Case Report Open Access
Advanced Gastric Cancer Identified during Acute Myocardial Infarction: Report of a Case
Takehiro Wakasugi, Yoichi Matsuo* and Hiromitsu Takeyama
Department of Gastroenterological Surgery, Nagoya City University Graduate School of Medical Sciences, Nagoya, Japan
- *Corresponding Author:
- Yoichi Matsuo
Department of Gastroenterological Surgery
Nagoya City University, Nagoya 4678601, Japan
Tel: +81-52-853-8226
Fax: +81-52-842-3906
E-mail: matsuo@med.nagoya-cu.ac.jp
Received date: September 15, 2014; Accepted date: October 29, 2014; Published date: November 03, 2014
Citation: Wakasugi T, Matsuo Y and Takeyama H (2014) Advanced Gastric Cancer Identified during Acute Myocardial Infarction: Report of a Case. J Gastrointest Dig Syst 4:233. doi:10.4172/2161-069X.1000233
Copyright: © 2014 Wakasugi T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
A 71-year-old man was admitted for acute myocardial infarction. The occluded right coronary artery was immediately reperfused by balloon angioplasty during percutaneous coronary intervention and plans were made to stent the left anterior descending artery and circumflex branch at a later date. Anti-platelet therapy was started after intervention. Ten days after intervention, the patient developed hematemesis. An advanced gastric cancer was identified as the source of the bleed. Although an urgent gastrectomy was indicated, the remaining coronary artery stenoses complicated surgical management.
The decision was made to perform the second intervention prior to gastrectomy and to treat the remaining stenoses with bare metal stents. Two months after the second intervention, a total gastrectomy was performed under general anesthesia. No severe cardiac complications occurred during the postoperative period.
Keyword
Gastric cancer; Acute myocardial infarction; Revascularization
Introduction
As the average age of the general population increases, the number of patients with gastric cancer who require coronary revascularization is also increasing [1]. Management of these patients presents several problems, including the priority of surgical resection of the neoplasm versus coronary revascularization [1].
Here, we present a case of advanced gastric cancer identified during the treatment of acute myocardial infarction. We review the recent literature on this topic and consider various strategies for the treatment of patients with gastric cancer who require coronary revascularization.
Case Report
A 71-year-old man presented to our hospital with sudden onset chest pain. His medical history was significant for right nephrectomy due to renal cell carcinoma and grafting of an abdominal aneurysm 4 years prior to presentation. His chest pain was accompanied by ischemic changes on electrocardiogram and elevated cardiac markers. He was thus admitted for acute coronary syndrome and emergent cardioangiography was performed. Three branch stenoses were observed, including a 100% occlusion of the right coronary artery (RCA). Percutaneous coronary intervention (PCI) was performed. The RCA diameter was found to be greater than 5 mm and multiple sequential thrombi were observed, hence balloon angioplasty was performed rather than stent placement. This intervention improved the RCA occlusion to 25%. Plans were made to stent the remaining left anterior descending artery (LAD) and circumflex branch (CX) stenoses (90% and 99%, respectively) at a later date (Figure 1). Dual anti-platelet therapy was initiated after the procedure.
On the tenth day after PCI, the patient developed hematemesis that caused acute blood loss anemia. The hemoglobin level was decreasing from 13.3 g/dL to 8.9 g/dL and the patient required 4 units of red blood cell transfusion. An urgent gastroscopy revealed an oozing type 3 tumor at the upper posterior gastric wall. Biopsy specimens were positive for adenocarcinoma. In order to prevent more bleeding, anti-platelet therapy was immediately stopped and heparin was administered instead. Since then the progression of anemia was not found. An enhanced CT scan showed thickening of the upper posterior gastric wall with subserosal (SS) tumor invasion. No metastases were detected on imaging. He was diagnosed with clinical stage IIA gastric cancer according to the Japanese classification of gastric carcinoma (third English edition) [2].
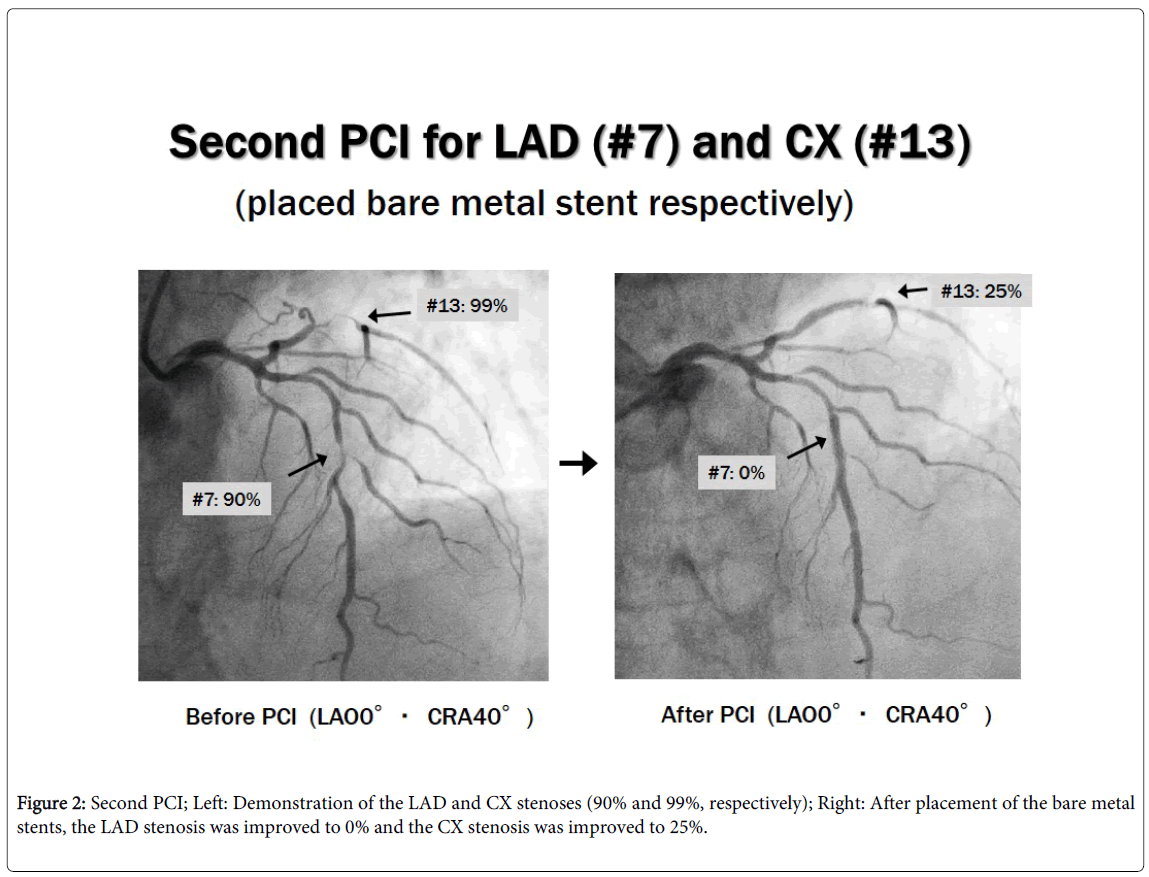
Although the patient required urgent total gastrectomy with curative intent for his advanced gastric cancer, he also required revascularization of the remaining LAD and CX stenoses. The decision was made to perform coronary revascularization prior to surgical resection in order to reduce perioperative cardiac risk. Bare metal stents were placed in the LAD and CX, improving both occlusions to 0% and 25%, respectively (Figure 2).
Total gastrectomy was performed two months after the second PCI. A Roux-en-Y anastomotic reconstruction was performed under general anesthesia with intra-abdominal balloon pump on stand-by. Lymph node dissection was limited to the level of D1 to reduce surgical damage, and the spleen was preserved. The operation lasted 370 minutes and the total blood loss was 489ml. The patient was admitted to the intensive care unit for post-surgical monitoring and he had no recurrence of myocardial infarction or heart failure. He was discharged on post-operative day 18.
Pathological review of the resected specimens revealed a poorly-differentiated adenocarcinoma extending beyond the serosa. Lymph node metastasis was found in 1 of 4 nodes. The final diagnosis was stage IIIA gastric cancer. Adjuvant chemotherapy was not administered due to his comorbid conditions.
Discussion
Both gastric cancer and acute myocardial infarction are life-threating conditions and management of patients with both of these problems can be difficult. That is, which is the highest priority treatment, surgical resection or coronary revascularization? If surgical resection is performed prior to coronary revascularization, the perioperative cardiac risk is much higher. On the other hand, if coronary revascularization is performed prior to surgical resection, surgery must be delayed which may influence postoperative outcomes. Furthermore, the risk of gastric bleeding due to anti-platelet therapy will continue until the tumor is removed.
McFalls et al. reported the results of a large randomized trial which found that routine coronary revascularization in patients with stable cardiac symptoms before elective vascular surgery should not be performed [3]. The American College of Cardiology (ACC)/ American Heart Association (AHA) 2007 guidelines on perioperative cardiovascular evaluation and care prior to noncardiac surgery state that PCI before noncardiac surgery does not prevent perioperative cardiac events, except in those patients for whom PCI is independently indicated for an acute coronary syndrome [4]. In our case, the advanced gastric cancer was found during the interval between staged PCI procedures and bleeding from the tumor ceased after discontinuation of anti-platelet therapy, thus, we decided to proceed with coronary revascularization prior to surgical resection.
Coronary revascularization can be achieved by either PCI or coronary artery bypass grafting (CABG). PCI is less invasive and offers the possibility of repeated interventions, so it is currently performed more frequently than CABG [5]. Japanese guidelines for perioperative cardiovascular evaluation and management of noncardiac surgery indicate that PCI is preferable to CABG in patients in whom the noncardiac disease has a poor prognosis and in those patients in poor general health, including those with bleeding diathesis [6]. We chose PCI for the remaining coronary stenoses by following these recommendations.
Compared to PCI, conventional CABG has the disadvantage of severe perioperative damage and high short-term morbidity [4]. Furthermore, it has been suggested that cardiopulmonary bypass might affect the spread of any coexisting tumors [7]. However, CABG has the advantage of long-term patency of the grafting vessels without the need for anti-platelet agents [5,8]. Moreover, off-pump CABG has become increasingly popular because it can be performed without cardiopulmonary bypass.
Revascularization by PCI can be achieved in several ways, including balloon angioplasty or placement of bare metal or drug-eluting stents. The type of PCI performed depends on the timing of any subsequent surgeries (Figure 3) [9]. We chose bare metal stent for the second PCI and performed total gastrectomy two months after the second PCI. Although no cardiac complication was found during the postoperative period, the gastric cancer might be progressed during the waiting time for the surgical resection. It might be preferable to reduce the waiting time for the surgery to one month.
Current strategies for coronary revascularization in patients who require noncardiac surgery are summarized in Figure 3. The revascularization strategy depends on the timing of surgery and both PCI and CABG (conventional or off-pump) should be considered. As progress is made in the further development of coronary stents and off-pump CABG, these strategies may change, and large randomized trials are required to determine the most appropriate management of these complicated cases.
References
- Darwazah AK (2012) Surgical management of coronary artery disease associated with malignancy. J Card Surg 27: 581-588.
- Japanese Gastric Cancer Association (2011) Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer 14: 101-112.
- McFalls EO, Ward HB, Moritz TE, Goldman S, Krupski WC, et al. (2004) Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 351: 2795-2804.
- Fleisher LA, Beckman JA, Brown KA, Calkins H, Chaikof E, et al. (2007) ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: executive summary. J Am CollCardiol 50: 1707-1732.
- Yokoyama H (2005) [Preoperative evaluation and management of patients with ischemic heart disease undergoing noncardiac surgery]. Nihon GekaGakkaiZasshi 106: 323-327.
- JCS Joint Working Group1 (2011) Guidelines for perioperative cardiovascular evaluation and management for noncardiac surgery (JCS 2008)--digest version. Circ J 75: 989-1009.
- Ochi M, Yamada K, Fujii M, Ohkubo N, Ogasawara H, et al. (2000) Role of off-pump coronary artery bypass grafting in patients with malignant neoplastic disease. JpnCirc J 64: 13-17.
- Dedeilias P, Nenekidis I, Koletsis E, Baikoussis NG, Hountis P, et al. (2010) Simultaneously performed off-pump coronary artery bypass grafting and colectomy: a case report. World J SurgOncol 8: 50.
- Satler LF (2004) Recommendations regarding stent selection in relation to the timing of noncardiac surgery postpercutaneous coronary intervention. Catheter CardiovascInterv 63: 146-147.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 14902
- [From(publication date):
November-2014 - Aug 16, 2025] - Breakdown by view type
- HTML page views : 10300
- PDF downloads : 4602