A Successful Treatment of Infant Who Had Two Significant Intracranial Hemorrhage with Factor X Replacement: Case Report
Received: 28-May-2018 / Accepted Date: 20-Aug-2018 / Published Date: 27-Aug-2018 DOI: 10.4172/2572-4983.1000163
Keywords: Factor X deficiency; Infant; Intracranial hemorrhage; Prothrombin complex concentrate
Introduction
Factor X is a vitamin K-dependent plasma glycoprotein that has a critical role in blood coagulation cascade [1]. Congenital factor X deficiency is a rare autosomal recessive bleeding disorder [2]. The bleeding tendency can be once or recurrent and can be ranged from mild to severe according to the level of the factor X in the blood. The most common symptoms are epistaxis, gingival bleeding, and menorrhagia [2,3]. In addition, infants and neonates with severe factor X deficiency are at high risk of intracranial hemorrhages (ICH) [4]. Here, we report an infant with severe factor X deficiency who developed intracranial hemorrhage. He was treated with fresh frozen plasma (FFP) and prothrombin complex concentrate (PCC). After control of bleeding and normalization of coagulation profile, treatment was maintained with PCC.
Case Report
A full-term baby boy, with weight 3.1 kg admitted to our neonatal intensive care unit at age of 21 days transferring from peripheral hospital due to severe bleeding, frequent apnea and convulsion. The baby was born to a first-degree consanguineous couple. The mother is 21 years old. Antenatal ultrasound scan was normal with no complications during pregnancy. On the physical examination, he had dilated pupils, tense and bulging anterior fontanel.
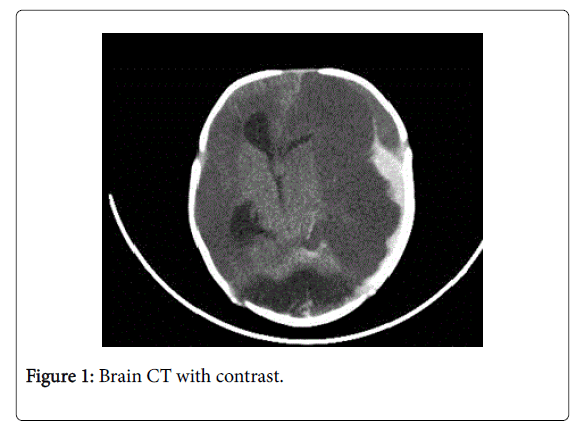
Upon the admission, the baby was intubated and connected to mechanical ventilator. Phenobarbitone started to control convulsion. Also, antibiotics with ampicillin and cefotaxime initiated for suspected meningitis. The baby had a history of acute umbilical bleeding at age 3 days of life and intracranial hemorrhage that developed at 11 days of life. In addition, there is no history of preceding trauma or injury; however there is a family history of bleeding disorder. Thus, an urgent cerebral computed tomography (CT) scan was done at 23 days of life that showed large intracranial areas of fluid density likely left-side subdural hematoma with substantial mid line shift, and hydrocephalus consistent with acute hemorrhage at the same time there are cerebral atrophy (Figure 1).
Bilateral large intra-cranial areas of fluid density (likely extra-axial) showing left temporo-parietal fresh blood density (likely sub-dural) (about 11.7 mm in maximum thickness). Subsequent cerebral atrophy and mass effect (marked left lateral ventricle compression and shift of midline structures to the right side).
Patient had low Glasgow coma scale and bilateral dilated pupils not reacting to light stimulus. Thus, an emergency craniotomy with evacuation of large subdural hematoma was done and due to dramatic swelling and severe herniation of the brain, craniotomy was extended and the bone-laps were removed and saved in the freezer. CT scan was repeated at age of 24 days to evaluate post craniotomy statues in left parietal area (Figure 2).
The coagulation studies revealed a prolonged activated partial thromboplastin time of 87.1 sec and prothombin time 114.6 sec, and INR 8.8. Specific anticoagulation levels were as follows: factor X: 2%, factor VII: 83%, factor II: 87.7%, factor V: 113%, factor IX: 69.7%. All are within the normal range except factor X. Thus inherited factor X deficiency was confirmed.
Post craniotomy status in left parietal area.
• There is redemonstration of diffuse hypodensity involving the cerebral hemisphere bilaterally.
• Interval evacuation of left subdural hematoma with linear areas of hyperdensity/blood products seen in left parietal area with air foci.
• There is an interval development of right parietal cerebral hematoma measuring 2.3 x 2.0cm.
• Diffuse posterior parieto occipital subgaleal soft tissue swelling with air foci in left parietal area.
• Rest of the disease is same as before.
• Right subdural hematoma (1.3 cm) and midline shift to the contralateral side.
• There is interval improvement of the intraventricular hematoma.
• There is redemonstration of diffuse hypodensity involving the cerebral hemisphere bilaterally.
• Left subdural fluid collection, likely related to chronic hematoma.
• Interval improvement of the previously noted diffuse posterior parieto occipital subgaleal soft tissue swelling with air foci in left parietal area.
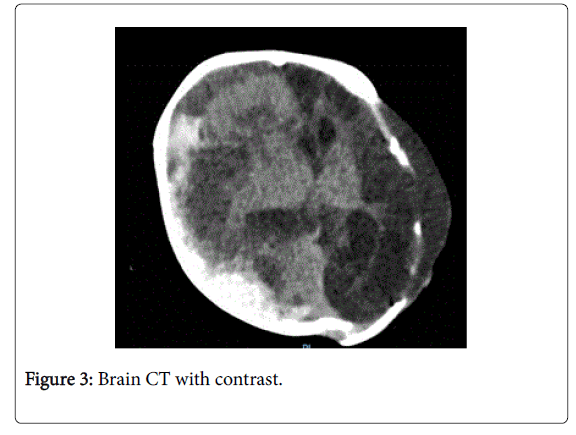
Others laboratory results were as follow: Htc: 26.2%, Hb: 8.2 g/dl, platelet count: 252 103/mm3, retic: 0.27, white blood cell count: 18.19103/mm3 (with 52.3% segmented neutrophils, 30% lymphocytes, and 16.8% monocytes). Fibrinogen and C-reactive protein levels were 380 mg/dl and16.5 respectively. Liver and renal function tests were normal. After initiation of FFP, activated partial thromboplastin time and prothrombin time prolongation were normalized. We continued on regular infusions with FFP 15 ml per kg three times as treatment for acute bleeding. Later at age of 48 days, the hemoglobin dropped to 5 g/dl. An urgent brain CT scan done and revealed the developing again of second intracranial hemorrhage in spite of regular replacement of FFP (Figure 3). At age of 84 days, the infants started on PCC (Factor X concentrate, Behring®) which contains 30–60 IU/ml of factor X 40 IU per kg per day daily for 14 days. At age 87 days of life, cranioplasty was done and the removed bone flaps were reinserted. Later, broviac line was inserted and PCC shifted to twice weekly as secondary prophylaxis due to high risk of bleeding. Currently patient gives a good eye contact and behaves normally. Feeding orally, and gain in length and weight is normal. He is moving all limbs symmetrically and does not show any signs of paresis. He has dilatation of the cerebral ventricles probably due to parenchyma loss secondary to cerebral insults, but the head is growing within normal limits. Regular MRI follow up is planned in future.
Discussion
Factor X deficiency is a rare autosomal recessive bleeding disorder [1]. It was firstly described by Tefler in 1956 [5]. Factor X is a glycoprotein that has a crucial role in blood coagulation cascade. Factor X is a vitamin K-dependent plasma protein with half-life around 40 hours [6]. The initial manifestation of congenital factor X deficiency is bleeding and severe elevations of coagulations profile. Intracranial hemorrhage secondary to congenital factor X deficiency considered one of serious complications and has been described in a few patients between 9-26% of all infants with factor X deficiency [4,7,8]. ICH usually occurs in the first month of life; median 9.7 days of life [9]. Our patients had initially bleeding from umbilical at age of 3 days of life and age of 11 days of life he developed the first episode of ICH. Later at age of 48 days of life and in spite of regular treatment with FFP, hemoglobin was drop to 5 and he had a second episode of intracranial hemorrhage. Additionally, there are severe elevations in activated partial thromboplastin time and prothrombin time. The diagnosis was confirmed after measuring of factor X. Congenital factor X deficiency can be categorized into three types: mild, moderate and sever deficiency. Mild deficiency if factor X level between 10-40%, moderate deficiency if level of factor X>40% and severe deficiency if level of factor X<10% [10,11]. In the literature, the majority of reported cases have severe factor X deficiency [9,12]. According to these classifications, our patient had a major factor X deficiency that manifests by major and recurrent spontaneous bleeding which with level of factor X=2% and the two episode if intracranial hemorrhage. S. El Kalla, et al. reported two cases who developed a twice ICH. The first case in which ICH developed at age of 3 days old after that secondary prophylaxis was not started until a second ICH developed at age of 4 months. The second case ICH developed at age of 3 days and in spite of regular replacement, a second episode of ICH developed [13]. Eric Sandler, report an infant who developed a three times of ICH in first 6 months but later, after initiation of secondary prophylaxis no more bleeding [14]. Bahri Ermis, also reported a case of infants who developed ICH twice despite the appropriate prophylaxis [15]. The treatment options for factor X deficiencyare depend on severity, locations of bleeding, risk of recurrence. Before FFP [16], and PCC [17] are only available options for treatment. Recently, purified factor X is available in market, but still not available in our hospital. The target from the treatment is to keep level of factor X between 10-40% as this is usually sufficient to keep hemostasis [18]. FFP is widely used for treatment especially in acute phase of bleeding. FFP has several complications. The most common complications, allergic reactions, transfusion infection and transfusion related acute lung injury, and fluid overload [16,19,20]. PCC contains significant amounts of factor X. Although, it is a drug of choice for long term especially if factor X alone is not available, it should be used with caution as risk of thrombosis either arterial or venous thrombosis may occur. This is due to different amount form specific clotting factors [12,16]. The decision if secondary prophylaxis should be initiated or not depends on availability of wither PCC, or purified factor X concentrate, severity of disease, bleeding frequency, and risk of developing long-term disability after bleeding such as ICH [17]. Our patient initially received FFP three times a day as treatment for acute bleeding. Factor X level at that time is in range between 30-50%. Later, after closing of skull opening, the PCC initiated with 40 units per kg per day daily for 14 days; Factor X P Behring® which contains 30-60 IU/ml of factor X. Due to risk of bleeding, we keep monitoring of factor X to be maintain between 25-40%. Our patient had severe factor X deficiency with recurrent ICH. Therefore the secondary prophylaxis must be initiated. Due to unavailability of factor X alone, we started him on PCC. The half-life of Factor X P Behring® is between 24 - 40 hours making it suitable for twice weekly prophylaxis. Our patient received 40 units per kg per day twice weekly. We got the approval from our hospital to bring purified factor X by direct purchasing process. Once it is available, we will start on 40 units per kg per day and adjust the dose according to the level of factor X. He is wake, sucking normally, and no more convulsions on phenobarbitone.
Conclusion
A possible factor deficiency should be considered when the activated partial thromboplastin time and prothrombin time prolongation persist despite vitamin K administration or in case of early ICH that is not related to trauma or injury. In these patients, early diagnosis is crucial for the prevention of central nervous system bleeding by aggressive replacement therapy. In addition the presence of family history can help to achieve the diagnosis rapidly. Our patient has a cousin with same diagnosis. Therefore, a proper educations to family with a history of factor X deficiency and early diagnosis with secondary prophylaxis if need it are very important steps to avoid the complications especially ICH and long term neurological sequences related to these complications.
References
- Uprichard J, Perry DJ (2002) Factor X deficiency. Blood Rev 16: 97-110.
- Peyvandi F, Mannucci PM, Lak M, Abdoullahi M, Zeinali S, et al. (1998) Congenital factor X deficiency: spectrum of bleeding symptoms in 32 Iranian patients. Br J Haematol 102: 626-628.
- Palla R, Peyvandi F, Shapiro AD (2015) Rare bleeding disorders: diagnosis and treatment. Blood 125: 2052-2061.
- Escobar MA (2013) Advances in the treatment of inherited coagulation disorders. Haemophilia 19: 648-659.
- Telfer R, Denson KW, Wright DR (1956) A new coagulation defect. Br J Hematol 2: 308-316.
- Hidehiko S (1984) Normal Hemostatic Mechanisms. In: Ratnoff O, Forbes C (eds.) Disorders of Hemostasis. Grune & Stratton, Orlando, USA, p: 32.
- Machin SJ, Winter MR, Davies SC, Mackie IJ (1980) Factor X deficiency in the neonatal period. Arch Dis Child 55: 406-408.
- Girolami A, Molaro G, Calligaris A, De Luca G (1970) Severe congenital Factor X deficiency in a 5-month old-child. Thromb Diath Haemorrh 24: 175-184.
- Herrmann FH, Auerswald G, Ruiz-Saez A, Navarrete M, Pollmann H, et al. (2006) Greifswald Factor XDSG. Factor X deficiency: clinical manifestation of 102 subjects from Europe and Latin America with mutations in the factor 10 gene. Haemophilia 12: 479-489.
- Mota L, Shetty S, Idicula-Thomas S, Ghosh K (2010) Molecular basis of factor X deficiency cases from India. Haemophilia 16: 693-697.
- Peyvandi F, Menegatti M, Palla R (2013) Rare bleeding disorders: worldwide efforts for classification, diagnosis, and management. Semin Thromb Hemost 39: 579-584.
- Bowles L, Baker K, Khair K, Mathias M, Liesner R (2009) Prophylaxis with prothrombin complex concentrate in four children with severe congenital factor X deficiency. Haemophilia 15: 401-403.
- el Kalla S, Menon NS. Neonatal Congenital Factor X Deficiency (1991) Pediatr Hematol Oncol 8: 347-354.
- Sandler E, Gross S (1992) Prevention of Recurrent Intracranial Hemorrhage in a Factor X-Deficient Infant. Am J Pediatr Hematol Oncol 14: 163-165.
- Ermis B, Ors R, Tastekin A, Orhan F (2004) Severe congenital factor X deficiency with intracranial bleeding in two siblings. Brain Dev 6: 137-138.
- Perry DJ (1997) Factor X and its deficiency states. Haemophilia 25: 159 -172.
- Kouides PA, Kulzer L (2001) Prophylactic treatment of severe factor X deficiency with prothrombin complex concentrate. Haemophilia 7: 220-223.
- Jeanne LM (1987) Diseases of Coagulation: The Fluid Phase. In: Nathan DG, Oski FA (eds.). WB Saunders, Philadelphia, USA. pp: 1322-1323.
- Furie B, Furie BC (1988) The molecular basis of blood coagulation. Cell 53: 505-518.
- Karimi M, Vafafar A, Haghpanah S, Payandeh M, Eshghi P, et al. (2012) Efficacy of prophylaxis and genotype-phenotype correlationin patients with severe Factor X deficiency in Iran. Haemophilia 18: 211-215.
Citation: Al-Mouqdad M, Abdelrahim A, Kahlon BMM, Almahmoud L and Asfour S (2018) A Successful Treatment of Infant Who Had Two Significant Intracranial Hemorrhage with Factor X Replacement: Case Report. Neonat Pediatr Med 4: 163. DOI: 10.4172/2572-4983.1000163
Copyright: © 2018 Al-Mouqdad M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 5036
- [From(publication date): 0-2018 - Dec 07, 2025]
- Breakdown by view type
- HTML page views: 4141
- PDF downloads: 895