A Pediatric Case of Very Late Onset Non-infectious Pulmonary Complication (LONIPC) after Allogeneic Hematopoietic Stem Cell Transplantation
Received: 19-Apr-2017 / Accepted Date: 22-Jun-2017 / Published Date: 28-Jun-2016 DOI: 10.4172/2475-7640.1000104
Abstract
17-months old infant with Wiskott-Aldrich syndrome was transplanted with genetically two-locus mismatched unrelated cord blood cells under the conditioning regimen of busulfan, cyclophosphamide and anti-thymocyte globulin. GVHD prophylaxis was cyclosporine (CSP) plus short-term methotrexate with prednisolone (1 mg/kg). Acute and chronic GVHD were not observed during the course. About six years later, he suffered from severe cough and dyspnea with no fever, and diagnosed as late onset non-infectious pulmonary complications (LONIPC). We have to be careful for LONIPC after SCT even in the absence of chronic GVHD for younger children because the sign of insidious obstructive pulmonary symptoms is difficult to monitor. To avoid the unexpected LONIPC, introduction of RIC regimen should be considered for younger children, although it has been reported that RIC regimen may increase the risk for GVHD and graft rejection.
Keywords: Non-infectious pulmonary complication; Chronic GVHD; Allogeneic hematopoietic stem cell transplantation
7654Introduction
Late onset non-infectious pulmonary complications (LONIPC) consist of major causes of morbidity and mortality after allogeneic hematopoietic stem cell transplantation (allo-HSCT). These include bronchiolitis obliterans (BO), bronchiolitis obliterans with organizing pneumonia (BOOP), and idiopathic pneumonia syndrome (IPS), and usually occur three months after HSCT. According to the literature, the incidence has been reported to be 10-25% [1]. Not well elucidated, it is speculated that allo-immune reactions play a major role in the pathogenesis, because they are closely associated with the presence of chronic GVHD. Concerning to LONIPC among the pediatric patients, few reports are available. Kojima et al have reported as a single institutional experience that the incidence of LONIPC in the pediatric patients was 10.3%, the median onset was 187 days (123-826 days), and it was also related with chronic GVHD as in the cases of adults [2].
Case Report
17-months old infant with Wiskott-Aldrich syndrome was transplanted with genetically two-locus mismatched unrelated cord blood cells. Major clinical symptoms were eczema and thrombocytopenia (4/μl) and he had no significant infection episodes before SCT. No expression of WASP protein was found in T-lymphocytes, and genetic analysis revealed splicing anomaly due to point mutation in intron2. Conditioning regimen consisted of busulfan (4 mg/kg/day for 4 days), cyclophosphamide (50 mg/kg/ day for 4 days) and horse anti-thymocyte globulin (10 mg/kg/day for 4 days), and GVHD prophylaxis was cyclosporine (CSP) plus shortterm methotrexate with prednisolone (1 mg/kg). Dose of oral busulfan was determined by the prior test dose and pharmacokinetic analysis. Methotrexate on day 6 and day 11 was omitted because of elevated serum transaminase. Neutrophil engraftment was on day 19, platelet engraftment on day 71 and complete chimerism was determined on day 21.
Because no GVHD was observed, CSP was stopped on day 95, and prednisolone on day 251. He became free of immunoglobulin supplementation one year after CBT. There were no significant infection episodes after CBT.
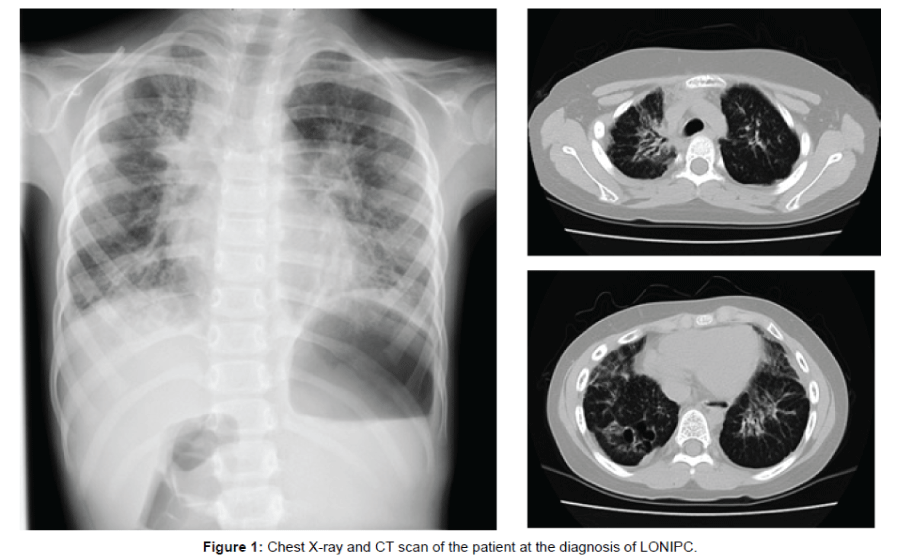
5 and 1/2 years after CBT, he began to complain of occasional dry cough, which resolved easily under cough remedy. A couple of months later, he suffered from severe cough and dyspnea with no fever. Auscultation revealed fine crackles on both lungs, and his oxygen saturation was 92% in room air. Chest X-ray presented marked consolidation of bronchioles (Figure 1). Under the suspicion of LONIPC, prednisolone (1 mg/kg/day) was started, which ameliorated his symptoms rapidly. Serum soluble IL-2R was slightly elevated to 937 U/ml (normal value: 145-519), and serum KL-6 was 458 U/ml (normal value: 0-500). Involvement of pathogens including RSV, CMV and fungi was excluded from the later examinations. He has been placed on oral prednisolone with clarithromycin (5 mg/kg/day) and fluticasone inhalation (twice daily). Three years later, he has been placed on home oxygen therapy and lung transplantation has been scheduled.
Discussion
Between 2001 and 2011, 72 pediatric patients (7m-22y8m; 7y3m ± 6y8m [mean ± SD]) have received 77 allogeneic stem cell transplantations in our institute. 9 patients (13.4%) in 67 patients who survived more than 3 months after SCT developed LONIPC (Table 1: observation period; 1155-5145 days). Multivariate analysis revealed myeloablative conditioning regimen and chronic GVHD as significant risk factors of LONIPC (data not shown). Unexpectedly, two patients developed LONIPC more than five years after HSCT and one patient died of respiratpry failure 30 months after diagnosis of LONIPC.
| S.No | Age at SCT | Source of SCT | Conditioning regimen | GVHD prophylaxis | aGVHD | cGVHD | Onset of NIPC | Outcome | Cause of death | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | CAEBV | 18y8M | UBMT | TBI+CY+VP | CSP + sMTX | II | (+) | 126 | Dead (631) | Respiratory failure |
| 2 | AML | 14y4M | UBMT | BU+LPAM+CY | CSP + sMTX | III | (+) | 90 | Dead (335) | Respiratory failure pneumoniae |
| 3 | WAS | 11y7M | UBMT | BU+CY+ATG | TAC + sMTX | IV | (+) | 234 | Dead (373) | Respiratory failure pneumoniae |
| 4 | AML | 13y4M | Mismathced-related (mother) | TBI+CY | TAC + sMTX | III | (+) | 107 | Dead (374) | AML |
| 5 | AML | 1y10M | MSB | iBU+LPAM | CSP + sMTX | IV | (+) | 98 | Alive (>2065) | - |
| 6 | ALL | 7y | Mismathced-related (mother) | TBI+CY+VP+ATG | TAC + sMTX + PSL | I | (+) | 131 | Alive (>1582) | - |
| 7 | ALL | 17y7M | MSB | TBI+CY+VP | CSP | II | (+) | 345 | Dead (720) | Subdralhemorrhage |
| 8 | WAS | 8M | UBMT | BU+CY+ATG | TAC + sMTX | II | (-) | 3578 | Dead (4470) | Respiratory failure |
| 9 | WAS | 1y5M | UCBT | BU+CY+ATG | CSP + sMTX + PSL | (-) | (-) | 2118 | Alive (>3821) | - |
CAEBV: Chronic Active EB Virus Infection; MDS: Myelodysplastic Symdrome; WAS: Wiskott-Aldrich syndrome; AML: Acute Myelogenous Leukemia; ALL: Acute Lymphocytic Leukemia; UBMT: Unrelated Bone Marrow Transplantation; MSB: Matched Sibling; UCBT: Unrelated Cord Blood Transplantation; TBI: Total Body Irradiation; CY: Cyclophosphamide; VP: Etoposide; BU: Busulfan; LPAM: Melphalan; ATG: Anti-Thymocyte Globulin; CSP: Cyclosporine; MTX: Methotrexate; PSL: Predonisolone
Table 1: Profile of patients who developed LONIPC.
As shown in Table 1, LONIPC was closely related with chronic GVHD also in our experience. Unexpectedly, two patients had no history of chronic GVHD before the onset of LONIPC contrary to other patients. The mean onset except for two cases was 161 days (90-345 days), which is comparable to previous reports. Four patients were dead due to LONIPC or its associated complications such as pneumonia. Described case was peculiar in that he had no apparent history of acute and chronic GVHD, and that the onset of LONIPC was extremely late. One possible risk factor is busulfan used in the conditioning regimen. It has been reported that busulfan may be associated with greater longterm lung toxicity than TBI in pediatric patients [3]. In our preliminary data, 18 patients with reduced intensity conditioning regimen during the same period developed no LONIPC.
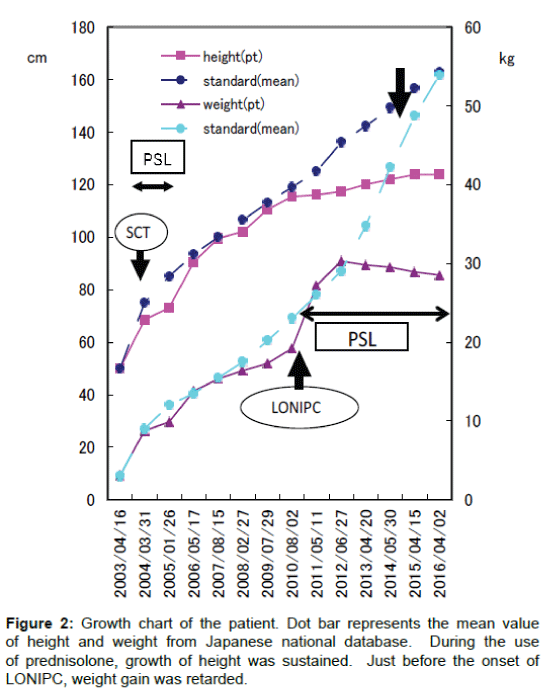
It has been reported that decline of air flow could be a good hall mark of non-infectious pulmonary complication after SCT [4-6]. However, it is difficult to monitor FEV1/FVC for younger children such as this case. Figure 2 showed the growth chart of this patient after SCT. Retrospectively, retardation of weight gain was found before the onset of LONIPC, although it is difficult to determine whether it was an early sign of pulmonary symptoms or not. We have to be careful to check the development of LONIPC after SCT even in the absence of chronic GVHD especially for infants or younger children because the sign of insidious obstructive pulmonary symptoms is difficult to monitor. It has been recently reported that RIC regimen stabilized longterm pulmonary function in adult patients with allogeneic SCT [7]. To avoid the unexpected LONIPC, introduction of RIC regimen should be considered for younger children, although it has been reported that RIC regimen may increase the risk for GVHD, CMV reactivation and graft rejection [8,9].
References
- Tomas LHS, Loberiza FR, Klein JP, Layde PM, Lipchik RJ, et al. (2005) Risk factors for bronchiolitis obliterans in allogeneic hematopoietic stem-cell transplantation for leukemia. CHEST 128:153-161
- Nishio N, Yagasaki H, Takahashi Y, Muramatsu H, Hama A, et al. (2009) Late-onset noninfectious pulmonary complications following allogeneic hematopoietic stem cell transplantation in children. Bone Marrow Transplant 44:303-308
- Bruno B, SouilletG, BertrandY, Werck-Gallois MC, Satta AS, et al. (2004) Effects of allogeneic bone marrow transplantation on pulmonary function in 80 children in a single paediatriccentre. Bone Marrow Transplant 34: 143-147
- Park M, Koh KN, Kim BE, Im HJ, Seo JJ (2011) Clinical features of late onset non-infectious pulmonary complications following pediatric allogeneic hematopoietic stem cell transplantation. Clin Transplant 25: 168-176.
- Chien JW, Martin PJ, Gooley TA, Flowers ME, Heckbert SR(2003) Air flow obstruction after myeloablative allogeneic hematopoietic stem cell transplantation. Am J RespirCrit Care Med 168: 208-214
- Sritippayawan S, Keens TG, Horn MV, Starnes VA, Woo MS (2003) What are the best pulmonary function test parameters for early detection of post-lung transplant bronchiolitis obliterans syndrome in children?Pediatric Transplantation 7: 200-203
- Dirou S, Malard F, Chambellan A, Chevallier P, Germaud P, et al (2014). Stable lomg-term pulmonary function after fludarabine, antithymocyte globulin and i.v. BN for reduced-intensity conditioning allogeneic SCT. Bone Marrow Transplantation 49: 622-627.
- George B, Kerridge I, Gilroy N, Huang G, Hertzberg M, et al. (2010)Fludarabine-based reduced intensity conditioning transplants have a higer incidence of cytomegalovirus reactivation compared with myeloablative transplants.Bone Marrow Transplant 45: 849-855.
- Burroughs LM, Storb R, Leisenring WM, Pulsipher MA, Loken MR, et al. (2007) Intensive postgrafting immune suppression combined with nonmyeloablative conditioning for ytansplantation of HLA-identical hematopoietic cell graft: results of a pilot study for treatment of primary immunodeficiency disorders.Bone Marrow Transplant 40: 633-642.
Citation: Nagasawa M, Ono T, Aoki Y, Isoda T, Takagi M, et al. (2016) A Pediatric Case of Very Late Onset Non-infectious Pulmonary Complication (LONIPC) after Allogeneic Hematopoietic Stem Cell Transplantation. J Clin Exp Transplant 1: 104. DOI: 10.4172/2475-7640.1000104
Copyright: © 2016 Nagasawa M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 13539
- [From(publication date): 7-2016 - Jul 06, 2025]
- Breakdown by view type
- HTML page views: 12609
- PDF downloads: 930