Research Article Open Access
A Randomized Controlled Pilot Trial of Chinese Medicine (Di-Tan Decoction) in the Treatment of Alzheimer's Disease
Ka-Kit Chua1,2, Adrian Wong3,4, Pauline Wing-Lam Kwan3,4, Ju-Xian Song1, Lei-Lei Chen1, Andrew Lung-Tat Chan5, Zhao-Xiang Bian1,2, Vincent Mok3,4* and Min Li 1,2*1Chi-Ming Centre for Parkinson’s Disease Research, School of Chinese Medicine, Hong Kong Baptist University, Hong Kong
2Hong Kong Chinese Medicine Clinical Study Centre, Hong Kong Baptist University, Hong Kong
3Institute of Integrative Medicine, Department of Medicine and Therapeutics, the Chinese University of Hong Kong, Hong Kong
4Therese Pei Fong Chow Research Centre for Prevention of Dementia, the Chinese University of Hong Kong, Hong Kong
5Divisions of Neurology and Geriatrics, Department of Medicine, Queen Elizabeth Hospital, Hong Kong
- *Corresponding Author:
- Min Li, Director
Teaching and Research Division
Director of Mr. & Mrs. Ko Chi-Ming Centre for Parkinson’s Disease Research
Associate Director of Clinical Division, School of Chinese Medicine
Hong Kong Baptist University, Kowloon Tong, Kowloon, Hong Kong
Tel: +852-3411 2919
E-mail: limin@hkbu.edu.hk - Vincent Mok
Director, Therese Pei Fong Chow Research Centre for Prevention of Dementia
Director of Master Programme in Stroke and Clinical Neurosciences
Department of Medicine and Therapeutics, Prince of Wales Hospital
The Chinese University of Hong Kong, Shatin, New Territories, Hong Kong
Tel: + 852-2632 2195
E-mail: vctmok@cuhk.edu.hk
Received date: August 08, 2016; Accepted date: August 11, 2016; Published date: August 18, 2016
Citation: Chua K, Wong A, Kwan PW, Song J, Chen L, et al. (2016) A Randomized Controlled Pilot Trial of Chinese Medicine (Di-Tan Decoction) in the Treatment of Alzheimer’s Disease. J Alzheimers Dis Parkinsonism 6:256. doi:10.4172/2161-0460.1000256
Copyright: © 2016 Chua K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Alzheimers Disease & Parkinsonism
Abstract
Objective: This double-blind, randomized, placebo-controlled, add-on pilot study aimed at providing information for conducting a full-scale trial assessing “Di-tan decoction” (DTD), which is a traditional Chinese medicine (TCM) formula frequently used in TCM to treat symptoms that are now defined as Alzheimer’s disease (AD), in treating AD in the future. Methods: We randomly assigned 38 patients with AD to receive either DTD or placebo for 24 weeks. Primary outcome was changes in the total score of AD Assessment Scale-cognitive subscale (ADAS-cog) and secondary outcome was changes in the total score of Chinese version of the Disability Assessment for Dementia (C-DAD). Results: Although we observed some improvement in the total scores of ADAS-cog in the DTD group comparing to the placebo group, the changes were not statistically significant. The ADAS-cog sub-scores of the DTD group also showed non-significant trends of improvement in ideational praxis (p=0.100) and in comprehension (p=0.106) comparing to placebo group. Adverse events were mild and comparable between two groups. Conclusion: This is the first rigorous randomized control trial of DTD focusing on AD. At least five factors could explain the failure of the trends to be significant, namely length of trial, size of trial, stage of AD, palatability of the drug, and sensitivity of the scoring system. Given the limitation but with the safety and century’s use of DTD, a modified pilot study is needed to support the clinical effects of DTD. In conclusion, there is no evidence supporting the efficacy of DTD to act as a single treatment for AD.
Keywords
Alzheimer’s disease; Pilot study; Randomized controlled trial; Di-Tan decoction; Chinese medicine; Efficacy; Safety
Abbreviations
AD: Alzheimer’s Disease; Aβ: Beta-Amyloid-Peptide; ChEIs: Cholinesterase Inhibitors; TCM: Traditional Chinese Medicine; PTOO: Phlegm Turbidity Obstructing the Orifices; DTD: Di-Tan Decoction; Ach: Acetylcholine; ChAT: Acetylcholine Transferase; AchE: Acetylcholine Esterase; CONSORT: Consolidated Standards of Reporting Trials; NINCDS/ADARA: National Institute of Neurological and Communicative Disorders and Stroke and Alzheimer’s Disease and Related Disorders Association; CDR: Clinical Dementia Rating Scale; ADAS-cog: Alzheimer’s Disease Assessment Scale-cognitive Subscale; C-MMSE: Chinese Version of Mini-Mental State Examination; C-DAD: Chinese Version of Disability Assessment for Dementia; AE: Adverse Event; LOFC: Last-Observation-Carried-Forward; MoCA: Montreal Cognitive Assessment
Background
Alzheimer’s disease (AD) is an irreversible, progressive and fatal neurodegenerative disease. Biochemically, it is characterized by accumulation of beta-amyloid-peptide (Aβ) in the brain. Symptomatically it is manifested as cognitive and memory deterioration, progressive impairment of motor skills needed in the activities of daily living as well as a variety of neuropsychiatric symptoms and behavioral disturbances [1]. It is the most common type of dementia in the elderly and imposes a great economic stress on both individuals and society [2]. The prevalence of AD is about 2% for people aged 65-69 and up to more than 25% for people aged over 90 [3]. The global AD population is predicted to increase to 114 million by 2050 if there is no new preventive intervention [4].
Cholinesterase inhibitors (ChEIs) and memantine are the two major types of pharmacological treatment for AD. While these chemicals ameliorate symptoms, they do not stop or slow the progressive neural death and malfunction in the AD brain [5]. They are also associated with unwanted effects, such as nausea, vomiting, dizziness and anorexia [6]. Given that current medication cannot halt disease progression and has undesirable side effects, patients often seek alternative treatments [7].
When described in terms of the principles of Traditional Chinese Medicine (TCM), AD is described as: (1) deficiency of vital energy of the kidney, heart and spleen; and (2) stagnation of blood and/or phlegm [8]. The guidelines for classifying dementia into different sub-types according to the TCM theory was published in 1990 [9]. Symptoms of “phlegm turbidity obstructing the orifices” (PTOO) (Additional file 1) is considered to be one of the major contributing factors to AD, and “resolving phlegm to open the orifices” is a primary TCM strategy for treating AD [10].
“Di-tan decoction” (DTD), a TCM formula, was developed by TCM doctor Dong Su in 1449 with the specific function of “resolving phlegm to open the orifices” [11]. It has been frequently used in TCM clinics to treat symptoms that are now defined as AD [12,13]. Some laboratory studies have shown a significant reduction in memory impairment of AD model mice treated with DTD [14,15].In the brain tissue of these mice, acetylcholine (Ach) and acetylcholine transferase (ChAT) were significantly increased, while acetylcholine esterase (AchE) was decreased [16]. The results of another study indicate that DTD may inhibit the decline of dopamine content in model mice brain tissue [17].
Nevertheless, our previous systematic review found no clinical trial studying DTD as the main intervention for treating AD. We did find AD clinical trials that had involved DTD as an intervention adjunct, but they were methodologically flawed. They not only lacked randomization, blinding and the use of control groups, but also failed to define inclusion and exclusion criteria and even the quality of the intervention medicine [18]. Thus, there is a lack of conclusive evidence for whether DTD can relieve AD clinically, and there is no basic information, such as sample size needed, for conducting a clinical trial assessing the value of DTD in treating AD.
In this double-blind, randomized, placebo-controlled, add-on pilot trial (ChiCTR-TRC-12004548, data of registration: 2012/11/21), we aimed to study the efficacy and safety of DTD in treating patients with AD. It is the first rigorous study of DTD focusing on AD, and it provides useful information for conducting a full-scale clinical trial if further study is warranted in the future.
Methods
Study design
As reported in our previous published study protocol [19], this study was a double-blind, randomized, placebo-controlled, add-on trial. Patients with mild to moderate AD were randomly assigned to a 2-week run-in period to fix their routine medication. Then they had received 24 weeks of active herbal treatment, DTD or placebo (in a 1:1 ratio) and followed for a further 6 weeks’ observation period without treatment. It was carried out at the Hong Kong Baptist University Chinese Medicine Specialty Centre. It had been approved by the Ethics Committee of the Hong Kong Baptist University’s Institutional Review Board (code: HASC/11-12/24). The study result was reported following the guidelines of Consolidated Standards of Reporting Trials (CONSORT).
Participants
Inclusion criteria: Adults were eligible for this study who (1) had been clinically diagnosed with AD based on the criteria of National Institute of Neurological and Communicative Disorders and Stroke and Alzheimer’s Disease and Related Disorders Association (NINCDS/ ADARA) [20] and (2) presented symptoms classified as PTOO (Additional file 2) as defined by the Guidance for Clinical Research of New Chinese Herbal Medicine [21] during a screening visit.. They all had mild to moderate dementia with 0.5-2 on Clinical Dementia Rating Scale (CDR) [22] with normal liver and renal function as well as a stable medication history.
Exclusion criteria: Patients who had any other type of dementia (i.e., vascular dementia), and/or other neurodegenerative disorder (i.e., Parkinson’s disease), depression (defined by a score of ≥ 8 on the 15- item Chinese version of the Geriatric Depression Scale [23]), or who were unwilling to cooperate with treatment procedures were excluded.
Study medication
The herbal medicine being studied was “Di-tan Decoction” (�?�?� 湯in Chinese), or DTD. It was composed of 11.42% Arisaema Cum Bile (DanNanXing�?��?�?? in Chinese), 11.42% Pinelliae Rhizoma (FaBanXia �?�?夏 in Chinese), 9.13% Aurantii Immaturus Fructus (ZhiShi �?�實 in Chinese), 9.13% Poria (FuLing �?��?? in Chinese), 6.84% Citri Reticulatae Pericarpium (ChenPi �?��?� in Chinese), 4.55% Acori Tatarinowii Rhizoma (ShiChangPu �?��?�?� in Chinese), 4.55% Ginseng Radix (RenShen 人�? in Chinese), 3.19 Bambusae in Taeniam Caulis (ZhuRu 竹�?� in Chinese), 2.29% Glycyrrhizae Radix (GanCao �??�? in Chinese), 18.1% Zingiberis Recens Rhizoma (ShengJiang �??�?? in Chinese) and 19.36% dextin [11]. The placebo was made of caramel (2%), gardenia yellow pigment (0.05%), sunset yellow (0.02%), tartrazine (0.02%), dextrin (95%) and broad leaf holly leaf (2.91%) [24]. The DTD granules and the placebo granules had identical appearance and smell, and both were packed in sealed opaque aluminum sachets and put in zip lock bags (10 sachets each). All DTD and placebo granules were distributed by L.L. Chen with both written and verbal instructions for each participant. Patients were instructed to take the granules orally by dissolving a sachet of granules in 150ml hot water, stirring well, then drinking, two times per day, with at least two hours apart from taking any routine Western medication.
Recruitment procedures
All recruited patients diagnosed with AD were referred to a TCM doctor who is the Principle Investigator of the study (M. Li), or to a Research Assistant (K.K. Chua), for further assessment and recruitment. The aim, procedures, nature of study and possible side effects of DTD were explained by the PI or RA. Patients had to sign a written consent before they started the study treatment. They were informed they were free to withdraw at any time during the study.
Randomization and masking
The randomization sequence was generated by SPSS 19.0 package (SPSS, Chicago, IL) and password-protected and kept in a computer by L.L. Chen. Group allocation was simple randomization in a ratio of 1:1 to either active treatment group or placebo group. The number sequences was kept in sealed opaque envelopes and distributed to assessors. Patients, investigators and all sponsoring parties were masked to treatment allocation until the end of the study. When there was a serious adverse event, the event was discussed between the principal investigator TCM expert M. Li, and the co-investigator neurology specialist V. Mok to consider unblinding.
Outcome measurements and its assessment
The positive primary outcome of this study was a decrease [25] in the total score of Alzheimer’s disease Assessment Scale-cognitive subscale (ADAD-cog) [26]. Increase in the total scores of the Chinese version of Mini-Mental State Examination (C-MMSE) [27,28] and Chinese version of Disability assessment for dementia score (C-DAD) [29] were used as secondary outcomes.
Outcome measurements were carried out during the study visits at weeks 0 (baseline), 12 (half of treatment), 24 (end of treatment) and 30 (end of observation period). Safety assessment, which included reporting of adverse events (AE), measurement of vital signs and physical examination, were carried out throughout the study. The laboratory safety screening of liver and renal function was performed at week 24.
A home diary was given to each study participant’s caregiver to record treatment and changes in the participant’s medical condition. Formal instruction for the home diary was given during the first visit. Revision and checking of the diary was carried out with the caregiver during each formal visit by the assessors. Compliance in taking the treatments was determined by the record of the diary and the number of the returned medicine / placebo packages.
Statistical Analysis
The differences of the total score between the measurement points and baseline in ADAS-cog, C-MMSE and C-DAD were compared between the treatment group and placebo group using the Chi-Squared test and independent sample t test, respectively. Post-hoc analysis of differences in the change of ADAS-cog and C-DAD sub-scores were also compared between the treatment group and placebo group.
To reduce the number of statistical comparisons, analyses were performed with a hierarchical approach. To begin, the change in total score of ADAS-cog at week 24 (end of treatment) for the treatment group and placebo group were compared. If the difference was deemed to be statistically significant at a 2-sided α-level of 0.1, the change of ADAS-cog total score at week 12 (half of treatment) and week 30 (end of observation period, i.e. without treatment) was compared between the treatment group and placebo group. Other outcome measurements were also analyzed in the same manner.
Missing data was input using the last-observation-carried- forward (LOFC) approach. All patients randomized with at least one post-randomization measurement were included in the primary analysis to follow the intention-to-treat principle. Analyses were done by SPSS 19.0 package (SPSS, Chicago, IL).
Results
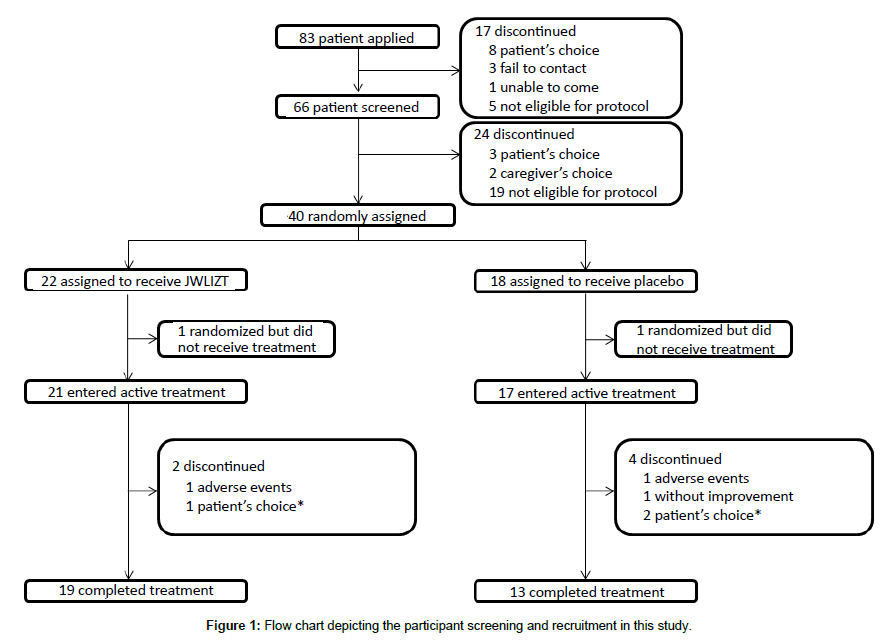
Figure 1 is a flow chart depicting the participant screening and recruitment in this study. Demographic data and baseline scores are summarized in Table 1. A total of 66 patients were screened for eligibility, and 40 participants were enrolled between 1st December 2012 and 30th November 2014. Two patients withdrew from the study due to personal reasons after randomization and before the start of treatment. Among the remaining 38 patients (14 male; 24 female; mean ages 74.45 ± 9.01 years; mean duration of AD 2.32 ± 1.70 years), 21 were assigned to the DTD group, 17 to the placebo group. Six participants dropped out during the study due to reasons listed in Figure 1. Nineteen participants in the DTD group and 13 in the placebo group completed the study.
| Parameter | DTD group (n=21) | Placebo group (n=17) | p-valuea |
|---|---|---|---|
| Age (years) | 76.57 ± 8.16 | 71.82 ± 9.55 | 0.107b |
| Gender (M/F) | 8/13 | 6/11 | 0.859 |
| Disease duration (years) | 2.58 ± 1.81 | 1.99 ± 1.54 | 0.290b |
| ChEIs, n (%) | 16 (76.2) | 15 (88.2) | 0.341c |
| Memantine, n (%) | 2 (9.5) | 1 (5.9) | 0.679c |
| Baseline scores | |||
| CDR | 1.05 ± 0.67 | 0.77 ± 0.40 | 0.134b |
| MMSE | 17.43 ± 5.61 | 16.82 ± 4.88 | 0.728b |
| Systolic Blood Pressure | 140.88 ± 18.35 | 145.68 ± 22.17 | 0.470b |
| Diastolic Blood Pressure | 72.79 ± 9.73 | 74.27 ± 10.50 | 0.656b |
| Heart Rate | 65.02 ± 8.48 | 68.06 ± 11.72 | 0.361b |
Data are expressed as mean ± S.D
ap-value was comparing the difference between two groups in baseline
btreatment group compared with placebo group by independent t-test
ctreatment group compared with placebo group by Chi-square test with continuity correction
Table 1: Baseline characteristics of AD patients in DTD group and placebo group.
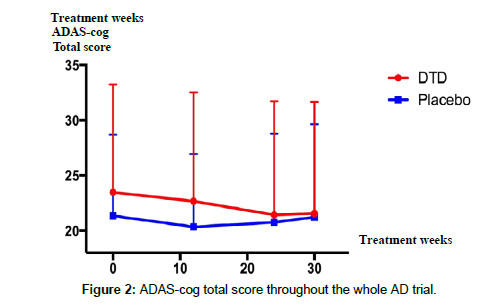
No statistically significant improvement (p=0.315, Table 2) was obtained in either group according to the primary outcome, i.e., the change in total score of ADAD-cog. However, we observed a trend of improvement, namely, a decreasing score at week 24 that suggests there may have been cognitive improvement in the DTD group compared to the control group (Table 2). Also, analyses of ADAS-cog sub-scores between the two groups reveals that the DTD group showed non-significant trends of improvement in ideational praxis (p=0.100) and in comprehension (p=0.106) compared to the placebo group (Table 2). Post-hoc analysis suggested that the improvement trend continued throughout the treatment period, and declined after DTD treatment stopped (Table 3 and Figure 2).
| Cognitive subscale of Alzheimer’s Disease Assessment Scale (ADAS-cog) | ||||||
|---|---|---|---|---|---|---|
| DTD | Placebo | p-value | ||||
| Total Score | -2.05 ± 5.26 | -0.59± 2.98 | 0.315 | |||
| D1 Word recall task | -0.10 ± 0.94 | -0.18 ± 1.24 | 0.820 | |||
| D2 Naming objects and fingers | 0.00 ± 0.71 | 0.24 ± 0.66 | 0.302 | |||
| D3 Commands | -0.14 ± 0.79 | -0.12 ± 0.60 | 0.914 | |||
| D4 Constructional praxis: figures | -0.14 ± 0.66 | -0.12 ± 0.78 | 0.914 | |||
| D5 Ideational praxis | -0.19 ± 0.87 | 0.35 ± 1.12 | 0.100 | |||
| D6 Orientation | -0.14 ± 1.24 | -0.53 ± 1.13 | 0.325 | |||
| D7 Word recognition test | -0.76 ± 3.03 | -0.06 ± 1.25 | 0.377 | |||
| D8 Remembering test instructions | -0.48 ± 1.47 | -0.24 ± 0.83 | 0.551 | |||
| D9 Spoken language ability | 0.00 ± 0.32 | 0.00 ± 0.00 | 1.000 | |||
| D10 Word finding difficulty | 0.05 ± 0.38 | -0.06 ± 4.3 | 0.425 | |||
| D11 Comprehension | -0.14 ± 0.57 | 0.12 ± 0.33 | 0.106 | |||
| Chinese Version-Disability assessment for dementia score (C-DAD) | ||||||
| Total Score | 3.35 ± 7.95 | 4.42 ± 12.91 | 0.755 | |||
| D1 Hygiene | 2.89 ± 9.76 | 3.78 ± 15.23 | 0.828 | |||
| D2 Dressing | 2.04 ± 10.39 | 4.20 ± 8.40 | 0.493 | |||
| D3 Continence | 1.59 ± 7.27 | 1.96 ± 8.08 | 0.882 | |||
| D4 Eating | 2.38 ± 10.91 | 1.47 ± 6.06 | 0.760 | |||
| D5 Meal preparation | 7.29 ± 20.17 | -5.13 ± 32.90 | 0.222 | |||
| D6 Telephoning | 10.71 ± 29.95 | 6.25 ± 19.36 | 0.607 | |||
| D7 Going on an outing | -2.58 ± 26.46 | 2.06 ± 17.24 | 0.540 | |||
| D8 Finance and correspondence | 1.75 ± 28.27 | 2.94 ± 34.48 | 0.910 | |||
| D9 Medications | 3.17 ± 17.97 | 11.11 ± 37.09 | 0.399 | |||
| D10 Housework | -3.57 ± 17.79 | 8.33 ± 25.17 | 0.097 | |||
| D11 Leisure | 0.00 ± 55.28 | 4.76 ± 17.82 | 0.760 | |||
| Chinese Version-Mini-Mental State Examination (C-MMSE) | ||||||
| Total Score | 0.86 ± 2.85 | 1.94 ± 3.09 | 0.269 | |||
| Clinical Dementia Rating Scale (CDR) | ||||||
| Stage | -0.24 ± 0.33 | 0.09 ± 0.26 | 0.268 | |||
ap-value was comparing the score changes at week-24 between DTD group and placebo group by independent sample t-tests
Values are given as mean ± S.D. Values in DTD group and placebo group are the score changed in the same group between week 24 and baseline (score at week-24 minus score at the baseline)
Table 2: Outcome of DTD group and placebo group at week 24.
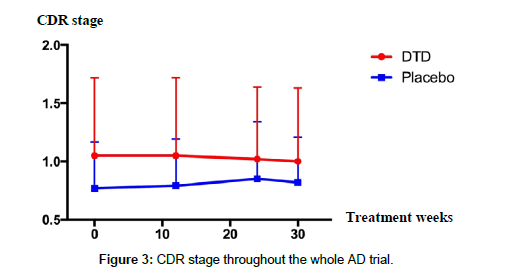
In the secondary analysis, there were no significant differences in the change of total score and sub-scores of C-DAD or C-MMSE at week 24 (Table 2). A trend of slowing the disease progress was observed in the DTD group in the CDR stage at the end of the whole trial; however, the change was not statistically significant (Table 3 and Figure 3).
| Parameter | Week 12 | Week 24 | Week 30 | |||
|---|---|---|---|---|---|---|
| DTD | Placebo | DTD | Placebo | DTD | Placebo | |
| ADAS-cog Total score | -0.81 ± 4.30 | -1.00 ± 3.14 | -2.05 ± 5.26 | -0.59 ± 2.98 | -1.90 ± 5.31 | -0.12 ± 3.31 |
| p-value=0.880 | p-value=0.315 | p-value=0.235 | ||||
| C-DAD Total score |
2.68 ± 9.38 | 2.20 ± 12.99 | 3.35 ± 7.95 | 4.42 ± 12.91 | 1.85 ± 7.31 | 2.18 ± 11.08 |
| p-value=0.918 | p-value=0.755 | p-value=0.913 | ||||
| C-DAD D10 Housework |
-1.98 ± 24.14 | -1.47 ± 25.72 | -3.57 ± 17.79 | 8.33 ± 25.17 | 1.59 ± 23.07 | 0.98 ± 24.63 |
| p-value=0.950 | p-value=0.097 | p-value=0.938 | ||||
| C-MMSE Total score |
1.00 ± 2.88 | 1.71 ± 2.62 | 0.86 ± 2.85 | 1.94 ± 3.09 | 1.43 ± 2.06 | 1.35 ± 3.08 |
| p-value=0.439 | p-value=0.269 | p-value=0.929 | ||||
| CDR stage | 0.00 ± 0.16 | 0.03 ± 0.12 | -0.24 ± 0.33 | 0.09 ± 0.26 | -0.48 ± 0.31 | 0.06 ± 0.17 |
| p-value=0.532 | p-value=0.268 | p-value=0.214 | ||||
ap-value was comparing the score changes at different time point between DTD group and placebo group by independent sample t-tests
Values are given as mean ± S.D. Values in DTD group and placebo group are the score changed in the same group between different time point and baseline (score at different time point minus score at the baseline)
Table 3: Further analyses of the general condition throughout the whole trial.
As for withdrawal and adverse events, six patients (2 (9.5%) [DTD] vs. 4 (23.5%) [Placebo], p=0.239) discontinued treatment after randomization. Among these 6 patients, 2 withdrew because of AE (1 in each group). During the treatment phase, two patients (9.5%) in the DTD groups and two patients (11.8%) in the placebo group had serious AE: one patient had hyperglycaemia (placebo), one had skin rashes (placebo), one had cramp (DTD), and one fell (DTD). Liver and renal functions were normal in the two groups at the end of treatment. No deaths were recorded during the trial. AE were reported by at least 5% of patients in each group, as shown in Table 4. Nausea and vomiting may be a possible AE for DTD.
| Adverse events | No. of patients (%) | |
|---|---|---|
| DTD(N=21) | Placebo (N=17) | |
| Joint pain | 1 (4.8) | 4 (23.5) |
| Back pain | 0 (0.0) | 3 (14.3) |
| Skin rashes | 2 (9.5) | 2 (11.8) |
| Abdominal pain | 1 (4.8) | 2 (11.8) |
| Nausea and Vomiting | 3 (14.3) | 1 (5.9) |
| Diarrhea | 1 (4.8) | 2 (11.8) |
Table 4: AE reported by >10% of AD patients in each group.
Discussion
In this study, results did not confirm the hypothesis that DTD improves the cognitive and daily activity including motor abilities of AD patients, as represented by the total score of ADAS-cog and total score of C-DAD. However, results did reveal a trend, which suggests an effect. A number of factors could explain the failure of this study to capture the beneficial effect of DTD.
Analyses revealed a trend of reduced cognitive impairment in the ADAS-cog total scores, and a trend of improvement in daily activity was suggested by C-DAD total score. Also, a trend of improvement in ideational praxis and comprehension is suggested by changes in the ADAS-cog sub score. Discontinuation due to AE occurred with the same frequency in the DTD group (1 patient) as in the placebo group (1 patient). DTD was well tolerated.
Although these trends suggest that DTD may be effective, this study has not fully captured its beneficial effect. Several factors could explain the failure to see significant results, as are described below.
Length of study
The most likely reason is the length of study. TCM works slowly in general. Its effect is gentler as well as more comprehensive compared to pharmaceuticals. Besides, AD is a slow, progressive disease; it may not be reasonable to see a protective effect, in a relatively short treatment period.
Sample size
The sample size in this study may have been too small to detect the effect of DTD. In fact, dementia patients in Hong Kong are much likely to present as a mixed dementia of AD and vascular dementia. It is difficult to recruit pure AD subjects. We should increase the sample size by either extend the recruitment period, or conduct in a multi-center design to increase the chance recruiting AD subjects.
Severity of AD
We aimed at recruiting mild to moderate AD patients, but most of the recruited subjects were only in mild condition. In fact, it is quite difficult to recruit AD patients in Hong Kong. AD patients must get the support and approval from their caregivers in order to participate in our clinical trial. As a number of moderate AD patients in Hong Kong have been sent to nursing homes, their caregivers often refuse to support them participating in an out-patient clinical trial. Future clinical trials carried out in a nursing home or hospital could be a possible way to recruit more moderate or even advanced AD patients.
Sensitivity of assessment tool
While some improvement trend in cognitive had been observed by ADAS-cog and CDR, it is strange that this trend was not supported by the total score of C-MMSE. C-MMSE is one of the most commonly used assessment tools for dementia; it is used as a standard measurement in most disciplines. It is doubtful that AD patients had already developed learning effect on C-MMSE even before they participated in our trial. This may have reduced the assessment accuracy of C-MMSE. A more sensitive, lesser used but valid cognitive assessment scale, like the Montreal Cognitive Assessment (MoCA) [30], could be used to replace C-MMSE in the future study.
Adverse events
For the AE, there were two more patients in DTD group (3 patients) who had nausea or vomiting compared to the control group (1 patient). All four patients complained about the bitter taste of the medicine. Although both the DTD granules and placebo granules had identical smell, they differed in bitterness and the tolerance of bitter differs among people. Thus, it appears worthwhile to try to make the taste of DTD more palatable. This may also reduce the AE of nausea and vomiting.
Conclusion
This randomized trial was the first rigorous testing of DTD for the treatment of AD patients. DTD was safely tolerated with few side effects. Although there were no significant differences in the primary and secondary outcomes, an improvement trend was observed throughout the trial, as revealed in chances in the ADAS-cog total scores, C-DAD total scores and CDR stages. At least five factors could explain the failure of these trends to be significant, particularly length of trial, size of trial, stage of AD, palatability of the drug, and sensitivity of the scoring system. Given the limitation but with the safety of DTD and its centuries of use in TCM, a further modified pilot RCT is suggested to retest the potential clinical effects of DTD in AD therapy. Currently, there is no evidence to support the efficacy of DTD to use as a single treatment for AD.
Declaration
We certify that we have each made a substantial contribution so as to qualify for authorship and that we have approved the contents.
Acknowledgement
This work is supported by an anonymous donation clinical research grant (GDS 0216), PuraPharm International (H.K.) Ltd. and S.H. Ho Foundation. This study was also support by HMRF 12132091 from the Food and Health Bureau, Hong Kong Government, research grants (RC-IRMS/15-16/04, FRG I/15-16/042 and FRG II/15-16/034) and Mr. & Mrs. Ko Chi Ming Centre for Parkinson’s Disease Research from Hong Kong Baptist University. The authors would like to thank Dr. Feng Sun, Ms. Vicky Keng from Clinical Division, School of Chinese Medicine, Hong Kong Baptist University and Hong Kong Alzheimer’s Disease Association for their great support on this clinical study. The authors would also like to thank Dr. Martha Dahlen for her English editing of this manuscript.
Availability of Data and Materials
The dataset supporting the conclusions of this article is included within the article and its Additional file 2.
References
- Cummings JL (2004) Alzheimer's disease. New England Journal of Medicine 351: 56-67.
- Hampel H, Prvulovic D, Teipel S, Jessen F, Luckhaus C, et al (2011) The future of Alzheimer's disease: The next 10 years. ProgNeurobiol 95: 718-728.
- Qiu C, Kivipelto M (2009) von Strauss E: Epidemiology of Alzheimer's disease: Occurrence, determinants and strategies toward intervention. Dialogues in Clinical Neuroscience 11:111-128.
- Brookmeyer R, Johnson E, Ziegler-Graham K, Arrighi HM (2007) Forecasting the global burden of Alzheimer's disease. Alzheimers Dement 3:186-191.
- (2013) Alzheimer's disease facts and figures. Alzheimers Dement 9:208-245.
- Massoud F, Leger GC (2011) Pharmacological treatment of Alzheimer disease. Canadian Journal of psychiatry Revue canadienne de psychiatrie 56:579-588.
- Kelley BJ, Knopman DS (2008) Alternative medicine and Alzheimer disease. Neurologist 14:299-306.
- Ho YS, So KF, Chang RC (2011) Drug discovery from Chinese medicine against neurodegeneration in Alzheimer's and vascular dementia. Chinese Medicine 6:15.
- Geriatric Society of the Traditional Chinese Medicine Society IMSotTCMS (1995) Diagnosis and rating standard of geriatric dementia. Chinese Journal of Rural Medicine and Pharmacy 2:19-20.
- Hui ML CK, Li M (2011)A literature review on the treatment of Chinese medicine in neurodegenerative diseases through supplying kidney Qi, promote blood circulation and Dispel Phlegm. Hong Kong Chinese Medicine Journal 6:35-40.
- Li F (2002) Chinese medicine formulae. Beijing: People's Medical Publishing House.
- Yuan WL HJ, Luo CM (2000) Review of Alzheimer disease treated by Chinese medicine. Modern Journal of Integrated Traditional Chinese and Western Medicine 9:203-205.
- Yu SMLL, Wang P (2007)Thetheory of dementia treated by Ditan Decoction. Fujian Journal of TCM 38:49-50.
- Liu LYR, Yan C, Dong H (2010) Effects of modified Ditandecoction on the neural degeneration and neuroethology in model mice with learning defect. Henan Traditional Chinese Medicine 30:553-554.
- Liu LLY, Jiang YM, Yu SM (2010) Experimental study on effect of Ditan decoction on ethology in model rats with Alzheimer's disease. Modern Journal of Integrated Traditional Chinese and Western Medicine 19:3154-3158.
- Liu LJY, Yu SM, Li Y (2009) Effect of Ditan decoction on central cholinergic system in model rats with Alzheimer’s disease. Hubei Journal of TCM 31:10-11.
- Liu LDH, Jiang YM, Yu SM (2010) Effects of Ditandecoction on learning and memory function in multiple models. Chinese Journal of Guang Ming Chinese Medicine 25:1364-1367.
- Chua KK, Chen LL, Liu LF, Kumar DS, Lu JH, et al. (2014) Efficacy of classic Chinese medicine formula Ditan Decoction for Alzheimer's disease. Chin J Integr Med.
- Chua KK, Wong A, Kwan PW, Song JX, Chen LL, et al. (2015) The efficacy and safety of the Chinese herbal medicine Di-Tan decoction for treating Alzheimer's disease: Protocol for a randomized controlled trial. Trials 16:199.
- McKhann G, Drachman D, Folstein M, Katzman R, Price D, et al. (1984) Clinical diagnosis of Alzheimer's disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology 34:939-944.
- Zheng XY (2002)Theguidance for clinical research of New Chinese Herbal Medicine. Beijing: Chinese Medical Scientific Publishers.
- Hughes CP, Berg L, Danziger WL, Coben LA, Martin RL (1982) A new clinical scale for the staging of dementia. The British Journal of Psychiatry: The Journal of Mental Science 140:566-572.
- Wong MT, Ho TP, Ho MY, Yu CS, Wong YH, et al. (2002) Development and inter-rater reliability of a standardized verbal instruction manual for the Chinese Geriatric Depression Scale-short form. Int J Geriatr Psychiatry 17:459-463.
- Tang XD,BianLQ, GaoR (2009) Exploration into the preparation of placebos used in Chinese medicinal clinical trial.Chinese Journal of Integrated Traditional and Western Medicine 29:656-658.
- Stern RG, Mohs RC, Davidson M, Schmeidler J, Silverman J, et al. (1994) A longitudinal study of Alzheimer's disease: Measurement, rate and predictors of cognitive deterioration. The American journal of psychiatry 151:390-396.
- Graham DP, Cully JA, Snow AL, Massman P, Doody R (2004) The Alzheimer's disease assessment scale-cognitive subscale: Normative data for older adult controls. Alzheimer Diseaseand Associated Disorders 18:236-240.
- Chiu HFLH, Chung WS, Kwong PK (1994) Reliability and validity of the Cantonese version of the mini-mental state examination-a preliminary study. Journal of Hong Kong College of Psychiatrists 2:25-28.
- Soto ME, Andrieu S, Cantet C, Reynish E, Ousset PJ, et al. (2008) Predictive value of rapid decline in mini mental state examination in clinical practice for prognosis in Alzheimer's disease. Dementia and Geriatric Cognitive Disorders 26:109-116.
- Mok CC, Siu AM, Chan WC, Yeung KM, Pan PC, et al. (2005) Functional disabilities profile of Chinese elderly people with Alzheimer's disease - a validation study on the Chinese version of the disability assessment for dementia. Dementia and Geriatric Cognitive Disorders 20:112-119.
- Dong Y, Lee WY, Basri NA, Collinson SL, Merchant RA, et al. (2012) The Montreal cognitive assessment is superior to the mini-mental state examination in detecting patients at higher risk of dementia. Int Psychogeriatr 24:1749-1755.
Relevant Topics
- Advanced Parkinson Treatment
- Advances in Alzheimers Therapy
- Alzheimers Medicine
- Alzheimers Products & Market Analysis
- Alzheimers Symptoms
- Degenerative Disorders
- Diagnostic Alzheimer
- Parkinson
- Parkinsonism Diagnosis
- Parkinsonism Gene Therapy
- Parkinsonism Stages and Treatment
- Stem cell Treatment Parkinson
Recommended Journals
Article Tools
Article Usage
- Total views: 13754
- [From(publication date):
August-2016 - Aug 30, 2025] - Breakdown by view type
- HTML page views : 12720
- PDF downloads : 1034