Beyond Proteinuria: Apoptosis and Vitamin D3 State in Children with Diabetic Nephropathy
Received: 04-Feb-2016 / Accepted Date: 15-Mar-2016 / Published Date: 18-Mar-2016 DOI: 10.4172/2161-0681.1000266
Abstract
Diabetic nephropathy (DN) is the leading cause of death in patients with diabetes mellitus type I (T1D). Formation of diabetic nephropathy depends on the quality of blood glucose control. The aim of the study was to investigate the level of metabolic-hypoxic disorders, condition of apoptosis controlling system and Vitamin D3 levels in children with diabetic nephropathy. The study involved 36 children with type I diabetes and diabetic nephropathy (aged 6 to 17 years). The affinity of haemoglobin to oxygen determined by spectrophotometric method. The levels of the marker of cellular hypoxia HIF-1alfa, levels of the apoptotic factor caspase-3 studied by Western Blotting, Vitamin D3 levels were analyzed using ELISA assay. In the group of children with newly diagnosed T1D an increased rate of dissociation of oxygen and haemoglobin compared to the control group was detected. Children with DN the levels of this index were significantly lower than the control group. We show stage-dependent manner in increase of the cellular hypoxia and apoptotic effector caspase-3 levels. Both markers detected at significantly higher rate in DN patients as compared to T1D. Vitamin-D3 deficiency is documented in patients with DN. Thus, the development of DN in children is associated with violation of Hb/oxygen dissociation, a sign of the high degree of the Hb glycosylation resulting to the formation of cellular hypoxia and activation of apoptosis, as one of the basic mechanisms of cell damage kidneys in DN. All mentioned above disorders accompanied by Vitamin D3 deficiency.
Keywords: Diabetes mellitus; Diabetic nephropathy; Metabolic; Cellular hypoxia; Apoptosis; Vitamin D3
312872Introduction
Diabetes mellitus type I (T1D) nowadays is a major public health problem in the world. According to recent data, about 140 million people worldwide suffer from diabetes. The main complication of the T1D is a diabethis nephropathy (DN). The main biochemical disorder in diabetes and DN is hyperglycemia, causing a toxic effect of activating a chain of different intracellular enzymatic reactions, apoptosis, inflammation, oxidative stress. [1-3]. Adequate control of blood glucose in patients with diabetes is a critical aspect of therapy. Glucose present in the blood in excessive concentrations can enzymatically bind to proteins such as hemoglobin and causes glycosylation. The level of glycosylated hemoglobin formation depends on the concentration of glucose in the environment. People with high blood glucose levels will have higher levels of glycated hemoglobin. Non-enzymatic glycation that occurs as a result of spontaneous interaction between glucose and amino groups of proteins, leading to the formation of products of advanced glycation (AGA) [3,4].
Tubulointerstitial fibrosis and tubular atrophy appear to be better predictors of renal disease progression than glomerular pathology than glomerular disorders. Nephrons from proteinuric diabetic patients show that 71% of glomeruli display glomerulotubular junction abnormalities and 8-17% of glomeruli are atubular glomeruli [5].
The mechanisms underlying tubular atrophy in diabetic patients are incompletely understood. In vitro and in vivo studies have shown that high glucose (HG) concentrations are associated with increased reactive oxygen species (ROS) production, inflammation which inhibits proximal tubular function and induces apoptosis. Apoptosis has been detected in renal proximal tubular cells (RPTCs) of diabetic mice and rats [5,6]. Moreover, in vitro data show the role of Vitamin D3 in rescuing kidney cells from apoptosis induced by glucose and albumin. These processes in pediatric patients with T1D and DN are not fully studied.
Aim of the study. The aim of the study was to investigate the level of metabolic-hypoxic disorders, condition of apoptosis controlling system and Vitamin D3 levels in children with diabetic nephropathy.
Materials and Methods
Patients
A survey of 36 patients’ children with T1Ds (aged 6 to 17 years) who were hospitalized in the Endocrinology unit on Children’s Clinical Hospital (Kyiv in 2014-2015) was done. Complex examination including conventional methods (inspection, monitoring blood pressure, blood tests, study of urinary sediment, renal ultrasound, etc.) was done to all patients. Other than conventional methods i.e. spectral analysis of the forms of hemoglobin, plasma levels of HIF-1alfa, caspase-3, Vitamin D3 was done. The control group included 13 healthy children. Study performed based on ethical standards National Ukrainian committee on human experimentation (National O.O. Bogomolets medical University) and on the Helsinki Declaration.
Spectrometry
The affinity of hemoglobin to oxygen was determined using spectrophotometric metod. Hemolysis of red blood cells was performed by freezing/melting of blood samples. Electronic hemoglobin spectra were recorded using a spectrophotometer T70 UV/VIS (PG Instruments Limited, UK). Quantitative evaluation of spectra was performed using the software Image J (NIH, USA).
Western blotting
Levels of the marker of cellular hypoxia HIF-1alfa and proapoptotic factor caspase-3 studied by immunoblotting. Briefly, proteins solubilized in Laemmli sample buffer were resolved in polyacrylamide gels by SDS-PAGE and transferred to a polyvinylidene difluoride membrane. Membranes were then blocked in 5% non-fat milk in TBST (136 mM NaCl, 10 mM Tris, 0.05% Tween 20) and immunoblotted using the caspase-3 and HIF-1α Ab (Cell Signaling Technology, Danvers, MA USA) and actin mouse mAb (BD, Lexington KY, USA) for 1 h at room temperature. The actin mouse mAb was used as a loading control. The protein bands were visualized by chemiluminescent substrate ECL. Quantification of the protein content was done by densitometric analysis. Vitamin D3 levels were studied using ELISA assay using commercially available kit (Vitamin D3 (human) ELISA kit (BioVision, USA).
Statistical Analysis
Material processed using the methods of variation statistics (STATISTICA 6.0) and nonparametric statistical approaches (Mann- Whitney test). Results are presented as Mean ± SEM, was considered statistically significant level of P<0.05.
Results
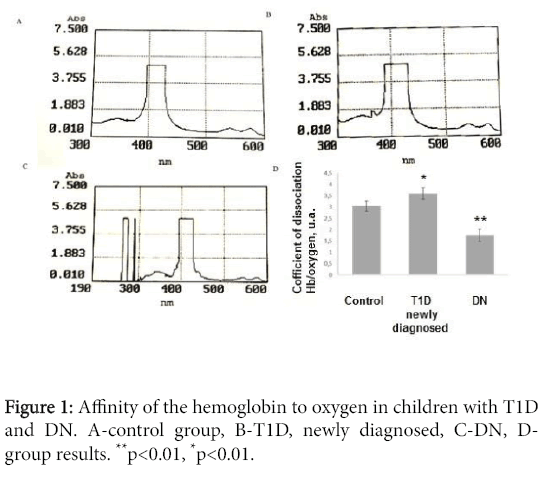
Depending on the general clinical data, all patients were divided into groups (Table 1). In all children the levels of hemoglobin affinity to oxygen were studied based on Sore peak analysis. Children from control show this index at the level 3.05 ± 0.23 a.u. In the group of children with newly diagnosed diabetes hemoglobin/ogygen dissociation was enhanced in comparison to control group -3.61 ± 0.25 a.u. (p<0.05). In the group of children diabetic nephropathy the level of index was documented at level 1.76 ± 0.27 a.u. (p<0.01, compared control group (Figure 1).
| Parameter | T1D, firstly diagnosed | DN | Control |
|---|---|---|---|
| Age | 13±5 (11-16) | 12±4 (12-16) | 12±5 (11-17) |
| Boys/girls | 12-Oct | 8-Jun | 6-Jul |
| Body weight, kg | 55±2 (49-60) | 52±4 (28-61) | 61±4 (35-71) |
| Hight, cm | 161±6 (157-171) | 163±4 (155-174) | 163±5 (159-171) |
| Serum creatinine, mcmol/mL | 41±4 (36-49) | 59±5 (45-73) | 40±4 (34-45) |
| Proteinuria, mg/day | Absent | 250±40 (170-360) | Absent |
| Gltcosylatedhemogobin, % | 4±3 (2-6) | 11±7 (9-14) | 4±3 (2-5) |
| Systolic blood pressure, mmHg | 110±4 (100-120) | 122±20 (115-140) | 112±7 (100-118) |
| Diastolic blood pressure, mmHg | 88±3(68-89) | 100±12 (92-116) | 88±3 (68-87) |
Table 1: Clinical characteristics of patients.
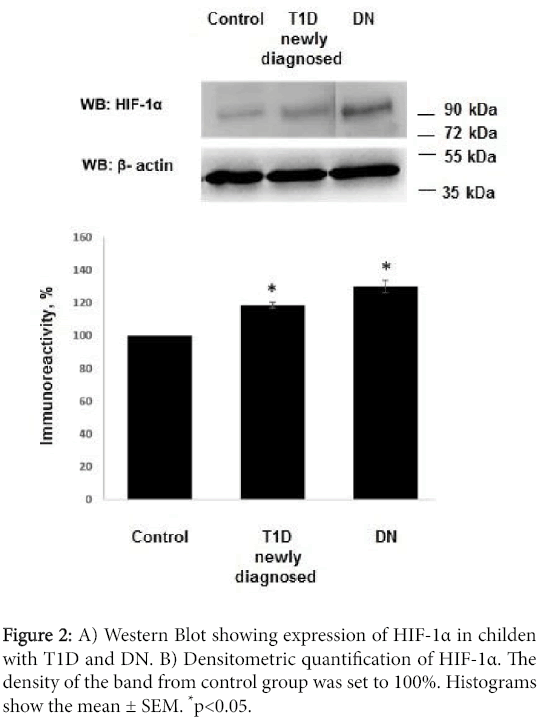
We have detected an increased level of marker of cellular hypoxia (HIF-1 α) in blood serum of all patients with T1D and DN as compared to control group. The levels of HIF-1α in group with T1D exceeded control group by 18.5 ± 1.76% (p<0.01 compared to control group) and by 30.03 ± 3.75% (?<0.01 compared to control group) in patients with DN (Figure 2).
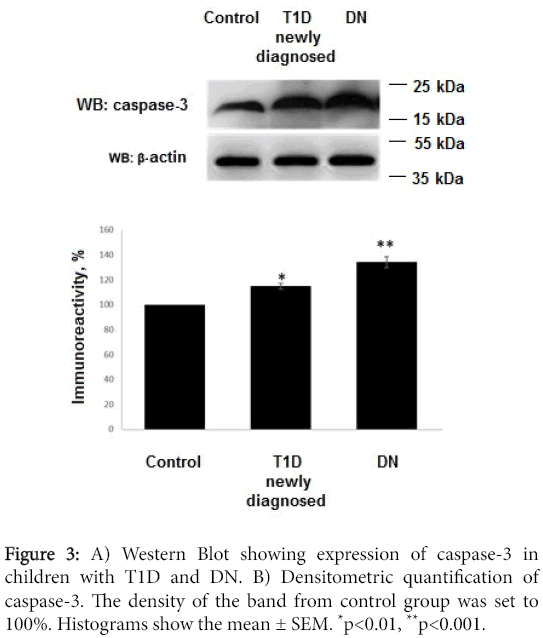
We studied the levels of apoptotic effector caspase-3. Increased levels of caspase-3 have been documented in all patients. In patients with DN expression of caspase-3 exceeded level of control group by 34.19 ± 4.36% (?<0,001). This marker in T1D group was documented at level exceeded control group by 14.82 ± 2.35% (?<0,01). Levels of caspase-3 in DN patients exceeded levels of T1D group. Control group marker assumed as 100% (Figure 3).
Normal level, insufficiency and deficiency of the Vitamin D3 were defined as ≥30 ng/mL, 21-29 ng/mL and ≤20 ng/mL, respectively. Endogenous levels of the Vitamin D3 are season-dependent. All patients have been recruited to the study during the September- Fabruary.
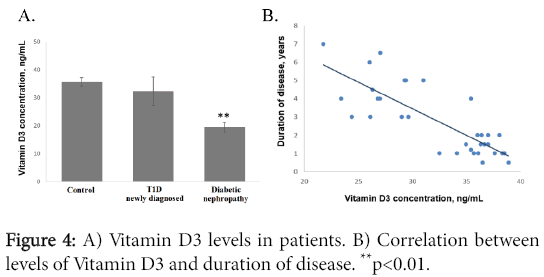
We show that the most prominent Vitamin D3 deficiency was detected in the group of patients with diabetic nephropathy. In control group Vitamin D3 was detected at level 35.68 ± 1.56 ng/mL, in patients with T1D-32.37 ± 5.1 ng/mL, in patients with diabetic nephropathy-19.39 ± 1.76 ng/mL (?<0.01 as compared to control group) (Figure 4A). Analysis of the Vitamin D3 levels and duration of the disease found negative correlation between them (R=-0.79, ?<0.001) (Figure 4B).
Discussion
It has been widely accepted, and histological studies have confirmed, that the deterioration of renal function in chronic kidney disease (CKD) better correlates with tubular and interstitial changes than with glomerular changes. Tubular dysfunction has been reported in the absence of microalbuminuria in patients with diabetes mellitus. Early tubular injury has been reported in patients with diabetes mellitus whose glomerular function is intact [7].
Chronic hypoxia of the tubulointerstitium has been recognized as a mechanism of progression that is common to many renal diseases. The hypoxic milieu in early stage diabetic nephropathy is aggravated by manifestations of chronic hyperglycemia-abnormalities of red blood cells, oxidative stress, sympathetic denervation of the kidney due to autonomic neuropathy, and diabetes-mellitus-induced tubular apoptosis; as such, tubulointerstitial hypoxia in diabetes mellitus might be an important early event [7-9].
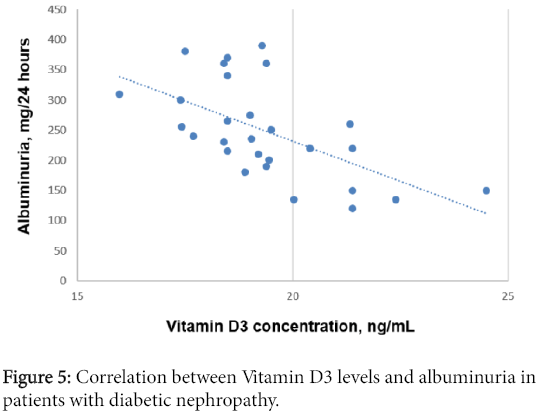
Analysis of the Vitamin D3 levels in relation to albuminuria levels in patients with diabetic nephropathy shows negative correlation (R=-0.59, ?<0.001) (Figure 5).
Here we show that patients with newly diagnised T1D have incresed levels of cellular hypoxia and its dramatic increase in patients with DN. At the same time chronic hypoxia is a potent inducer of apoptotic factors activation and cell death through apoptosis especially in tubular segment of nephrosis. We show the link between these two conditions-cellular hypoxia and apoptosis whereas the high levels of apoptotic effector caspase-3 activation both in T1D patients and DN patients was documented.
Hemoglobin belongs to the family of proteins containing prosthetic group and protoporphyrin IX (heme). In addition to transport oxygen hamohlobin performs a number of important functions: binding of nitric oxide and sulphides, interaction with reactive oxygen and nitrogen, and other sensory functions. As a result of hyperglycemia in diabetes there is an increase oxyhemoglobin formation and its transformed forms. Damage to the porphyrin ring followed by degradation of heme appears. Heme degradation is carried out in two stages and includes heme iron oxidation and nitration of tyrosine residues. Nitration of hemoglobin leads to changes in protein conformation leading to changes of its affinity to oxygen [10].
Hemoglobin-the first protein for which it was shown the nonenzymatic glycosylation resulted in glycosylated hemoglobin (HbA1c) formation. Blood levels of HbA1c is a marker for disorders of glucose metabolism and/orls adequate control of blood glucose levels in diabetic patients [10,11]. In this study we found a significant increase of HbA1 in patients with diabetic nephropathy as compared to newly diagnosed T1D.
Hemoglobin glycosylation sites mostly are amino N-terminal amino acid valine in β-chains and ε-amino groups of lysine residues of some α- and β-chains. The presence of glucose molecule at the N-β-globin chains leads to the decrease of hemoglobin affinity to oxygen [12,13]. This phenomenon was shown by us in the group of patients with established diabetic nephropathy. In contrast, group of children with T1D have enhanced hemoglobin affinity to oxygen was enhanced. This might be an explanation that the organism provides a compensatory reaction after the general metabolic disorders induced by hyperglycaemia.
The kidney plays a central role in vitamin D3 metabolism and regulation of its circulating levels. Beyond its role in calcium and phosphate homeostasis, vitamin D is an important modulator of cellular proliferation, inflammation, differentiation, and immunity [14]. Vitamin D3 attenuates kidney injury by suppressing fibrosis, inflammation, and apoptosis, by inhibiting multiple pathways known to play a role in kidney injury, including the renin-angiotensinaldosterone system (RAAS), the nuclear factor-κB (NF-κB) [15], the transforming growth factor-β (TGF-β)/Smad [16].
It is known that renal inflammation is an essential pathological process in the evolution of diabetic nephropathy [17]. Several studies have shown the role of active vitamin D3 in the modulation of renal inflammation. The anti-inflammatory properties of active vitamin D and its analogues may be attributed to their ability to suppress the NF- κB pathway [18].
It has also been reported that an increased Bax/Bcl-2 ratio and the cleaved form of caspase-3, which are apoptotic markers, are reversed by paricalcitol treatment in gentamicin-induced kidney injury [19]. Here we show that patients with diabetic nephropathy have increased activity of caspase-3. In addition, D3 prevents puromycin aminonucleoside-induced apoptosis of glomerular podocytes by activating the phosphatidylinositol 3-kinase/Akt-signalling pathway.
In this study we show that children with diabetic nephropathy have Vitamin D3 deficiency, activation of the apoptotic effector caspase-3, and high level of cellular hypoxia due to the disorders in dissociation of the Hb/O2. We speculate that negative correlation between Vitamin D3 levels and albuminuria is an evidence that adequate levels of Vitamin D3 provides protective effects on the glomerular barrier in children with diabetic nephropathy possibly due to its anti-apoptotic and anti-inflammatory activity. These aspects are perspective in terms of further investigation of the vitamin D3 system in children with diabetic nephropathy especially from the point of view on it’s possible anti-apoptotic effects.
References
- (2015) American diabetes association standartsof medical care in diabetes. The journal of clinical and applied research and education.
- Atkins RC, Zimmet P; World Kidney Day Steering Committee (2010) Diabetic kidney disease: act now or pay later. J Am SocHypertens 4: 3-6.
- Forbes JM, Thallas V, Thomas MC, Founds HW, Burns WC, et al. (2003) The breakdown of preexisting advanced glycation end products is associated with reduced renal fibrosis in experimental diabetes. FASEB J 17: 1762-1764.
- Goldin A, Beckman JA, Schmidt AM, Creager MA (2006) Advanced glycation end products: sparking the development of diabetic vascular injury. Circulation 114: 597-605.
- Heilig CW, Deb DK, Abdul A, Riaz H, James LR, et al. (2013) GLUT1 regulation of the pro-sclerotic mediators of diabetic nephropathy. Am J Nephrol 38: 39-49.
- Franko B, Brault J, Jouve T, Beaumel S, Benhamou PY, et al. (2014) Differential impact of glucose levels and advanced glycation end-products on tubular cell viability and pro-inflammatory/profibrotic functions. BiochemBiophys Res Commun 451: 627-631.
- Reidy K, Kang HM, Hostetter T, Susztak K (2014) Molecular mechanisms of diabetic kidney disease. J Clin Invest 124: 2333-2340.
- Rojas A, Morales MA (2004) Advanced glycation and endothelial functions: a link towards vascular complications in diabetes. Life Sci 76: 715-730.
- Shapiro R, McManus MJ, Zalut C, Bunn HF (1980) Sites of nonenzymatic glycosylation of human hemoglobin A. J BiolChem 255: 3120-3127.
- Vinod PB (2012) Pathophysiology of diabetic nephropathy. Clinical Queries: Nephrology 1: 121-126.
- Artyukhov VG (1995)Gemoproteidy: zakonomernostifotokhimicheskikhprevrashcheniy v usloviyakhrazlichnogomikrookruzheniya. Voronezh: IzdatelstvoVoronezhskogouniversitetata 280.
- Hayashi A, Suzuki T, Shin M (1973) An enzymic reduction system for metmyoglobin and methemoglobin, and its application to functional studies of oxygen carriers. BiochimBiophysActa 310: 309-316.
- Tan X, Li Y, Liu Y (2007) Therapeutic role and potential mechanisms of active Vitamin D in renal interstitial fibrosis. J Steroid BiochemMolBiol 103: 491-496.
- Park JW, Bae EH, Kim IJ, Ma SK, Choi C, et al. (2010) Renoprotective effects of paricalcitol on gentamicin-induced kidney injury in rats. Am J Physiol Renal Physiol 298: F301-313.
- Park JW, Bae EH, Kim IJ, Ma SK, Choi C, et al. (2010) Paricalcitol attenuates cyclosporine-induced kidney injury in rats. Kidney Int 77: 1076-1085.
- Segerer S, Nelson PJ, Schlöndorff D (2000) Chemokines, chemokine receptors, and renal disease: from basic science to pathophysiologic and therapeutic studies. J Am SocNephrol 11: 152-176.
- Guijarro C, Egido J (2001) Transcription factor-kappa B (NF-kappa B) and renal disease. Kidney Int 59: 415-424.
- Suh SH, Lee KE, Park JW, Kim IJ, Kim O, et al. (2013) Antiapoptotic Effect of Paricalcitol in Gentamicin-induced Kidney Injury. Korean J PhysiolPharmacol 17: 435-440.
- Xiao H, Shi W, Liu S, Wang W, Zhang B, et al. (2009) 1,25-Dihydroxyvitamin D(3) prevents puromycinaminonucleoside-induced apoptosis of glomerular podocytes by activating the phosphatidylinositol 3-kinase/Akt-signaling pathway. Am J Nephrol 30: 34-43.
Citation: Burlaka I (2016) Beyond Proteinuria: Apoptosis and Vitamin D3 State in Children with Diabetic Nephropathy. J Clin Exp Pathol 6:266. DOI: 10.4172/2161-0681.1000266
Copyright: © 2016 Burlaka I, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Select your language of interest to view the total content in your interested language
Share This Article
Recommended Journals
Open Access Journals
Article Tools
Article Usage
- Total views: 12595
- [From(publication date): 4-2016 - Aug 23, 2025]
- Breakdown by view type
- HTML page views: 11619
- PDF downloads: 976