Research Article Open Access
An Orally-Bioavailable Glutathione-Based Hepatoprotective Compound in Experimental Acute Liver Injury: More Effective than Silymarin and YHK
Kantah MK1, Kumari A2, He F3, Sollano J4, Alagozlu H5, Min CH1, Lorenzetti A1, Morita Y1 and Marotta F1,6*1ReGenera Research Group for Aging-Intervention, Milano, Italy
2Institut de Biologie Intégrative et des Systèmes, Université Laval, Québec, QC, Canada
3Department of Nutrition and Food Hygiene, West China School of Public Health, Sichuan University, China
4Gastroenterology Department, University of Santo Tomas, Manila, The Philippines
5Medical Park Ankara Hospital, Gastroenterology Department, Ankara,Turkey
6Milano Medical-Gender Healthy Aging Unit by Genomics & Biotechnology, Milano, Italy
- *Corresponding Author:
- Prof. Francesco Marotta, MD, PhD
Milano Medical-Gender Healthy Aging Unit by Genomics and Biotechnology
Piazza Firenze, 12, 20154 Milano, Italy
E-mail: fmarchimede@libero.it
Received date: May 10, 2016; Accepted date: July 19, 2016; Published date: August 9, 2016
Citation: Kantah MK, Kumari A, He F, Sollano J, Alagozlu H, et al. (2016) An Orally-Bioavailable Glutathione-Based Hepatoprotective Compound in Experimental Acute Liver Injury: More Effective than Silymarin and YHK. J Gastrointest Dig Syst 6:462. doi:10.4172/2161-069X.1000462
Copyright: © 2016 Kantah MK, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License; which permits unrestricted use; distribution; and reproduction in any medium; provided the original author and source are credited.
Visit for more related articles at Journal of Gastrointestinal & Digestive System
Abstract
Many hepatoprotective non-chemical preparations have been recommended as complementary medicine for the treatment of liver disorders. We have utilized a well-defined model of liver injury to study the efficacy of GLU-9599 (a oral bioavailable glutathione-based compound added with selenium, L-cysteine and vitamin C) designed to support liver detox function. Wistar albino rats were divided into five groups. Group I represented the healthy control group; Groups II-IV were given carbon tetrachloride (CCl4) intragastrically. Rats were exposed to CCl4 after they had been pretreated for five days with either saline, silymarin extract, YHK (am herbal Japanese compound with putative hepatoprotective effects) or GLU-9599. After inducing hepatic damage, Group II served as control CCl4; Group III and IV were given silymarin (reference hepatoprotective) and YHK whereas Groups V was administered GLU-9599. Animals were sacrificed 24 hours after receiving CCl4. Liver enzymes and hepatic histology formed the basis for evaluating the efficacy of the treatments. Samples of livers were observed under microscope and electron microscopy (TEM) for the histopathological changes. Levels of marker enzymes such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST) significantly increased in CCl4 treated rats (Group II). Groups III, IV and V intoxicated with CCl4 and treated with the above hepatoprotective agents showed a significant decrease of the activities of these two enzymes (p<0.05). However, GLU-9599 provided a significantly stronger effect than YHK (p<0.05) and a trend improvement as compared to sylimarin. This was confirmed at histology and TEM whereas the others hepatoprotective compounds had only limited benefits on morphological abnormalities. In conclusion GLU-9599 could be a promising hepatoprotective compound of safe profile although extensive clinical studies are warranted.
Keywords
Acute liver injury; Oral GSH; Silymarin; YHK
Introduction
Liver is an important organ actively involved in metabolic functions and is a frequent target of a number of toxicants. Carbon tetrachloride (CCl4) has been widely used for inducing experimental hepatic damage due to free radicals generation during its metabolism by hepatic microsome, leading to lipid peroxidation and consequent liver damage [1-3]. Indeed, it is well documented that the hepatotoxicity of CCl4 is related to its reductive dehalogenation by cytochrome P450 2E1 [4] in the hepatic endoplasmic reticulum to the highly unstable trichloromethyl radical (•CCl3). This is subsequently converted into a trichloromethyl peroxyl radical (•OOCCl3) damaging cellular membranes [5]. The resulting hepatic injury occurs within 5 min after oral administration of CCl4 and is characterized by leakage of cellular enzymes into the blood stream and by liver necrosis and fibrosis [6-8]. Besides increased lipid peroxidation, CCl4 also depletes intracellular glutathione (GSH) levels, indicating that GSH loss might result from the detoxification of CCl4 by GSH conjugation [9].
Glutathione is a low-molecular-weight, water-soluble reduced-thiol and tripeptide molecule mostly concentrated in hepatocytes which modulates intracellular redox balance by preferentially binding electrophilic molecules [10]. This represents a key antioxidant by exerting a most important function in the detoxification of either endogenous metabolic products and xenobiotic compounds such as pollutants, heavy metals and drugs [11]. Indeed, depending on the availability of cysteine and the enzymatic activity of glutamate cysteine ligase, its rate of formation depends on its generation from GSH disulfide form or, on the contrary, its depletion reflects xenobiotics burden. Although restoration of GSH by de novo formation is usually a quick process, when the amount and rate of its destruction or consumption exceeds the regeneration rate of GSH, excess oxidated products are free to initiate hepatotoxicity by conjugating hepatic macromolecules [12].
The aim of the present study was to test the hepatoprotective properties of a GSH-based compound on CCl4-induced experimental liver injury model as compared to other hepatoprotective molecules.
Materials and Methods
Animal population and study design
Sixty adult male albino Wistar rats weighing 200-220 g and with identical numbers of male and female in each group. were used in the experiments. The animals had free access to normal standard chow diet (Rodent LaboChow, Purina) and tap water for 3 weeks. Throughout the experiment, the animals were housed, three per cage, in laminar flow cages maintained at 22 ± 2°C, 50-60% relative humidity and under a 12 hour light/dark lighting cycle. The animals were kept in these facilities for at least one week before the experiment. Animal care and treatment was carried out in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication no. 85-23, revised in 1996). The protocols used in this study were approved by the ethics committee of ReGenera Research Group.
Animals were randomly divided into 5 groups (I-V), each having 12 animals, Group uI served as normal control. As in vivo hepatotoxicity model, animals of Groups II-V were administered intraperitoneally a single dose of 2 ml/kg body weight of 50% CCl4 dissolved in olive oil (15% v/v) [7]. After CCl4 intoxication, Group II served as untreated control CCl4. Group III served as positive control and was given silymarin (20 mg/kg in water) once a day for 21 days. Group IV followed the same schedule but treated with YHK (panax pseudoginseng, eucommia ulmoides, glycyrrhiza radix, Kyotsu Jigyo, Tokyo, Japan) purchased from the market, at the dosage of 20 mg/kg and Group V was administered orally GLU-9599 (content per 100 mg: GSH 36 mg in a patented orally-bioavailable form, vitamin C 6 mg, Lcystein 7.4 mg, selenium 8 mcg, Olimed ltd, London, UK) at a dose of 20 mg/kg body weight per day diluted in 2 ml of water and instilled in the mouth, for a period of 21 days. To rule out the hepatotoxic effect of the anaesthetics employed, also the animals from the healthy control group were anesthetized.
Assessment of Liver Damage
Biochemical evaluation
After completing the treatment, rats were anesthetized with pentobarbital (50 mg/kg, ip), the abdomen was cut opened and the blood samples were collected from the aorta artery to be kept at room temperature for 90 min, and then centrifuged (12,000 rpm for 10 min at 4°C) to obtain serum.. This was finally was kept at -20°C until assay. Liver damage was assessed by testing serum levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) using a standard photometric method using a bichromatic analyzer (Integra 700, Roche Diagnostics).
Five-hundred milligrams of liver tissue was homogenized by employing a motor-driven Teflon-glass homogenizer with 5 up-anddown strokes in ice. The solution used for the homogenization consisted of 3.75 mL of 0.1 M sodium phosphate/0.005 M ethylendiamin- tetra-acetate (EDTA) buffer (pH 8.0) and 1.0 mL of 25% metaphosphoric acid. The homogenate was centrifuged at 60,000 X g for 60 min at 0°C t separate the acid supernatant. This supernatant was then employed for the assay of GSH and lipid peroxide (LPO). GSH and LPO were analyzed fluorometrically by the method of Hissin and Hilf [13] and Ohkawa [14], respectively by employing the supernatant prepared from liver samples.
Histological evaluation
The livers were perfused via the portal vein with 20 ml PBS to remove blood prior preservation in neutral buffered formalin and processing for paraffin embedding, following the standard microtechnique to fixation in 10% formalin. Staining was performed with haematoxylin and eosin as well as trichromic Mason´s technique and histopathological changes were semi-quantitatively assessed (scale ranging from 0: no changes to 6: maximum changes) by two experienced pathologists, blinded to the treatment received as for: steatosis, necrosis, inflammatory infiltrate, portal reaction, hepatocyte degeneration, Kupffer cell activity, vasodilatation, interstitial haemorrhage and cholestasis.
Transmission electron microscopy evaluation
For electron microscopic observation, the liver specimens were fixed with 2.5% glutaraldehyde and post-fixed in 1% osmium tetroxide. The tissue samples were dehydrated through a graded alcohol series and embedded in Epok 812. Ultrathin sections were cut on a Porter MT 5000 ultramicrotome with a diamond knife, then stained with uranyl acetate and lead citrate. The sections were examined at 75 kV under a Hitachi H-800 electron microscope.
Statistical analysis
All values are expressed as means ± SEM. Comparison between any two groups was performed using Student´s t-test. Statistically significant differences between groups were defined as P<0.05. Calculations were performed with the Microsoft Excel™ program.
Results
Biochemical changes
Results in Table 1 revealed a significant elevation of serum AST and ALT activity in CCl4 treated group (p<0.05), indicating, as expected, that CCl4 induced severe liver damage. As compared to untreated intoxicated controls, GLU-9599 significantly reduced the extent of AST and ALT elevation in the treated animals (p<0.05). Such effect was found to be more effective than what observed with silymarin and YHK (p<0.05), the latter yielding the weakest reduction of liver enzymes, although the two latter groups were statistically comparable.
| Group | Serum | Liver Tissue | ||
|---|---|---|---|---|
| AST (U/l) | ALT (U/l) | GSH (μg/g) | LPO (MDA nmol/g) | |
| Healthy control | 60.3 ± 0.9 | 70.3±0.8 | 3917.3±138.7 | 14.7±0.09 |
| CCl4 | 228.4 ±3.8* | 267.2±4.9* | 989.2±44.6* | 98.7±0.05* |
| CCl4 + sylimarin | 99.2±2.7§ | 121.1±3.9§ | 2598.6±89.9§ | 30.4±0.06§ |
| CCl4 + YHK | 112.6±3.2§ | 136.6±2.7§ | 2345.3±126.6§ | 33.5±0.02§ |
| CCl4 + GLU-9599 | 79.5±1.3** | 84.3±0.8** | 3878.3±211.7** | 22.2±0.05** |
Table 1: Biochemical analysis of the effect of three different potential hepatoprotective preparations in CCl4 induce acute liver injury. Values represent the mean ± SD of 10 determinations.*p<0.001 vs healthy control; §p<0.05 vs. untreated CCl4 group; **p<0.05 vs silymarin- and YHKtreated CCl4 group.
As compared to the normal control group, the animal treated with the CCl4 showed significant (p<0.01) decrease in the level of GSH and increase of LPO. Such changes were partially but significantly mitigated by either silymarin and YHK (p<0.05). YHK seemed to exert the least protective effect but this didn’t reach a statistically significant difference when compared to silymarin.
Histological changes
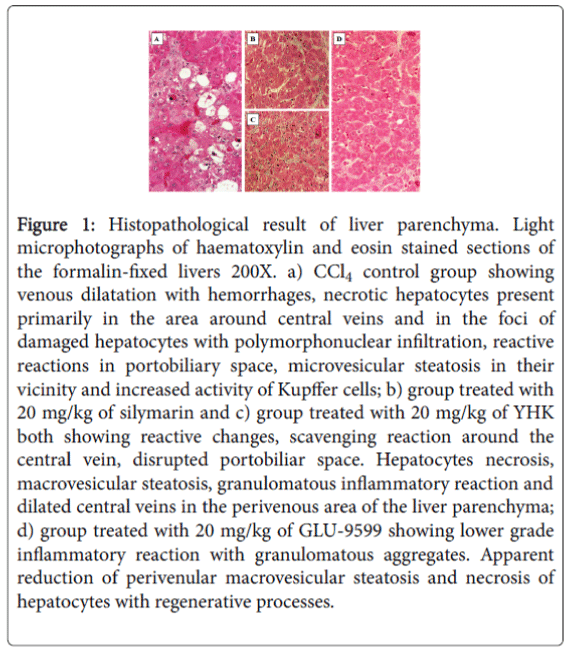
CCl4 induced liver injury occurs rapidly. In this study, hepatic necrosis was seen in the lobular regions 6 h after the administration of CCl4 (in-house data). Hepatocytes necrosis, macrovesicular steatosis, granulomatous inflammatory reaction, disrupted portobiliar space and dilated central veins were observed in the untreated CCl4 group (Table 2, Figure1A, p<0.0001 vs. healthy control). The changes with either silymarin and YHK provided some protection from the most severe inflammation but the histological scores were not significantly different from untreated animals (Figures 1B and 1C).
| Histological parameters | Healthy control | CCl4* | CCl4 + sylimarin § | CCl4 + YHK§ | CCl4 + GLU-9599 ** |
|---|---|---|---|---|---|
| Steatosis | 0 | 5 | 2 | 3 | 1 |
| Necrosis | 0 | 5 | 2 | 3 | 1 |
| Inflammatory infiltrate | 0 | 4 | 2 | 3 | 1 |
| Portal reaction | 0 | 3 | 1 | 2 | 0 |
| Hepatocyte degeneration | 0 | 4 | 1 | 1 | 0 |
| Kupffer cell activity | 0 | 4 | 2 | 1 | 1 |
| Interstitial haemorrhage | 0 | 5 | 1 | 2 | 1 |
| Vasodilatation | 0 | 4 | 3 | 3 | 1 |
| Cholestasis | 0 | 3 | 0 | 1 | 0 |
Table 2: Histological scoring of the effect of three different potential hepatoprotective preparations in CCl4 induce acute liver injury. Semiquantitative evaluation of the changes in liver parenchyma (0=no changes, 6=maximum changes). *cumulative evaluation: p<0.001 vs. healthy control; § cumulative evaluation: p<0.05 vs. untreated CCl4 group; **cumulative evaluation: p<0.05 vs. Silymarin and YHK-treated CCl4 group.
Figure 1: Histopathological result of liver parenchyma. Light microphotographs of haematoxylin and eosin stained sections of the formalin-fixed livers 200X. a) CCl4 control group showing venous dilatation with hemorrhages, necrotic hepatocytes present primarily in the area around central veins and in the foci of damaged hepatocytes with polymorphonuclear infiltration, reactive reactions in portobiliary space, microvesicular steatosis in their vicinity and increased activity of Kupffer cells; b) group treated with 20 mg/kg of silymarin and c) group treated with 20 mg/kg of YHK both showing reactive changes, scavenging reaction around the central vein, disrupted portobiliar space. Hepatocytes necrosis, macrovesicular steatosis, granulomatous inflammatory reaction and dilated central veins in the perivenous area of the liver parenchyma; d) group treated with 20 mg/kg of GLU-9599 showing lower grade inflammatory reaction with granulomatous aggregates. Apparent reduction of perivenular macrovesicular steatosis and necrosis of hepatocytes with regenerative processes.
In particular, such changes were only partly mitigated in the groups which were administered silymarin and YHK (Table 2, Figures 1B and 1C, p<0.05 vs. untreated CCl4 group). On the contrary, a significant beneficial improvement of all the above morphological abnormalities with minimal histological injury was scored in the group which was administered GLU-9599 (Table 2, Figure 1D, p<0.01 vs. sylimarineand YHK-treated animals).
Electron microscopy
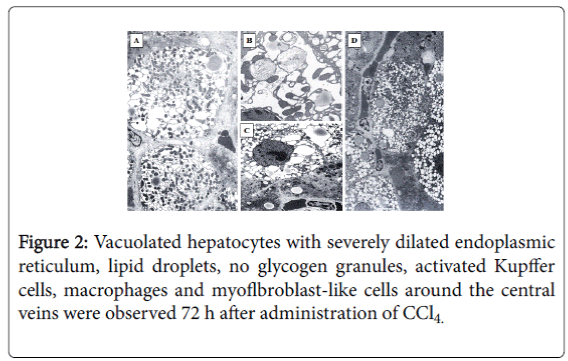
Normal control group showed a normal liver architecture, hepatocytes very well arranged, central and portal veins without alterations (not shown). Vacuolated hepatocytes with severely dilated endoplasmic reticulum, lipid droplets, no glycogen granules, activated Kupffer cells, macrophages and myoflbroblast-like cells around the central veins were observed 72 h after administration of CCl4 (Figure 2A). The group intoxicated with CCl4 and treated either with either silymarin or YHK had only minor prevention of the damage and the two treatments resulted to be quite comparable among them (Figures 2B and 2C, silymarin vs. YHK: p<0.09, not significant).
The group treated with GLU-9599 showed vacuolated hepatocytes with only a moderately dilated endoplasmic reticulum, many glycogen granules and ribosomes and reduced number of macrophages and hepatic stellate cells around the central veins without fibrosis as compared to rats treated with CCl4 alone, were observed (Figure 2D).
Discussion
CCl4 is metabolised in hepatocellular microsomes and generating a number of oxidative damage of lipid and protein cellular substrates. Indeed, this xenobiotic is a well established noxious compound affecting the generation of free radical oxidative [1-4]. This is because, under physiological conditions, there is an intricate interplay between multiple regulatory enzymes controlling the intracellular balance between the oxidized disulfide form and the reduced one depending on the intracellular need for activating conjugation reactions and extracellular requirements to decrease these phenomena [15]. When a proper cellular GSH concentration is lacking, several accumulating oxidative and nitrosative reactive intermediates such as peroxide, superoxide and peroxynitrite radicals, will invariably modify cellular lipidic macromolecules and enhance DNA adduct formation [15,16].
Some of our group had previously shown either in vitro and in vivo experimental studies that YHK, a known hepatoprotective compound in Japan, exerts a significant protective effect on hepatocellular damage and on liver microcirculation in an ischemia-reperfusion model [17,18] as well as exerting potent in vitro protective effect on metalinduced oxidative stress of hepatocytes [19]. Nonetheless, to date, there has been no further published experimental research or clinical study confirming its effectiveness. Moreover, any prove of a stringent batchto- batch control from producer raises some concern. This is often a crucial weakness in such kind of herbal compounds and may justify the virtual disappearance of YHK from the validated and properly published clinical scientific reports.
In the present study, administration of GLU-9599 decreased the CCl4 induced elevated enzyme levels in group V. This suggests the maintenance of structural integrity of hepatocytic cell membrane or regeneration of damage liver cells by the compound. Thus, it is evident from the present study that GLU-9599 has hepatoprotective effect in CCl4 induced liver damage. The hepatoprotective mechanism of this GSH-based compound requires further studies, although the high oral GSH bioavailability is likely to represent a major factor to be advocated for GLU-9599 proved to provide greater protection than silymarin and YHK preparation. This property seems to differentiate this formulation by other would-be oral GSH which indeed had failed to show any substantial benefit on redox balance [20].
The data, along with the most recent clinical pharmacokinetic studies [21] raise the possibility that this agent may be applied in the management of human alcoholic liver disease and those subjects taking multiple medications burdening liver metabolization mechanisms.
Acknowledgments and Memorial
We greatly appreciate the unbiased research grant support from Olimed ltd, London, UK and from Milano Medical Center, Milano, Italy, whose grant support helped concluding the study.
This work is dedicated to prof. Marzatico whose brilliant mind played a crucial role in devising this compound and carried out the related fundamental pharmacokinetic studies.
References
- Zavodnik IB (2015) Mitochondrial dysfunction and compensatory mechanisms in liver cells during acute carbon tetrachloride-induced rat intoxication. Biomed Khim61:731-736
- Uehara T, Pogribny IP, Rusyn I (2014) The DEN and CCl4 -Induced Mouse Model of Fibrosis and Inflammation-Associated Hepatocellular Carcinoma. Curr Protoc Pharmacol 66:1-14.
- Hafez MM, Al-Shabanah OA, Al-Harbi NO, Al-Harbi MM, Al-Rejaie SS, et al. (2014) Association between paraoxonases gene expression and oxidative stress in hepatotoxicity induced by CCl4. Oxid Med Cell Longev 2014: 893212.
- Bastien MC, Leblond F, Pichette V, Villeneuve JP (2000) Differential alteration of cytochrome P450 isoenzymes in two experimental models of cirrhosis. Can J Physiol Pharmacol 78: 912-919.
- Goeptar AR, Scheerens H, Vermeulen NP (1995) Oxygen and xenobiotic reductase activities of cytochrome P450. See comment in PubMed Commons below Crit Rev Toxicol 25: 25-65.
- Josan S, Billingsley K, Orduna J, Park JM, Luong R, et al. (2015) Assessing inflammatory liver injury in an acute CCl4 model using dynamic 3D metabolic imaging of hyperpolarized [1-(13) C]pyruvate. NMR Biomed28:1671-1677.
- Ariosto F, Riggio O, Cantafora A, Colucci S, Gaudio E, et al. (1989) Carbon tetrachloride-induced experimental cirrhosis in the rat: a reappraisal of the model. Eur Surg Res 21: 280-286.
- Smejkalová J, Simek J, Rouchal J, Dvorácková I (1985) The time course of biochemical and histological changes following carbon tetrachloride-induced liver damage in rats of both sexes. Physiol Bohemoslov34:494-501.
- Wu J, Danielsson A, Zern MA (1999) Toxicity of hepatotoxins: new insights into mechanisms and therapy. Expert Opin Investig Drugs 8: 585-607.
- Diaz-Vivancos P, de Simone A, Kiddle G, Foyer CH (2015) Glutathione--linking cell proliferation to oxidative stress. Free Radic Biol Med 89: 1154-1164.
- Hernández LE, Sobrino-Plata J, Montero-Palmero MB, Carrasco-Gil S, Flores-Cáceres ML, et al. (2015) Contribution of glutathione to the control of cellular redox homeostasis under toxic metal and metalloid stress. J Exp Bot 66:2901-2911.
- Weldy CS, Luttrell IP, White CC, Morgan-Stevenson V, Cox DP, et al. (2013) Glutathione (GSH) and the GSH synthesis gene Gclm modulate plasma redox and vascular responses to acute diesel exhaust inhalation in mice. Inhal Toxicol25:444-454.
- Hissin PJ, Hilf R (1976) A fluorometric method for determination of oxidized and reduced glutathione in tissues. Anal Biochem 74: 214-226.
- Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95: 351-358.
- Kalinina EV, Chernov NN, Novichkova MD (2014) Role of glutathione, glutathione transferase, and glutaredoxin in regulation of redox-dependent processes. Biochemistry (Mosc) 79:1562-1583.
- Huang KP, Huang FL (2002) Glutathionylation of proteins by glutathione disulfide S-oxide. Biochem Pharmacol 64: 1049-1056.
- Marotta F, Rouge A, Harada M, Anzulovic H, Idéo GM, et al. (2001) Beneficial effect of a controlled Chinese herbal remedy, K-17-22, in CCl4-induced liver toxicity: an in vivo and in vitro study. Biomed Res 22:167-174.
- Marotta F, Bertuccelli J, Albergati F, Harada M, Safran P, et al. (2001) Ischemia-reperfusion liver injury: effect of a nutritional approach with K-17.22 on hepatic antioxidant defense system. Biomed Res 22: 221-227.
- Marotta F, Lecroix P, Harada M, Masulair K, Safran P, et al. (2006) Liver exposure to xenobiotics: the aging factor and potentials for functional foods. Rejuvenation Res 9: 338-341.
- Allen J, Bradley RD (2011) Effects of Oral Glutathione Supplementation on Systemic Oxidative Stress Biomarkers in Human Volunteers. J Altern Complemen Med 17:827-833.
- Buonocore D, Grosini M, Giardina S, Michelotti A, Carrabetta M, et al. (2016) Bioavailability Study of an Innovative Orobuccal Formulation of Glutathione. Oxid Med Cell Longev 2016: 3286365.
Relevant Topics
- Constipation
- Digestive Enzymes
- Endoscopy
- Epigastric Pain
- Gall Bladder
- Gastric Cancer
- Gastrointestinal Bleeding
- Gastrointestinal Hormones
- Gastrointestinal Infections
- Gastrointestinal Inflammation
- Gastrointestinal Pathology
- Gastrointestinal Pharmacology
- Gastrointestinal Radiology
- Gastrointestinal Surgery
- Gastrointestinal Tuberculosis
- GIST Sarcoma
- Intestinal Blockage
- Pancreas
- Salivary Glands
- Stomach Bloating
- Stomach Cramps
- Stomach Disorders
- Stomach Ulcer
Recommended Journals
Article Tools
Article Usage
- Total views: 16415
- [From(publication date):
August-2016 - Aug 28, 2025] - Breakdown by view type
- HTML page views : 15300
- PDF downloads : 1115