Research Article Open Access
Rapid Analysis of Cocaine in Saliva by Surface-Enhanced Raman Spectroscopy
Kathryn Dana, Chetan Shende, Hermes Huang and Stuart Farquharson*
Real-Time Analyzers Inc., 362 Industrial Park Road, Unit 8, Middletown, CT 06457, USA
- *Corresponding Author:
- Stuart Farquharson
Real-Time Analyzers Inc., 362
Industrial Park Road, Unit 8
Middletown, CT 06457, USA
Tel: 860-635-9800-230
E-mail: stu@rta.biz
Received date: September 30, 2015 Accepted date: October 29, 2015 Published date: November 05, 2015
Citation: Dana K, Shende C, Huang H, Farquharson S (2015) Rapid Analysis of Cocaine in Saliva by Surface-Enhanced Raman Spectroscopy. J Anal Bioanal Tech 6:289. doi:10.4172/2155-9872.1000289
Copyright: © 2015 Dana K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Analytical & Bioanalytical Techniques
Abstract
Increases in illicit drug use and the number of emergency-room visits attributable to drug misuse or abuse highlight the need for an efficient, reliable method to detect drugs in patients in order to provide rapid and appropriate care. A surface-enhanced Raman spectroscopy (SERS)-based method was successfully developed to rapidly measure cocaine in saliva at clinical concentrations, as low as 25 ng/mL. Pretreatment steps comprising chemical separation, physical separation, and solid-phase extraction were investigated to recover the analyte drug from the saliva matrix. Samples were analyzed using Fourier-transform (FT) and dispersive Raman systems, and statistical analysis of the results shows that the method is both reliable and accurate, and could be used to quantify unknown samples. The procedure requires minimal space and equipment and can be completed in less than 16 minutes. Finally, due to the inclusion of a buffer solution and the use of multiple robust pretreatment steps, with minimal further development this method could also be applied to other drugs of interest.
Keywords
Raman; SERS; Cocaine; Drug detection
Introduction
Illicit drug use is a growing problem in the United States, with associated detrimental health effects to users that may cause them to seek urgent care. The number of recent users increased from 8.3 percent in 2002 to 9.4 percent in 2013, an estimated 24.6 million people [1]. This figure includes 1.5 million cocaine users. Within approximately the same period, the number of annual emergency-room visits attributable to drug misuse or abuse rose by 52% (between 2004 and 2011), and cocaine was detected in 40.3% of all illicit-drug-related admissions [2]. In order to accurately diagnose and treat patients in emergency room situations, it is critical to identify the cause of admission, e.g., the type of drug present in the patient’s system. The symptoms of illicit drug abuse are often similar and can sometimes be misdiagnosed as physical or psychiatric disorders [3]. In particular, cocaine may cause arrhythmias, myocardial infarction, hypothermia, seizures, and/or hallucinations [4]. Current serum and urine toxicological screening methods can be inconvenient and time-consuming, requiring the collection of samples from potentially uncooperative patients. Thus, there is a need for a rapid and minimally invasive analytical technique to accurately detect illicit substances in emergency room patients. To reduce the invasive nature of sample collection, saliva may be used in lieu of blood or urine. Typically, drugs are represented in saliva at concentrations similar to blood plasma [5,6], while saliva is characterized by better sample integrity than urine and can contain both the parent compound and metabolites [7]. Furthermore, saliva is 99.5% water, making it easy to chemically analyze [8]; and simple saliva collectors are readily commercially available.
In the case of cocaine, previous studies have shown that cocaine and metabolite concentrations in saliva are significantly correlated with blood plasma concentrations and that the metabolite-to-parent ratio detected in oral fluid can potentially be used as an indicator of the time of last use [9,10]. In one study, cocaine was detected in saliva at concentrations 4.9 times higher than in urine and serum, and all saliva samples yielded true positives for cocaine while some matched urine and serum samples had undetectable concentrations of the drug [11].
The current accepted cut-off threshold for the detection of most drugs in saliva falls within the 10-50 ppb (10-50 ng/mL) range, as defined by the U.S. Substance Abuse and Mental Health Services Administration (SAMHSA). For clinical applications, a device that can reliably measure these concentrations in just a few minutes, without false positives or negatives, is desirable [12]. To this end, we have been investigating the potential of surface-enhanced Raman spectroscopy (SERS) to both identify and quantify drugs of interest and their metabolites in an oral fluid matrix [13-15].
In this study, a rapid SERS-based method was developed to measure cocaine in saliva. Various sol-gel chemistries, doped with silver or gold as the SERS-active medium, were investigated. A solidphase extraction (SPE) pretreatment procedure, including steps to free cocaine from the saliva matrix and concentrate samples prior to measurement, was also developed. Clinically relevant concentrations of cocaine were measured in saliva, and the results were subjected to statistical analysis.
Materials and Methods
SERS-active sol-gel capillaries
Gold and silver sol-gels were prepared according to previously published procedures [16,17], with some modifications. Briefly, a metal-ligand precursor (e.g., silver amine or gold chloride) was mixed with a Si-alkoxide precursor (tetramethyl orthosilicate or methyltrimethoxysilane) in methanol. The SERS-active capillaries were prepared by drawing ~20 μL of the resulting gold- or silverdoped sol-gel into a 10 cm long, 1 mm diameter glass capillary (VWR International, Bridgeport, NJ, USA) to form a ~1 cm plug. The plug was allowed to gel and cure, after which the incorporated gold or silver ions were reduced by flowing 5 mL of 0.1M NaBH4 through the capillary. A sample photograph of a reduced gold sol-gel capillary is included in the Supporting Information (Figure S1).
Sample pretreatment
SPE cartridges were obtained from United Chemical Technologies (UCT C8+SCX Clean Screen SPE columns; Bristol, PA, USA). All solvents used during the extraction experiments were obtained from Sigma Aldrich (Allentown, PA, USA). For the SPE pretreatment procedure, the column was first conditioned with 1.3 mL of methanol, followed by 1.3 mL of water. Then, 1-2 mL of sample was flowed through the SPE cartridge at a rate of 1 mL/min using a GenieTouch syringe pump (Kent Scientific, Torrington, CT, USA). The column was then dried by flowing air through it for 1 minute using the syringe pump in draw mode, after which analytes were eluted off the column using 100 μL of 50/50 (v/v) hexane/ethyl acetate at a flow rate of 1 mL/ min. Finally, 25 μL of the collected eluate was loaded onto a SERSactive capillary and measured using SERS. The recovery efficiency of the SPE method was confirmed by gas chromatography (GC) analysis of samples with known concentration before and after extraction.
For physical separation using centrifugation, a Galaxy Mini centrifuge (VWR, Radnor, PA, USA) was employed for 5 minutes at 6000 rpm. For filtration steps, a 0.2-micron filter (Grace Davison Discovery Science, Deerfiled, IL, USA) was connected to a Luer-Lok plastic syringe (Becton-Dickinson, Franklin Lakes, NJ, USA), and the sample was passed manually through the filter.
Saliva samples
Saliva sample controls were collected from volunteers using an oral swab (Medimpex United Inc., PA, USA). The swabs were wiped along all inner mouth surfaces for 1 minute, collecting ~1 mL of saliva. Samples were collected throughout the day, then combined and stored in a freezer until use. Varying amounts of a standard cocaine solution were added to saliva samples to yield artificially spiked samples at a desired range of concentrations. These samples were loaded directly onto SERS-active capillaries for SERS measurement.
The SPE pretreatment method described above was modified for saliva samples as follows: A 0.5 mL saliva sample was collected using a swab and transferred to a centrifuge tube containing 0.5 mL of 0.1M pH 6 acetate buffer. A C8+SCX SPE cartridge was preconditioned by sequentially drawing 1 mL each of methanol and 0.1M acetate buffer through it at 2 mL/min. Then, the 1 mL buffered sample was drawn through the column at 1 mL/min. The column was washed by sequentially drawing 1 mL each of water, 0.1M HCl, and methanol through it at 2 mL/min, and then dried by drawing air through it for 1 min. The analyte was eluted from the column using 1 mL of 2% NH4OH in methanol at a flow rate of 0.5 mL/min. All fluid flow through the SPE cartridge was accomplished using a syringe. The eluate was evaporated to dryness under nitrogen flow and reconstituted using 10 μL of 50/50 (v/v) methanol/water, which was then drawn into a SERS-active capillary for measurement.
Analyte concentrations were verified using gas chromatography with flame ionization detection (GC-FID) (Model 17 A, Shimadzu, Columbia, MD, USA). The instrument was fitted with a split-splitless injector and an RTx-1 capillary column (30 m × 0.53 mm, 100% dimethylpolysiloxane; Restek, Bellefonte, PA, USA), and helium was used as the carrier gas. The injector was set to 300°C and a split ratio of 1. The oven temperature program was as follows: initial temperature 150°C; ramp to 305°C at 10°C/min; hold at 305°C for 4 min. The FID temperature was maintained at 300°C. Samples were injected at a volume of 1 μL.
SERS analysis
For all samples, a 20 μL aliquot was manually drawn into a prepared SERS-active capillary using a syringe. The capillary was mounted on an XY stage above a fiber-optic probe for immediate analysis. SERS measurements were performed using FT-Raman and dispersive Raman analyzers, both employing laser excitation at 785 nm and a power of 80 mW (models RamanID and SERS-Lab, respectively, Real-Time Analyzers, Middletown, CT, USA). Spectra were collected at 10 spots along the sol-gel plug, at 1 mm intervals, for 1 minute each using the FT-Raman analyzer and 10 seconds each using the dispersive Raman analyzer, unless otherwise noted; the results for each capillary were averaged using Raman VISTA software (Real-Time Analyzers).
Results
Throughout the development of this method, various aspects were investigated to achieve the detection of 10-50 ng/mL cocaine in saliva. This included silver vs. gold as the SERS-active metal; methods to preconcentrate the samples, such as SPE, sample pH, improvements in Raman analyzer technology, and methods to extract the cocaine from saliva.
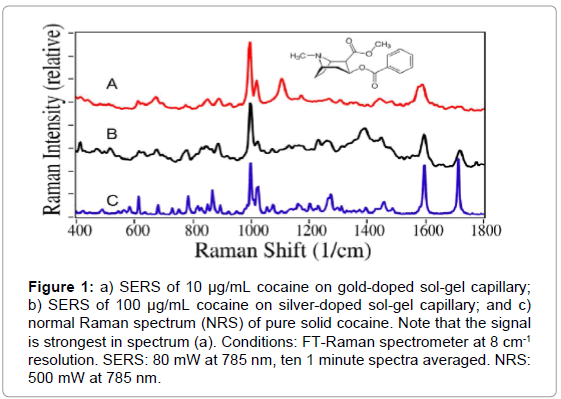
To determine the optimal sol-gel chemistry, cocaine in solution was measured using multiple variants of gold- and silver-doped solgels. The gold-doped sol-gels yielded greater sensitivity than the silverdoped sol-gels, by approximately two orders of magnitude. The lowest measured concentration using the silver-doped sol-gel capillaries was 100 μg/mL, while the lowest measured concentration for the golddoped sol-gel capillaries was 1 μg/mL. Representative spectra are shown in Figure 1.
Figure 1: a) SERS of 10 μg/mL cocaine on gold-doped sol-gel capillary; b) SERS of 100 μg/mL cocaine on silver-doped sol-gel capillary; and c) normal Raman spectrum (NRS) of pure solid cocaine. Note that the signal is strongest in spectrum (a). Conditions: FT-Raman spectrometer at 8 cm-1 resolution. SERS: 80 mW at 785 nm, ten 1 minute spectra averaged. NRS: 500 mW at 785 nm.
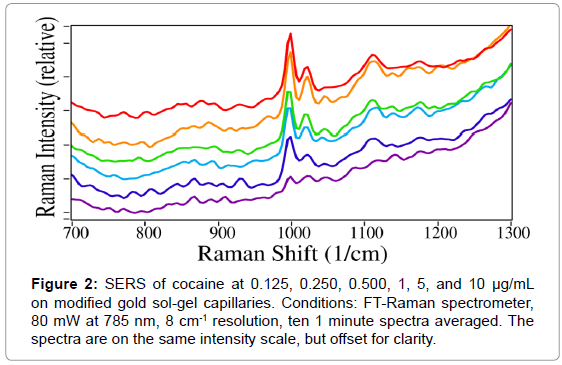
Given the better sensitivity obtained using gold, the gold sol-gel was selected for use in further experiments. For the first experiment, a concentration series of cocaine in water was examined to establish the SERS limit of detection using the gold capillaries. The intensity of the peaks, shown in Figure 2, follows a Langmuir curve (13). Furthermore, cocaine was consistently detected at concentrations as low as 125 ng/ mL without any sample pretreatment or spectral manipulation.
While 125 ng/mL is adequate for the federal mandatory workplace drug testing cutoff concentration of 150 ng/ mL [18], for clinical applications a lower detection limit is needed. In order to achieve the desired goal of 10-50 ng/mL in saliva, the possibility of a pretreatment step was investigated. In particular, SPE has been shown to be a reliable and inexpensive method for recovering low-concentration analytes from a matrix [19,20], and the authors have successfully used this method previously to recover drugs from saliva [21]. Because cocaine is a basic drug, it was necessary to select an SPE material that is appropriate for basic compounds. The UCT C8+SCX column, which contains monomerically bonded octyl and benzene sulfonic acid on silica and retains analytes with both neutral and amine functional groups, but passes analytes with carboxylate functional groups, was therefore used for pretreatment in all subsequent experiments.
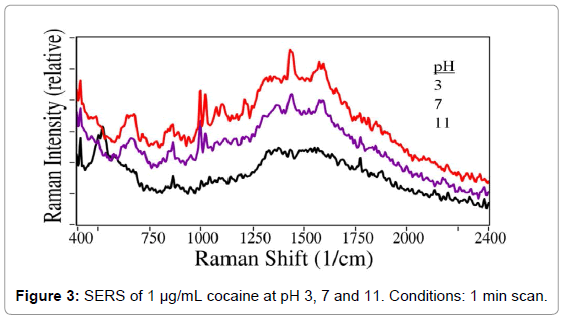
Since the pH of saliva can vary, the effect of pH on SERS measurements of cocaine in water was investigated. The pH of 1 μg/mL cocaine samples was varied by adding dilute HCl or NaOH to yield pH values of 3, 7, and 11. As shown in Figure 3, the characteristic cocaine peaks are relatively stable at acidic pH, but disappear at basic pH. This result was as expected, given that cocaine has a pKa of 8.6. Due to the demonstrated pH sensitivity, it was concluded that the saliva samples would have to be buffered in order to produce accurate results. For this purpose, acetate buffer was selected to mitigate the effects of pH in subsequent experiments.
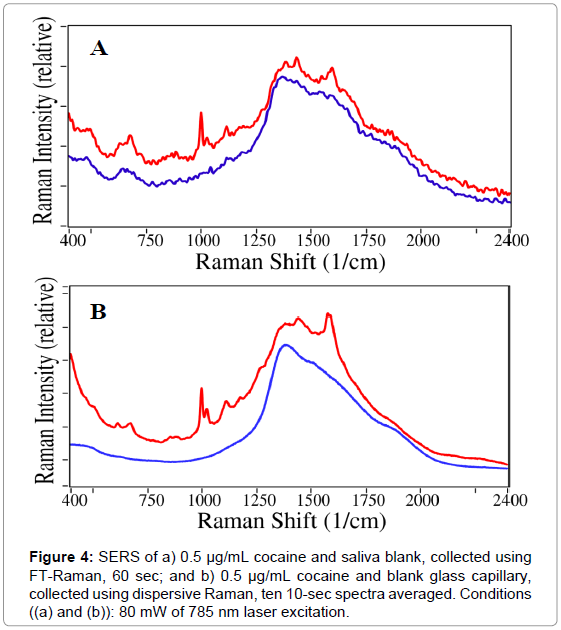
After collecting saliva from volunteers using an oral swab, the untreated samples were subjected to SERS analysis to ensure that the background spectrum of the saliva matrix would not interfere with cocaine measurements. First, a saliva blank was compared with 0.5 μg/ mL cocaine in saliva using the FT-Raman spectrometer. As shown in Figure 4 (a), the 1002 cm-1 cocaine peak is distinctly visible against the saliva background and the broad feature caused by luminescence of the glass capillary between 1200 and 2000 cm-1. A characteristic peak at 1020 cm-1 is also visible, attributed to the asymmetric phenyl ring breathing mode [22].
Since the FT-Raman spectra were relatively noisy, a dispersive Raman system was built employing a newly available low-noise chargecoupled- device 2-D array detector. While repeated measurements of the same cocaine sample yielded near-identical-intensity spectra with the same features, the dispersive spectrometer provided a 12-fold improvement in noise reduction based on the signal-to-noise ratio for the 1002 cm-1 peak relative to the spectrum baseline (Figure 4 (b)). It is also worth noting that the comparison of spectra from cocaine in a capillary, saliva in a capillary, and an empty capillary shows that saliva causes very little interference with the cocaine spectrum. Due to the improved signal quality, the dispersive Raman system was selected for use in further measurements.
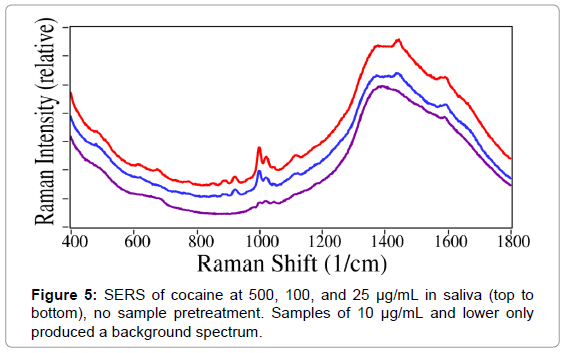
Cocaine-spiked saliva samples containing 1000, 500, 250, 100, 50, 25, 10, 5, and 1 μg/mL cocaine were loaded into SERS-active capillaries without pretreatment and measured using the dispersive Raman system. Spectral features specific to cocaine were detected at concentrations as low as 25 μg/mL (Figure 5). However, this is 3 orders of magnitude higher than the SAMHSA cut-off threshold.
In order to improve sensitivity, a series of solvents, specifically acetic acid, acetonitrile, chloroform, and hexane, were investigated to chemically separate cocaine from the saliva mucans. The best performance was observed for acetic acid. As noted above, due to the need to control the sample pH, acetate buffer was selected to meet both chemical separation and buffering requirements. Next, filtration and centrifugation were investigated to physically separate the cocaine from the saliva matrix. Both methods yielded similar results. Due to the ease of use and minimal equipment required for manual filtration compared with centrifugation, the former was selected for all further experiments.
Based on the results obtained for cocaine in water, an SPE pretreatment method was then developed for the spiked saliva samples, using a C8+SCX column as before. Several eluents were investigated, including acetate buffer, acetonitrile, ammonia, ethanol, hexane, hydrochloric acid, methanol, and water. Flow rates were varied from 0.1 to 5 mL/min. In addition to varying the solvent and flow rate, an additional step was added after the SPE process to further improve the sensitivity obtained from sample pretreatment. Following the extraction procedure, the solvent was evaporated from the eluate to concentrate the sample prior to measurement. The total time required for analysis of cocaine in saliva using this method, including pretreatment and SERS measurement, was 15.5 minutes. A schematic of the final method is shown in the Supporting Information (Figure S2).
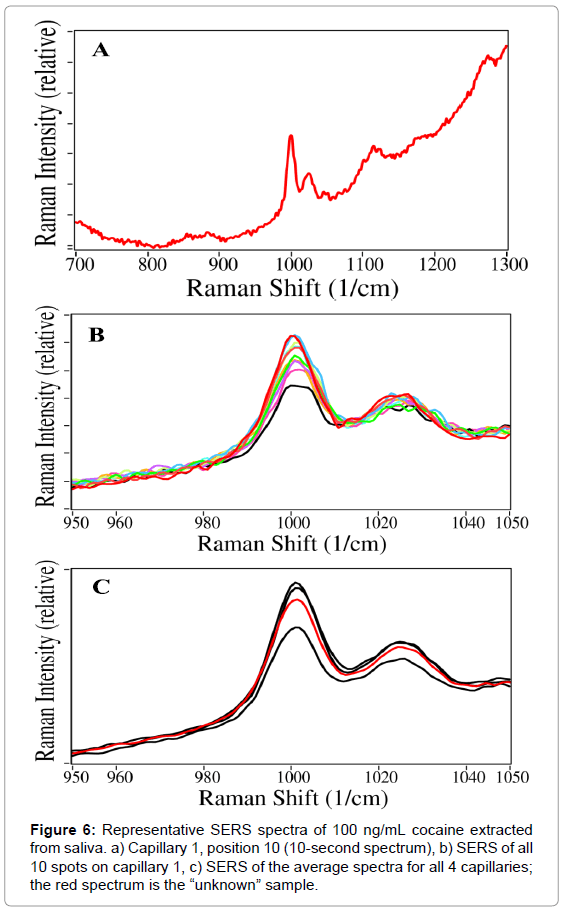
Artificially spiked cocaine samples in saliva at much lower concentrations were prepared at 1000, 500, 250, 100, 50, 25, and 10 ng/mL. At each concentration, four capillaries were prepared, three to be analyzed in triplicate and a fourth to be used later as an unknown; all capillaries were measured by SERS. This procedure yielded both sensitive and repeatable results, as illustrated in Figure 6.
The raw averaged spectra collected for spiked saliva samples were overlaid by setting the spectral intensity at 810 cm-1 to zero. The cocaine intensity was calculated as the peak height at 1002 cm-1 minus a baseline drawn from 950 to 1070 cm-1. The average standard deviation for the four capillaries individually was 17.1%, while the standard deviation between capillaries was 21.7%.
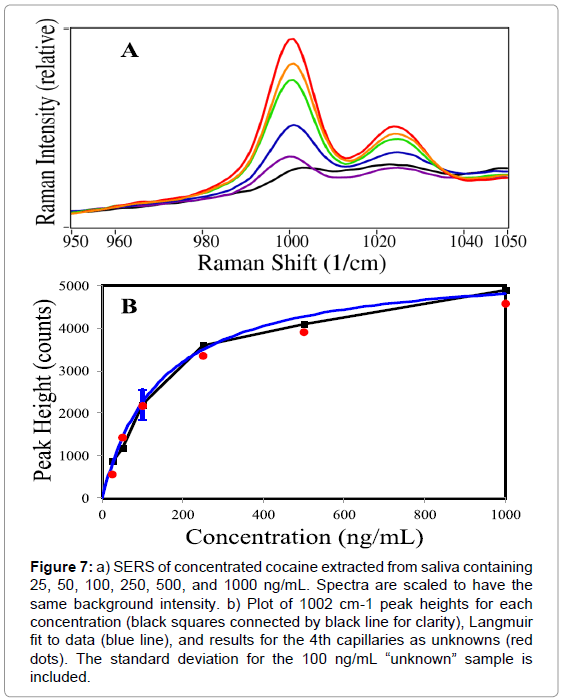
Figure 7 (a) shows the 3-capillary averaged spectra for cocaine in saliva at 1000, 500, 250, 100, 50, and 25 ng/mL (10 ng/mL was not observed). In Figure 7 (b), the peak heights at 1002 cm-1 are plotted as a function of concentration and fit with a Langmuir curve.
Figure 7: a) SERS of concentrated cocaine extracted from saliva containing 25, 50, 100, 250, 500, and 1000 ng/mL. Spectra are scaled to have the same background intensity. b) Plot of 1002 cm-1 peak heights for each concentration (black squares connected by black line for clarity), Langmuir fit to data (blue line), and results for the 4th capillaries as unknowns (red dots). The standard deviation for the 100 ng/mL “unknown” sample is included.
The Langmuir equation is Θ=[kC/(1+kC)]S, where Θ is the surface coverage (here expressed as the SERS peak height), k is a constant, C is the sample concentration and S is a scaling constant added to produce the correct SERS peak height. Here, k=0.007, C ranges from 0 to 1000 ng/mL, and S=2900. The Langmuir fit then represents a concentration curve for this analysis. For example, the Langmuir curve predicts the following concentrations for the “unknown” samples: 25=15, 50=50, 100=92, 250=225, 500=340, and 1000=750 ng/mL. It is worth noting that smaller errors occur at lower concentrations for a Langmuir curve. However, the SERS measurements produce different amounts of error at each different concentration, as reflected in their standard deviations (Table 1).
| 500 ng/ml | Cap 1 | Cap 2 | Cap 3 | Unknown | 4 Cap Ave |
|---|---|---|---|---|---|
| Spot 1 | 3290 | 1425 | 2100 | 1313 | |
| Spot 2 | 2550 | 2550 | 3000 | 2063 | |
| Spot 3 | 2025 | 1200 | 2550 | 1650 | |
| Spot 4 | 2400 | 1275 | 1875 | 1800 | |
| Spot 5 | 2550 | 1575 | 1575 | 1688 | |
| Spot 6 | 1800 | 2925 | 2700 | 2250 | |
| Spot 7 | 2588 | 1500 | 2700 | 1650 | |
| Spot 8 | 2700 | 2250 | 2700 | 1538 | |
| Spot 9 | 1838 | 2250 | 2925 | 2400 | |
| Spot 10 | 2625 | 2175 | 2775 | 1500 | |
| Average | 2436.5 | 1912.5 | 2490.0 | 1785.0 | 2156.0 |
| Std dev | 426.2 | 562.5 | 451.0 | 329.1 | 567.0 |
| % dev | 17.49% | 29.41% | 18.11% | 18.44% | 26.30% |
Table 1: Peak height measurements and statistics for 4 capillaries containing 100 ng/mL cocaine extracted from saliva (3 known and 1 unknown, 10 spots/capillary). Values shown are the peak height calculated at 1002 cm-1 minus a baseline drawn from 950 to 1070 cm-1.
Conclusion
A method was successfully developed to rapidly measure cocaine in saliva at concentrations as low as 25 ng/mL. The procedure requires minimal space and equipment and can be completed in less than 16 minutes, which makes it appropriate for use in settings such as hospital emergency rooms. Due to the inclusion of a buffer solution and the use of the C8+SCX SPE column, this method could also be applied to measure not only other basic drugs, but also acidic drugs in a saliva matrix. Ongoing research using saliva samples from regular drug users is currently being performed to confirm the accuracy of this method and determine its applicability in real-world conditions. Future studies of pretreatment options may allow the detection limit and analysis time to be improved, and experiments using other pharmaceuticals will confirm the applicability of this method to other compounds.
Acknowledgements
The authors would like to thank the National Institute on Drug Abuse for funding this research (NIH Grant Number 1R43DA032178-01).
References
- Substance Abuse and Mental Health Services Administration (SAMHSA) (2014) Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings. NSDUH Series H-48, HHS Publication No. (SMA) 14-4863.
- SAMHSA (2011) Drug Abuse Warning Network: National Estimates of Drug-Related Emergency Room Visits.
- Reidy LJ, Junquera P, Van Dijck K, Steele BW, Nemeroff CB (2014) Underestimation of substance abuse in psychiatric patients by conventional hospital screening. J Psychiatr Res 59: 206-212.
- Rainey PM, Schonfeld DJ (1997) The Yale University School of Medicine Patient's Guide to Medical Tests: Toxicology, Monitoring of Drug Therapy, and Testing for Substance Abuse. Houghton-Mifflin Co., New York, USA. pp: 587-596.
- Drummer OH (2005) Review: Pharmacokinetics of illicit drugs in oral fluid. Forensic Sci Int 150: 133-142.
- Crouch D, Day J, Baudys J, Fatah A (2004) Evaluation of Saliva/Oral Fluid as an Alternate Drug Testing Specimen. National Institute of Justice, USA Report Number 605-03.
- Allen KR (2011) Screening for drugs of abuse: which matrix, oral fluid or urine? Ann Clin Biochem 48: 531-541.
- Chamberlain J (1995) The Analysis of Drugs in Biological Fluids. 2nd edn. CRC Press, Boca Raton, FL, USA. p: 46.
- Scheidweiler KB, Kolbrich Spargo EA, Kelly TL, Cone EJ, Barnes AJ, et al. (2010) Pharmacokinetics of Cocaine and Metabolites in Human Oral Fluid and Correlation with Plasma Concentrations following Controlled Administration. Ther Drug Monit 32: 628-637.
- Dams R, Choo RE, Lambert WE, Jones H, Huestis MA (2007) Oral fluid as an alternative matrix to monitor opiate and cocaine use in substance-abuse treatment patients. Drug Alcohol Depend 87: 258-267.
- Schramm W, Craig PA, Smith RH, Berger GE (1993) Cocaine and benzoylecgonine in saliva, serum, and urine. Clin Chem 39: 481-487.
- SAMHSA (2010) Results from the 2009 National Survery on Drug Use and Health, Volume 1. NSDUH Series H-38A, HHS Publication No. SMA 10-4586.
- Farquharson S, Shende C, Inscore F, Maksymiuk P, Gift A (2005) Analysis of 5-fluoroacil in saliva using surface-enhanced Raman spectroscopy. J Raman Spectrosc 36: 208-212.
- Shende C, Inscore F, Gift A, Maksymiuk P, Farquharson S (2005) Ten-minute analysis of drugs and metabolites in saliva by surface-enhanced Raman spectroscopy. P Soc Photo-Opt Instrum Eng 6007: 165-171.
- Farquharson S, Shende C, Sengupta A, Huang H, Inscore F (2011) Rapid detection and identification of overdose drugs in saliva by surface-enhanced Raman scattering using fused gold colloids. Pharmaceutics 3: 425-439.
- Farquharson S, Lee YH, Nelson C (2003) Material for surface-enhanced Raman spectroscopy, and SER sensors and method for preparing same. US Patent No. 6623977.
- Farquharson S, Gift AD, Inscore FE, Shende CS (2008) SERS method and apparatus for rapid extraction and analysis of drugs in saliva. US Patent No. 7393691.
- SAMHSA (2008) Mandatory Guidelines For Federal Workplace Drug Testing Programs. Federal Register 73 FR 71858. p: 71880.
- Foulon C, Menet MC, Manuel N, Pham-Huy C, Galons H, et al. (1999) Rapid Analysis of Benzoylecgonine, Cocaine, and Cocaethylene in Urine, Serum, and Saliva by Isocratic High-Performance Liquid Chromatography with Diode-Array Detection. Chromatographia 50: 721-727.
- Hegstad S, Oiestad EI, Johansen U, Christophersen AS (2006) Determination of Benzodiazepines in Human Urine Using Solid-Phase Extraction and High-Performance Liquid Chromatography-Electrospray Ionization Tandem Mass Spectrometry. J Analytical Toxicology 30: 31-37.
- Inscore F, Shende C, Sengupta A, Huang H, Farquharson S (2011) Detection of drugs of abuse in saliva by surface-enhanced Raman spectroscopy (SERS). Appl Spectrosc 65: 1004-1008.
- Carter JC, Brewer WE, Angel SM (2000) Raman spectroscopy for the in situ identification of cocaine and selected adulterants. Applied Spectroscopy 54: 1876-1881.
Relevant Topics
Recommended Journals
Article Tools
Article Usage
- Total views: 18566
- [From(publication date):
December-2015 - Aug 24, 2025] - Breakdown by view type
- HTML page views : 13658
- PDF downloads : 4908