently at a sonic velocity. A pneumatic nebulizer is typically used to turn the solution into a supersonic spray of small droplets. Ions are formed when the solvent evaporates. Oligonucleotides have been studied with this method. In SSI, charged droplets are produced by the high-velocity gas flow. No ionization is observed when the gas flow rate is less than 1 L/min [34]. Charged droplet formation from SSI was explained based on the non-uniformity of positive and negative ion concentration distributions near the solution surface [35]. Therefore, non-polar compounds such as benzene are not expected to be observed with SSI. Recently, easy ambient sonic- spray ionization (EASI), which combines traditional desorption ionization with SSI, was developed for studying protein folding [36]. It was also used to produce ionization of solid analytes [37-39]. Nevertheless, the ionization mechanism of SSI is still not well known.
In this review article, we will focus more on atmospheric ionization without the need of external high voltage power supply. In the follows, only ultrasound ionization (UI), triboelectric spray ionization (TeSI) and Kevin spray ionization (KeSI) will be discussed in more details.
Ultrasound ionization (UI) [40]
In this approach, ultrasound produced by a simple piezoelectric device is used for soft ionization of biomolecules. Intact ions of proteins, saccharides and fatty acids can be obtained and spectra showed little fragmentation. Cavitation may be a primary mechanism for the ionization of biomolecules. This method doesn’t need any high voltage or laser for ionization. Up to now, there have been very few reports on the use of ultrasound for ionization in mass spectrometry applications. Ultrasound has only been used to eject charged droplets from micromachined array devices for mass spectrometry applications [41,42].
Ultrasound has been successfully used in various industrial applications, such as sound navigation and ranging (SONAR), ultrasound cleaning and ultrasound-induced chemical reactions. Many of these applications involve the process of cavitation. When the bubble collapses, the gas within the bubble dissipates into the surrounding liquid to release a significant amount of energy. Therefore, chemical reactions and even ionization can occur by cavitation. Cavitationinduced polymerization have been reported [43,44]. Therefore, the phenomenon of cavitation-induced chemical reactions has been established. In 2005, Flannigan and Suslick [45] observed atomic emission and ionic O2+. Flint and Suslick [46] estimated the temperature of cavitation of silicone oil as ~5000 K. It appears that ionization by cavitation can definitely occur.
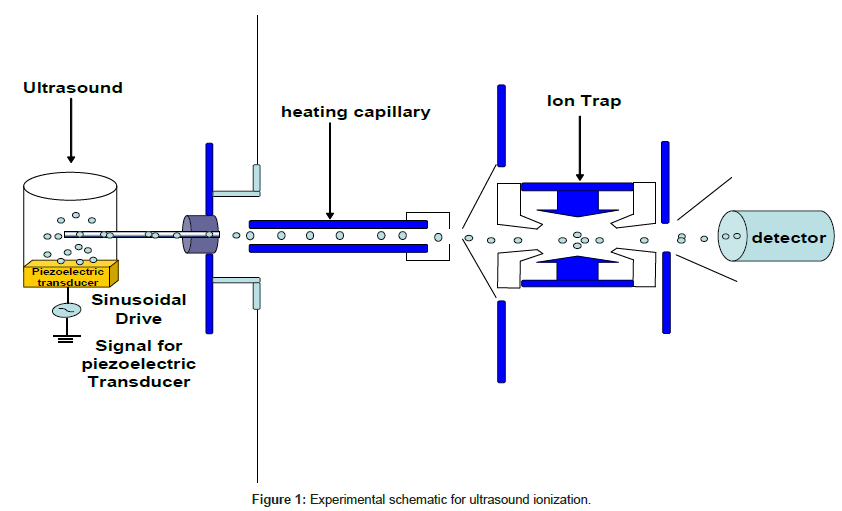
The typical ultrasound power used for ionization was 4 W or higher. The frequency was at 1.5~2 MHz. Solution samples were directly placed on the surface of the piezoelectric device. The desorbed small droplets were introduced into an ion trap mass spectrometer for analysis. The schematic of the device is shown in Figure 1. No high voltages on the tip of the capillary were applied. No ionization signals were observed prior to a high rate of cavitation, which was visible to the naked eye.
Ultrasound ionization (UI) of biomolecules was successfully demonstrated [40]. In general, most observed ions are singly charged. Therefore, the patterns of mass spectra by UI are more similar to those from MALDI. It indicates the ionization is more likely through ion molecule interactions between biomolecule and small molecular ions produced by cavitation. Mass spectra of oligosaccharide such as Man8 with UI were successfully measured when matrix was added [40]. The result indicates the ionization may be due to the protonation during the cavitation process. In general, much more alkali-attached oligosaccharide ions are produced than protonated oligosaccharides by either MALDI or ESI. With UI, most ions produced are protonated oligosaccharide ions that make spectra analysis much simpler.
Mass spectra of angiotensin in various solutions produced by UI were also observed [40]. Signals for biomolecular ions are increased by ~300% when a mixture of acetone and water was used instead of a pure water solvent. Since the density and viscosity of acetone are lower than those of water, it is easier for cavitation to occur in acetone than in water [47]. In addition, biomolecule protonation requires significantly more energy in water than in acetone [48]. Therefore, more ionization can be achieved and more protonated biomolecular ions can be observed with the addition of acetone.
Since ionization of small molecules has been observed in several different cavitation experiments and data not consistent with SSI, cavitation can be a major mechanism for producing biomolecular ions. A model for ultrasonic ionization of biomolecules consists of following major steps: (1) Biomolecules are included in the bubble during the bubble formation process. (2) Most energy released from the bubble burst lead to ionization. (3) Protonation and/or deprotonation can occur between water and biomolecules to produce biomolecular ions. Since the amount of water molecules is at least six orders of magnitude more than the amount of the impurity level of alkali metal ions, more protonation than alkalization can be expected. Therefore, ultrasound ionization is just like the MALDI inside of a bubble. Most biomolecular ions produced are singly charged instead of multiply charged.
The detection sensitivity of ultrasound ionization is comparable to that of desorption electrospray ionization (DESI). Since UI can be directly applied to liquid samples, there are no matrix effects due to crystallization, such as sweet spots in MALDI. On the other hand, some organic compounds that have been used as matrices for MALDI were observed to enhance ionization with ultrasound-induced ionization. The effect of impurities such as salts was found less important than in ESI. Few sodium-attached polysaccharide ions were detected, even after adding tenfold more NaCl than polysaccharide into the sample. The major disadvantage of UI is primarily on the difficulty to control cavitation so that quantitative determination becomes difficult.
Triboelectric spray ionization (TeSI) [49]
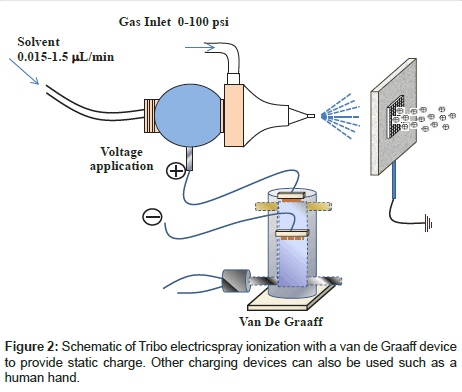
The major difference between ESI and TeSI is high voltage power supply. For ESI, a potential difference must be applied between the spray capillary and the counter electrode. This potential difference initiates the electro-hydrodynamic processes that create the small charged droplets. In contrast to the DC electrospray, another type of ionization method was developed by using an AC electric field [50-52]. TeSI is introduced by using static electricity which is readily available. Experimental scheme is shown in Figure 2. In general, it includes a capillary, a nebulizer and a high voltage connection. There is a metal screen in front of the spray part. Source can be powered by a van de Graaff [53] that can produce either positive or negative static charges. For positive ions, positive charges are applied to the spray side and metal screen is grounded, or negative charges are applied to metal screen and spray is grounded. For the negative ion spectra, the arrangement is just opposite to the above polarity. This source can also work without a van de Graaff device. In this case, spray component is grounded and screen is charged up by friction or contact to other materials such as polymers. This process is known as triboelectric effect or triboelectric charging. This design only needs a potential difference between spray and screen and ion formation can be observed. When the system is powered by a van de Graaff device, signal intensity is comparable with that from ESI.
For TeSI, mass spectra can be obtained with any mass analyzer. Nevertheless, a quadrupole ion trap (QIT) mass spectrometer is convenient for detection. Spray emitter was oriented axially to the entrance of the mass spectrometer. Nitrogen gas was used as a nebulizing gas at a pressure of 0-60 psi. The flow rate of solvent can be lower than 1 μL/min. The voltage induced by static charge was typically between 0.5-4 kV. Figure 3 shows comparison of TeSI and ESI spray profiles. In both cases the spray plums are similar and the size of the liquid droplets apparently became smaller than those observed without the DC voltage or static electricity.
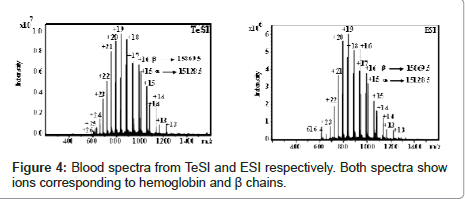
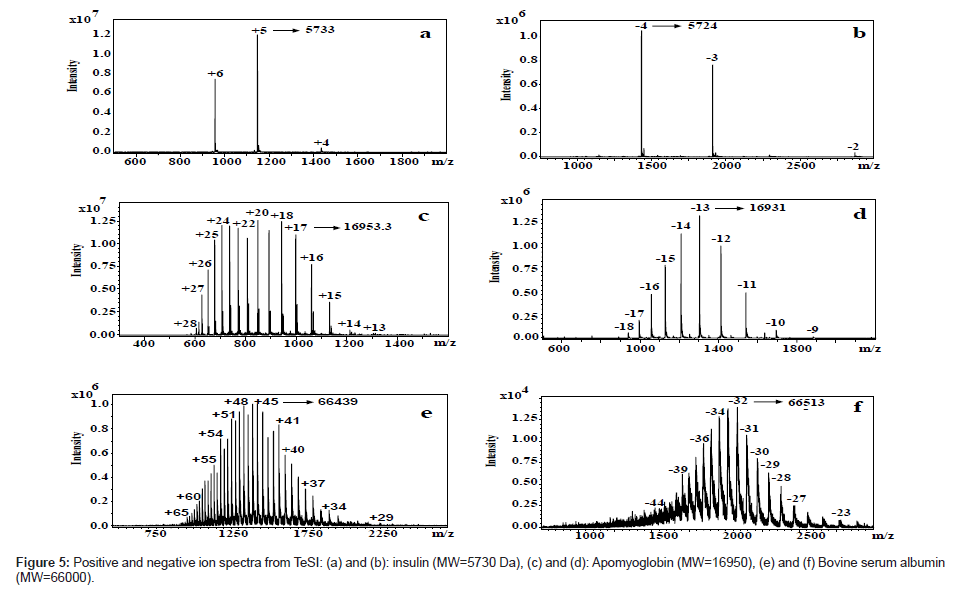
Figure 4 shows a side-by-side comparison of positive ion mode spectra using TeSI and ESI for blood samples. Both results are comparable. Figure 5 shows both positive and negative ion spectra for proteins from 5.7 to 66 KDa with TeSI.
TeSI has some advantages over other atmospheric ionization methods. TeSI is robust and provides reproducible results. TeSI is a very human friendly source. Even holding in hand TeSI can be used without the concern of high voltage shock. By changing the parameters, it is possible to study the non-covalent interactions and observe kinetics of protein folding with TeSI.
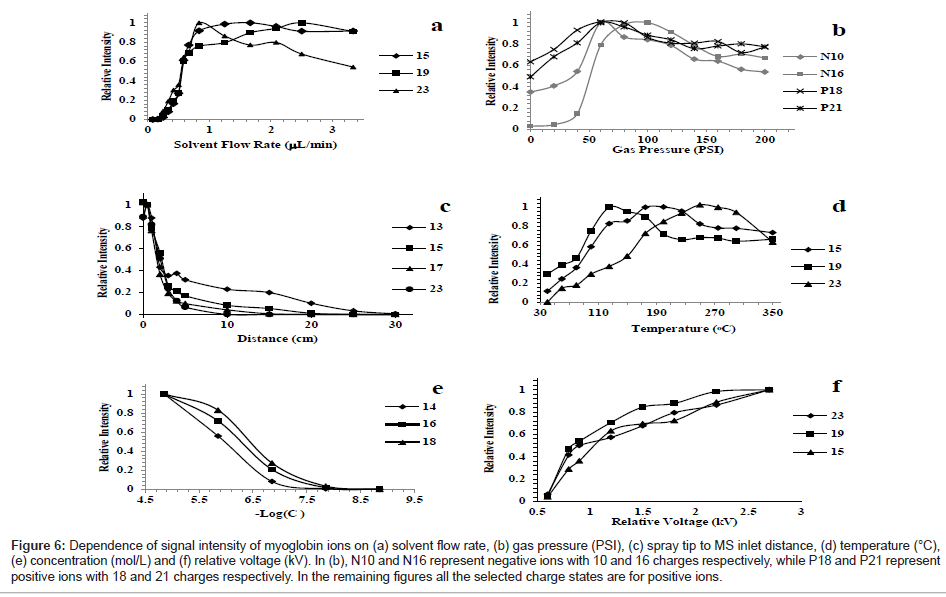
For TeSI, gas flow rate, capillary temperature, solution acidity and static charge have strong relationship with the signal intensity. Figure 6a shows the effect of solvent flow rate on the signal intensity. The experimental results show that TeSI does not require high solvent flow rate which is very convenient for biomedical experiments. The typical flow rate needed is 0.15 ~ 1.5 μl/min.
Figure 6: Dependence of signal intensity of myoglobin ions on (a) solvent flow rate, (b) gas pressure (PSI), (c) spray tip to MS inlet distance, (d) temperature (°C), (e) concentration (mol/L) and (f) relative voltage (kV). In (b), N10 and N16 represent negative ions with 10 and 16 charges respectively, while P18 and P21 represent positive ions with 18 and 21 charges respectively. In the remaining figures all the selected charge states are for positive ions.
The influence of nebulizing gas pressure to the peak intensities of multiply charged ions of myoglobin was studied (Figure 6b). Nebulizing gas and distance effects have opposite effect on the signal intensity. The shorter the distance between spray and MS, the lower pressure is required. Figure 6c shows the distance dependent signal intensity and charge state distribution. Protein solutions can be sprayed from 30 cm away from entrance of MS, with a signal-to-noise (S/N) of around 50. The relationship of transfer capillary temperature to the signal of myoglobin is shown in Figure 6d. The charge state distribution of ions did not change throughout the temperature studies. Figure 6e shows the concentration dependency of signal of myoglobin ion for three distinct charge states. Figure 6f shows the voltage versus signal intensity for three different charge states of myoglobin.
Another important two parameters in spray ionization process are pH in solution and composition of solvents. These two parameters change the charged state distribution of multiple charged ions and conformational structures. Changes in solvent composition by adding organic solvents or acidic solution can induce the unfolding or precipitation of proteins from solution.
The major advantage of TeSI is very soft ionization and the disadvantage is the difficulty to control static charge at a constant level when no external power supply is used.
Kelvin spray ionization (KeSI) [54]
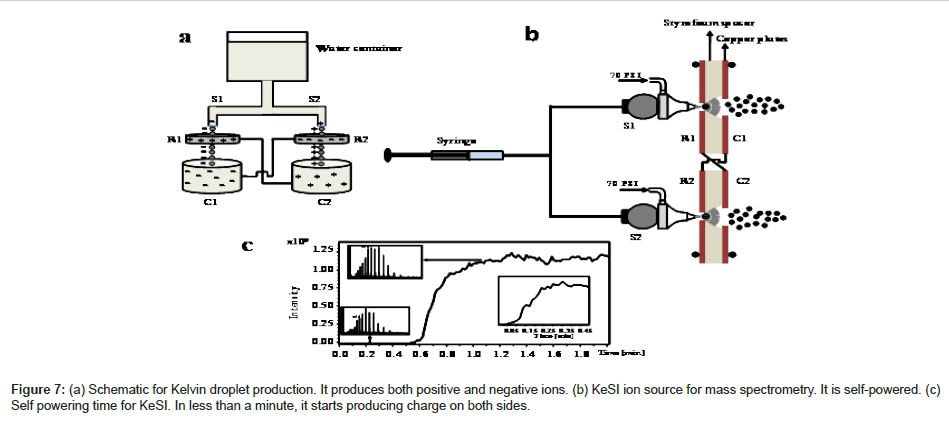
Sonic spray ionization (SSI) is a soft ionization since SSI does not use any power supply. Nevertheless, the detection sensitivity of SSI is often lower than that from ESI and very high gas flow is needed. A new approach without the need of external high voltage and to achieve high detection sensitivity is called Kelvin spray ionization (KeSI). The idea behind this ionization source comes from Kelvin water dropper. Experimental schematic of KeSI is shown in Figure 7. The facility consists of two homemade identical spray sources, two double layered copper plates in front of them and a syringe. The sprays consist of stainless steel unions including a capillary which extends 1 mm beyond the edge of the stainless steel fittings. The spray component can be moved with a XYZ translational stage. Two double layered copper plates are placed in front of the sprays. Double layered copper plates have a small hole on both spray side and the entrance of the mass spectrometer. Copper plates are isolated using polystyrene wafer between them. The locations of copper plates can be adjusted with a XYZ translation assembly. The spray solvent is provided using a syringe pump. Mass spectra can be obtained with a quadrupole ion trap (QIT) mass analyzer. Nitrogen gas is used as a nebulizing gas. The flow rate range is typically at 0.15 -2.5 μL/min. Potential difference can be created without any external high voltage power supplies.
If the main purpose of ionization is to ionize biomolecules and direct them into the mass spectrometer by keeping the structures same as in solution, the amount of internal energy that will be transferred to the ionized molecules should be controlled to the minimum. The applied high voltage can distort the conformational structures especially for non-covalent complex such as protein dimers. One indicator for structural change is the change of charge state distribution in mass spectra. The charge-state distribution is frequently used as an analytical tool for determining the degree of unfolding of proteins [55-57]. Narrow distribution with the peak at low charge number indicates native-like folded structures and distributions centered around high charge state shows unfolded structures.
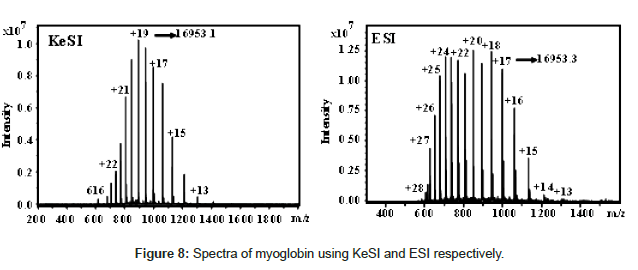
The idea of KeSI is simple and both positive and negative ions can be obtained simultaneously. Figure 8 shows a side-by-side comparison of positive mode mass spectra of myoglobin with KeSI and ESI. It is clear that the charge state distribution is lower in KeSI spectra. This implies the softer ionization of KeSI than that of ESI. KeSI can be achieved by external static electricity or self-powered.
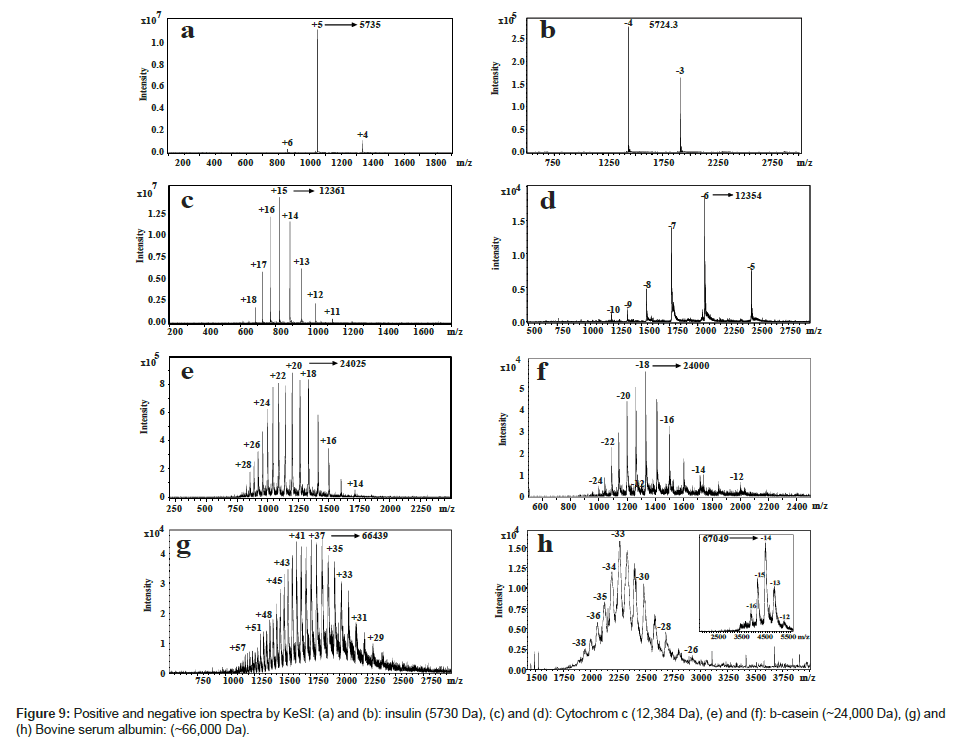
Mass spectra from KeSI for various sizes of proteins are shown in Figure 9. Signal intensity of KeSI is similar to ESI for small proteins like myoglobin, cytochrome c and casein. For big proteins such as albumin, the observed charge state distribution of KeSI is lower than ESI in the same m/z region. In this region, ESI does not give a well-resolved mass spectrum, indicates that KeSI is a softer ionization technique. To obtain similar spectra for big proteins, KeSI needs higher gas flow and solution flow to increase the voltage on copper plates.
The degree of a soft ionization is basically defined how successful an ionization method to carry the solution phase structures into the gas phase [58-61]. The main reason for this difference is the internal energy transferred to the molecules during the ionization. If the amount of internal energy transferred is low, the probability of dissociation will be less and the probability of the structure in ionic state in gas phase is more similar to the structure in solution phase. In KeSI, it is possible to decrease the transferred internal energy to minimum by decreasing the gas pressure or decreasing the solvent flow rate. Once solution droplets are induced by first copper plate, droplets become oppositely charged. This property makes KeSI a preferable method to investigate the noncovalent biological complex. Formation of gaseous ions from droplets in ESI is generally assumed to follow either the charge residue model [62-65] or the ion evaporation model depending mostly on the mass of the ion. Since the charge state distribution of ions is lower in KeSI than that in ESI, softer ionization mechanism is more likely. Observation of albumin negative ions supports this idea. ESI cannot produce the same spectra for albumin.
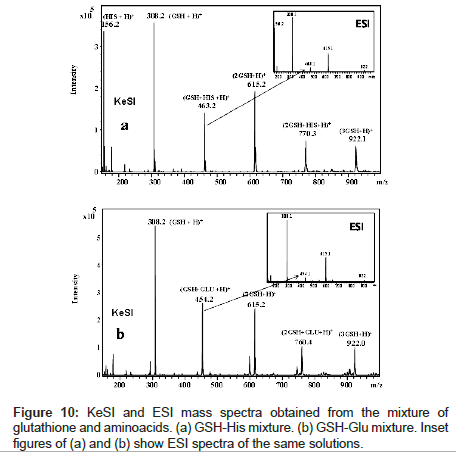
Figure 10 shows the glutathione (GSH)-histidine (HIS) and GSHL- glutamate (GLU) complex formations. For comparison, ESI results are also shown in the same figure as an inset for both GSH-amino acid complexes. In the comparison of complex ion peaks, KeSI gives higher peak intensity. Also peak intensities of dimer and trimer of GSH are higher than the results obtained by ESI. MS-MS experiments have been done to confirm the non-covalent interactions between GSH and an amino acid molecule.
KeSI is a very soft ionization and can become a powerful tool for studying non-convalent bonding between biomolecules which is critical to understand the disease mechanism and drug development. The disadvantage of KeSI is somewhat a more complex structure for the facility and it is difficult to predict the locations for each type of ions (positive or negative ions) to be produced.
Conclusion
UI, MAII, SAII, TeSI and KeSI are newly developed atmospheric soft ionization methods. They share one common advantage that no external HV power supply is absolutely required. There are no needs for a laser beam so that the device is cheaper. UI can be easily pursued under the environment of most biomedical experiments so that in situ and real-time analysis can become feasible. MAII and SAII have the advantages of simplicity and high sensitivity. MAII is expected to have similar advantages of measuring non-covalent bonding complex although detailed data have not been reported. TeSI and KeSI have the softer ionization than ESI so that biomolecule complex with noncovalent bonding can be studied. Although TeSI and KeSI are among the softest ambient ionization methods, there is no need to worry about electrical shock during operation.
Acknowledgement
This work was supported by the Genomics Research Center, Academia Sinica, the National Science Council of Taiwan (NSC 99-2113-M-001-002-MY3; NSC 101-2325-B-001-001) and the National Health Research Institute in Taiwan (Grant NHRI-EX101-9803EI). NGC likes to acknowledge the support provided by National Chiao Tung University and AO wants to acknowledge the support by Sakarya University.
References
- Cody RB, Laramée JA, Durst HD (2005) Versatile new ion source for the analysis of materials in open air under ambient conditions. Anal Chem 77: 2297-2302.
- Laramee JA, Cody RB (2006) Method for atmospheric pressure analyte ionization. U.S. Patent US7112785 B2.
- Yu S, Crawford E, Tice J, Musselman B, Wu JT (2009) Bioanalysis without sample cleanup or chromatography: the evaluation and initial implementation of direct analysis in real time ionization mass spectrometry for the quantification of drugs in biological matrixes. Anal Chem 81: 193-202.
- Nilles JM, Connell TR, Durst HD (2009) Quantitation of chemical warfare agents using the direct analysis in real time (DART) technique. Anal Chem 81: 6744-6749.
- Maleknia SD, Bell TL, Adams MA (2009) Eucalypt smoke and wildfires: temperature dependent emissions of biogenic volatile organic compounds. Int J Mass Spectrom279: 126-133.
- Haefliger OP, Jeckelmann N (2007) Direct mass spectrometric analysis of flavors and fragrances in real applications using DART. Rapid Commun Mass Spectrom 21: 1361-1366.
- Chernetsova ES, Morlock GE, Revelsky IA (2011) DART mass spectrometry and its applications in chemical analysis. Russian Chemical Reviews 80: 235-255.
- Tanaka K, Yaki H, Ido Y, Akida S, Yoshida Y, et al. (1988) Protein and polymer analyses up to m/z 100,000 by laser ionization time-of-flight mass spectrometry. Rap Comm Mass Spectrom 2: 151-153.
- Karas M, Hillenkamp F (1988) Laser desorption ionization of proteins with molecular masses exceeding 10,000 daltons. Anal Chem 60: 2299-2301.
- Fenn JB, Mann M, Meng CK, Wong SF, Whitehouse CM (1989) Electrospray ionization for mass spectrometry of large biomolecules. Science 246: 64-71.
- Laiko VV, Baldwin MA, Burlingame AL (2000) Atmospheric pressure matrix-assisted laser desorption/ionization mass spectrometry. Anal Chem 72: 652-657.
- Takáts Z, Wiseman JM, Gologan B, Cooks RG (2004) Mass spectrometry sampling under ambient conditions with desorption electrospray ionization. Science 306: 471-473.
- Lee CY, Shiea J (1998) Gas chromatography connected to multiple channel electrospray ionization mass spectrometry for the detection of volatile organic compounds. Anal Chem 70: 2757-2761.
- Chang DY, Lee CC, Shiea J (2002) Detecting large biomolecules from high-salt solutions by fused-droplet electrospray ionization mass spectrometry. Anal Chem 74: 2465-2469.
- Huang MZ, Hsu HJ, Lee JY, Jeng J, Shiea J (2006) Direct protein detection from biological media through electrospray-assisted laser desorption ionization/mass spectrometry. J Proteome Res 5: 1107-1116.
- Nemes P, Vertes A (2007) Laser ablation electrospray ionization for atmospheric pressure, in vivo, and imaging mass spectrometry. Anal Chem 79: 8098-8106.
- Cheng SC, Cheng TL, Chang HC, Shiea J (2009) Using laser-induced acoustic desorption/electrospray ionization mass spectrometry to characterize small organic and large biological compounds in the solid state and in solution under ambient conditions. Anal Chem 81: 868-874.
- Sampson JS, Hawkridge AM, Muddiman DC (2006) Generation and detection of multiply-charged peptides and proteins by matrix-assisted laser desorption electrospray ionization (MALDESI) Fourier transform ion cyclotron resonance mass spectrometry. J Am Soc Mass Spectrom 17: 1712-1716.
- Harper JD, Charipar NA, Mulligan CC, Zhang X, Cooks RG, et al. (2008) Low-temperature plasma probe for ambient desorption ionization. Anal Chem 80: 9097-9104.
- Özdemir A, Chen CH (2010) Electrode-assisted desorption electrospray ionization mass spectrometry. J Mass Spectrom 45: 1203-1211.
- Huang MZ, Yuan CH, Cheng SC, Cho YT, Shiea J (2010) Ambient ionization mass spectrometry. Annu Rev Anal Chem (Palo Alto Calif) 3: 43-65.
- Laiko VV, Moyer SC, Cotter RJ (2000) Atmospheric pressure MALDI/ion trap mass spectrometry. Anal Chem 72: 5239-5243.
- Takáts Z, Wiseman JM, Cooks RG (2005) Ambient mass spectrometry using desorption electrospray ionization (DESI): instrumentation, mechanisms and applications in forensics, chemistry, and biology. J Mass Spectrom 40: 1261-1275.
- Ifa DR, Jackson AU, Paglia G, Cooks RG (2009) Forensic applications of ambient ionization mass spectrometry. Anal Bioanal Chem 394: 1995-2008.
- Gode D, Volmer DA (2013) Lipid imaging by mass spectrometry - a review. Analyst 138: 1289-1315.
- Wang H, Liu J, Cooks RG, Ouyang Z (2010) Paper spray for direct analysis of complex mixtures using mass spectrometry. Angew Chem Int Ed Engl 49: 877-880.
- Hu B, So PK, Chen H, Yao ZP (2011) Electrospray ionization using wooden tips. Anal Chem 83: 8201-8207.
- Liu J, Wang H, Cooks RG, Ouyang Z (2011) Leaf spray: direct chemical analysis of plant material and living plants by mass spectrometry. Anal Chem 83: 7608-7613.
- Chen H, Venter A, Cooks RG (2006) Extractive electrospray ionization for direct analysis of undiluted urine, milk and other complex mixtures without sample preparation. Chem Commun (Camb): 2042-2044.
- Chakrabarty S, Pagnotti VS, Inutan ED, Trimpin S, McEwen CN (2013) A new matrix assisted ionization method for the analysis of volatile and nonvolatile compounds by atmospheric probe mass spectrometry. J Am Soc Mass Spectrom 24: 1102-1107.
- Pagnotti VS, Chubatyi ND, McEwen CN (2011) Solvent assisted inlet ionization: an ultrasensitive new liquid introduction ionization method for mass spectrometry. Anal Chem 83: 3981-3985.
- Inutan ED, Trimpin S (2013) Matrix assisted ionization vacuum (MAIV), a new ionization method for biological materials analysis using mass spectrometry. Mol Cell Proteomics 12: 792-796.
- McEwen CN, Pagnotti VS, Inutan ED, Trimpin S (2010) New paradigm in ionization: multiply charged ion formation from a solid matrix without a laser or voltage. Anal Chem 82: 9164-9168.
- Hirabayashi A, Sakairi M, Koizumi H (1995) Sonic spray mass spectrometry. Anal Chem 67: 2878-2882.
- Huang M, Hirabayashi A, Okumura A, Hirabayashi Y (2001) Matrix effect on the analysis of oligonucleotides by using a mass spectrometer with a sonic spray ionization source. Anal Sci 17: 1179-1182.
- Haddad R, Sparrapan R, Eberlin MN (2006) Desorption sonic spray ionization for (high) voltage-free ambient mass spectrometry. Rapid Commun Mass Spectrom 20: 2901-2905.
- Haddad R, Sparrapan R, Kotiaho T, Eberlin MN (2008) Easy ambient sonic-spray ionization-membrane interface mass spectrometry for direct analysis of solution constituents. Anal Chem 80: 898-903.
- Haddad R, Milagre HM, Catharino RR, Eberlin MN (2008) Easy ambient sonic-spray ionization mass spectrometry combined with thin-layer chromatography. Anal Chem 80: 2744-2750.
- Santos VG, Regiani T, Dias FF, Romão W, Jara JL, et al. (2011) Venturi easy ambient sonic-spray ionization. Anal Chem 83: 1375-1380.
- Wu CI, Wang YS, Chen NG, Wu CY, Chen CH (2010) Ultrasound ionization of biomolecules. Rapid Commun Mass Spectrom 24: 2569-2574.
- Aderogba S, Meacham JM, Degertekin FL, Fedorov AG, Femandez FM (2005) Nanoelectrospray ion generation for high-throughput mass spectrometry using a micromachined ultrasonic ejector array. Appl Phys Lett 86: 203110-203113.
- Hampton CY, Forbes TP, Varady MV, Meacham JM, Fedorov AG, et al. (2007) Analytical Performance of a Venturi-Assisted Array of Micromachined Ultrasonic Electrosprays Coupled to Ion Trap Mass Spectrometry for the Analysis of Peptides and Proteins. Anal Chem 79: 8154-8161.
- Diedrich GK, Kruus P and Rachlis LM (1972) Cavitation-induced Reactions in Pure Substituted Benzenes. Can J Chem 50: 1743-1750.
- Kuijpers MW, van Eck D, Kemmere MF, Keurentjes JT (2002) Cavitation-induced reactions in high-pressure carbon dioxide. Science 298: 1969-1971.
- Flannigan DJ, Suslick KS (2005) Plasma formation and temperature measurement during single-bubble cavitation. Nature 434: 52-55.
- Flint EB, Suslick KS (1991) The temperature of cavitation. Science 253: 1397-1399.
- Didenko YT, Suslick KS (2002) The energy efficiency of formation of photons, radicals and ions during single-bubble cavitation. Nature 418: 394-397.
- Ngyuen MT, Chandra AK, Zeegers-Huyskens T (1998) Protonation and deprotonation energies of uracil - Implications for the uracil-water complex. J Chem Soc Faraday Trans 94: 1277-1280.
- Özdemir A, Lin JL, Gillig KJ, Chen CH (2013) Triboelectric spray ionization. J Mass Spectrom 48: 154-163.
- Chetwani N, Cassou CA, Go DB, Chang HC (2011) Frequency dependence of alternating current electrospray ionization mass spectrometry. Anal Chem 83: 3017-3023.
- Chetwani N, Cassou CA, Go DB, Chang HC (2010) High-frequency AC electrospray ionization source for mass spectrometry of biomolecules. J Am Soc Mass Spectrom 21: 1852-1856.
- Nemes P, Goyal S, Vertes A (2008) Conformational and noncovalent complexation changes in proteins during electrospray ionization. Anal Chem 80: 387-395.
- Schnier PD, Price WD, Williams ER (1996) Modeling the maximum charge state of arginine-containing Peptide ions formed by electrospray ionization. J Am Soc Mass Spectrom 7: 972-976.
- Ozdemir A, Lin JL, Gillig KJ, Chen CH (2013) Kelvin spray ionization. Analyst 138: 6913-6923.
- Pagnotti VS, Inutan ED, Marshall DD, McEwen CN, Trimpin S (2011) Inlet ionization: a new highly sensitive approach for liquid chromatography/mass spectrometry of small and large molecules. Anal Chem 83: 7591-7594.
- Wang GD, Cole RB (1994) Effect of Solution Ionic Strength on Analyte Charge State Distributions in Positive and Negative Ion Electrospray Mass Spectrometry. Anal Chem 66: 3702-3708.
- Verkerk UH, Peschke M, Kebarle P (2003) Effect of buffer cations and of H3O+ on the charge states of native proteins. Significance to determinations of stability constants of protein complexes. J Mass Spectrom 38: 618-631.
- Fligge TA, Przybylski M, Quinn JP, Marshall AG (1998) Evaluation of heat-induced conformational changes in proteins by nanoelectrospray Fourier transform ion cyclotron resonance mass spectrometry. Eur Mass Spectrom 4: 401-404.
- Katta V, Chowdhury SK, Chait BT (1990) Electrospray ionization: A new tool for the analysis of ionic transition-metal complexes. J Am Chem Soc 112: 5348-5349.
- Ganguly AK, Pramanik BN, Tsarbopoulos A, Covey TR, Huang E, et al. (1992) Mass spectrometric detection of the noncovalent GDP-bound conformational state of the human H-ras protein. J Am Chem Soc 114: 6559-6560.
- Lightwahl KJ, Schwartz BL, Smith RD (1994) Observation of the Noncovalent Quaternary Associations of Proteins by Electrospray Ionization Mass Spectrometry. J Am Chem Soc 116: 5271-5278.
- Dole M, Mack LL, Hines RL, Mobley RC, Ferguson LD, et al. (1968) Molecular beams of macroions. J Chem Phys 49: 2240-2249
- Dole M, Hines RL, Mack LL, Mobley RC, Ferguson LD, et al. (1968) Gas Phase Macroions. Macromolecules 1: 96-97.
- Kebarle P, Tang L (1993) From ions in solution to ions in the gas phase. Anal Chem 65: 972A-986A.
- Smith JW, Siegel EE, Maze JT, Jarrold MF (2011) Image charge detection mass spectrometry: pushing the envelope with sensitivity and accuracy. Anal Chem 83: 950-956.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 15786
- [From(publication date):
specialissue-2014 - Dec 01, 2025] - Breakdown by view type
- HTML page views : 10999
- PDF downloads : 4787